Abstract
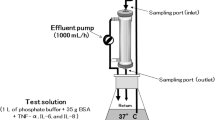
Background: Extracorporeal photopheresis (ECP) is an effective immunomodulatory therapy for various diseases. Autologous leukocytes are collected, photoactivated with a photosensitizer (8–methoxypsoralen, 8–MOP) and UVA light, and subsequently reinfused back to the patient. Leukapheresis and UVA rradiation systems can be integrated into one device (inline) or handled by two separate devices (offline) ECP works via intercalation of 8–MOP into DNA helices and UVA-based interactions to inhibit DNA replication. 8–MOP is known to adhere to plastic materials, which might reduce its availability for intercalation In the present study we examined the bioavailability of 8–MOP when different plastic materials and solvents are used as matrices. Methods: Varying amounts of shredded ethylene vinyl acetate (EVA) and poly-vinylchloride (PVC) from the MacoGenic irradiation bag (EVA1), UVA PIT irradiation bag (EVA2), UVA PIT recirculation bag (PVC A) and UVA PIT tubing (PVC B) by MacoPharma and PIT Medical Systems, respectively, were ncubated with 200 ng mL-1 8–MOP dissolved in diisopropyl ether (DIPE) plus toluene 90/10 vol%, deionized water or plasma. After 2 h 8–MOP concentrations were determined by GC-MS. Results: After incubation, 8-MOP concentrations varied depending on the amount and type of plastic (PVC > EVA) and solvent (water > plasma > DIPE/toluene). Absorption to 200 mg EVA1 or EVA2 resulted in 8–MOP concentrations of 57% or 32% in water, 91% or 80% in plasma, and 93% or 92% in DIPE/toluene, while 200 mg PVC A and PVC B yielded recovery rates of 26% and 10% in water, 76% and 75% in plasma, and 55% and 30% in DIPE/toluene, respectively. Remaining 8–MOP differed significantly between container materials (EVA vs. PVC; p < 0.022) but not manufacturers (MacoPharma vs. PIT Medical Systems). Conclusion: Absorption loss of 8–MOP depends on the type of plastic and solvent and is more pronounced with water than with plasma. As the DNA binding effect of 8–MOP is dose-dependent, ECP starting doses should be adjusted to ensure that a sufficient concentration of free bioavailable 8–MOP is present during UV irradiation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Schwartz, A. Padmanabhan, N. Aqui, R. A. Balogun, L. Connelly-Smith, M. Delaney, N. M. Dunbar, V. Witt, Y. Wu and B. H. Shaz, J. Clin. Apher., 2016, 31, 149–162.
J. Adamski, T. Kinard, T. Ipe and L. Cooling, Transfus. Apher. Sci., 2015, 52, 171–182.
A. Alfred, P. C. Taylor, F. Dignan, K. El-Ghariani, J. Griffin, A. R. Gennery, D. Bonney, E. Das-Gupta, S. Lawson, R. K. Malladi, K. W. Douglas, T. Maher, J. Guest, L. Hartlett, A. J. Fisher, F. Child and J. J. Scarisbrick, Br. J. Haematol, 2017, 177, 287–310.
P. S. Song and K. J. Tapley Jr., Photochem. Photobiol., 1979, 29, 1177–1197.
H. Cao, J. E. Hearst, L. Corash and Y. Wang, Anal. Chem., 2008, 80, 2932–2938.
G. D. Cimino, H. B. Gamper, S. T. Isaacs and J. E. Hearst, Annu. Rev. Biochem., 1985, 54, 1151–1193.
T. Bessho, J. Biol Chem., 2003, 278, 5250–5254.
D. Schmid, C. Grabmer, D. Streif, T. Lener, K. Schallmoser and E. Rohde, Vox Sang., 2015, 108, 82–88.
C. Franklin, E. Cesko, U. Hillen, B. Schilling and S. Brandau, PLoS One, 2015, 10, e0134518.
Senate Commission on Food Safety (SKLM) of the German Research Foundation (DFG), Mol Nutr. Food Res., 2007, 51, 367–373.
S. E. Shephard, F. O. Nestle and R. Panizzon, Photodermatol., Photoimmunol Photomed., 1999, 15, 64–74.
J. Kruger, E. Christophers and M. Schlaak, Br. J. Dermatol., 1978, 98, 141–144.
S. I. Smith and J. S. Brodbelt, Analyst, 2010, 135, 943–952.
J. Gazith, W. Schalla and H. Schaefer, Arch. Dermatol. Res., 1978, 263, 215–222.
J. Malakova, P. Zak, I. Jokesova, P. Zivny, M. Blazek, A. Zavrelova and V. Palicka, Chromatographia, 2009, 69, 779–783.
H. Ehrsson, S. Eksborg, I. Wallin, N. Kallberg and G. Swanbeck, J. Chromatogr., 1977, 140, 157–164.
A. D. Buhimschi and F. P. Gasparro, Photochem. Photobiol., 2014, 90, 241–246.
J. Li, Q. H. Zhang, J. He, E. W. Liu, X. M. Gao and Y. X. Chang, Sci. World J., 2015, 2015, 365093.
L. Karolak, M. Tod, A. Leon, A. M. Heudes, O. Petitjean and L. Laroche, PhotoDermatol., Photoimmunol Photomed., 1992, 9, 58–60.
V. Hähnel, F. Dormann, A. Nitsopoulos, A. Friedle and N. Ahrens, Photochem. Photobiol Sci., 2017, 16, 193–200.
M. Laulhe, S. Lefebvre, D. Le Broc-Ryckewaert, M. Pierre, A. Ferry and B. Delorme, PLoS One, 2019, 14, e0212835.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Hähnel, V., Weber, I., Tuemmler, S. et al. Matrix-dependent absorption of 8–methoxypsoralen in extracorporeal photopheresis. Photochem Photobiol Sci 19, 1099–1103 (2020). https://doi.org/10.1039/c9pp00378a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00378a