Abstract
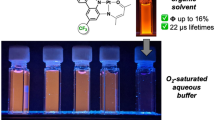
We report the first exocyclically metallated tetrapyridinoporphyrazine, [tetrakis-(trans-Pt(NH3)2Cl)-tetra(3,4-pyrido)porphyrazine-zinc(II)](NO3)4 (4), synthesized in a multistep synthesis starting from 3,4-pyridinedicarbonitrile (1). The synthetic procedure involved a platination reaction of the intermediate tetra(3,4-pyrido) porphyrazine-zinc(II) (2), whereby the zinc(II) enhanced the solubility of the intermediate enabling the platination reaction. A similar approach to synthesize [tetrakis-(trans-Pt(NH3)2Cl)-tetra(3,4-pyrido)porphyrazine] (NO3)4 (5) failed due to the unsuitable solubility properties of the intermediate tetra(3,4-pyrido)porphyrazine (3). The final product 4 and the intermediates were characterized, the photochemical and photophysical properties were determined and the photocytotoxicities were investigated. We demonstrate that the platinated tetra-pyridinoporphyrazine 4 is a potential photosensitizer for photodynamic therapy (PDT)
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
L. M. Moreira, F. V. dos Santos, J. P. Lyon, M. Maftoum- Costa, C. Pacheco-Soares and N. S. da Silva, Photodynamic therapy: Porphyrins and phthalocyanines as Photosensitizers, Aust. J. Chem., 2008, 61, 741–754.
I. Yoon, J. Z. Li and Y. K. Shim, Advance in photosensitizers and light delivery for photodynamic therapy, Clin. Endosc., 2013, 46, 7–23.
Z. Jiang, J. Shao, T. Yang, J. Wang and L. Jia, Pharmaceutical development, composition and quantitative analysis of phthalocyanine as the photosensitizer for cancer photodynamic therapy, J. Pharm. Biomed. Anal., 2014, 87, 98–104.
Y. Li, J. Wang, X. Zhang, W. Guo, F. Li, M. Yu, X. Kong, W. Wu and Z. Hong, Highly water-soluble and tumor-targeted photosensitizers for photodynamic therapy, Org. Biomol. Chem., 2015, 13, 7681–7694.
P. M. Antoni, A. Naik, I. Albert, R. Rubbiani, S. Gupta, P. Ruiz-Sanchez, P. Munikorn, J. M. Mateos, V. Luginbuehl, P. Thamyongkit, U. Ziegler, G. Gasser, G. Jeschke and B. Spingler, (Metallo)porphyrins as Potent Phototoxic Anti- Cancer Agents after Irradiation with Red Light, Chem. – Eur. J., 2015, 21, 1179–1183.
J. M. Dabrowski, B. Pucelik, A. Regiel-Futyra, M. Brindell, O. Mazuryk, A. Kyziol, G. Stochel, W. Macyk and L. G. Arnaut, Engineering of relevant photodynamic processes through structural modifications of metallotetrapyrrolic photosensitizers, Coord. Chem. Rev., 2016, 325, 67–101.
D. van Straten, V. Mashayekhi, H. S. de Bruijn, S. Oliveira and D. J. Robinson, Oncologic Photodynamic Therapy: Basic Principles, Current Clinical Status and Future Directions, Cancers, 2017, 9, 19.
J. Zhang, C. Jiang, J. P. Figueiró Longo, R. B. Azevedo, H. Zhang and L. A. Muehlmann, An updated overview on the development of new photosensitizers for anticancer photodynamic therapy, Acta Pharm. Sin. B, 2018, 8, 137–146.
S. G. Bown, Photodynamic therapy for photochemists, Philos. Trans. R. Soc., A, 2013, 371, 20120371.
K. Ishii, Functional singlet oxygen generators based on phthalocyanines, Coord. Chem. Rev., 2012, 256, 1556–1568.
H. Abrahamse and M. R. Hamblin, New photosensitizers for photodynamic therapy, Biochem. J., 2016, 473, 347–364.
J.-Y. Liu, X.-J. Jiang, W.-P. Fong and D. K. P. Ng, Highly photocytotoxic 1,4-dipegylated zinc(II) phthalocyanines. Effects of the chain length on the in vitro photodynamic activities, Org. Biomol. Chem., 2008, 6, 4560–4566.
D. E. J. G. J. Dolmans, D. Fukumura and R. K. Jain, Photodynamic therapy for cancer, Nat. Rev. Cancer, 2003, 3, 380–387.
A. Naik, R. Rubbiani, G. Gasser and B. Spingler, Visible- Light-Induced Annihilation of Tumor Cells with Platinum– Porphyrin Conjugates, Angew. Chem., Int. Ed., 2014, 53, 6938–6941.
A. Oniszczuk, K. A. Wojtunik-Kuleszaa, T. Oniszczuk and K. Kasprzak, The potential of photodynamic therapy (PDT)- Experimental investigations and clinical use, Biomed. Pharmacother., 2016, 83, 912–929.
L. Hassani, F. Hakimian, E. Safaei and Z. Fazeli, Antibacterial effect of cationic porphyrazines and anionic phthalocyanine and their interaction with plasmid DNA, J. Mol. Struct., 2013, 1052, 221–227.
M. Machacek, J. Demuth, P. Cermak, M. Vavreckova, L. Hruba, A. Jedlickova, P. Kubat, T. Simunek, V. Novakova and P. Zimcik, Tetra(3,4-pyrido)porphyrazines Caught in the Cationic Cage: Toward Nanomolar Active Photosensitizers, J. Med. Chem., 2016, 59, 9443–9456.
M. P. Donzello, E. Viola, X. Cai, L. Mannina, C. Ercolani and K. M. Kadish, Tetra-2,3-pyrazinoporphyrazines with Externally Appended Pyridine Rings. 8. Central (ZnII, CuII, MgII(H2O), CdII) and Exocyclic (PdII) Metal Ion Binding in Heteropentametallic Complexes from Tetrakis-2,3-[5,6- di(2-pyridyl)pyrazino]porphyrazine, Inorg. Chem., 2010, 49, 2447–2456.
I. Manet, F. Manoli, M. P. Donzello, E. Viola, A. Masi, G. Andreano, G. Ricciardi, A. Rosa, L. Cellai, C. Ercolani and S. Monti, Pyrazinoporphyrazines with Externally Appended Pyridine Rings. 13. Structure, UV-Visible Spectral Features, and Noncovalent Interaction with DNA of a Positively Charged Binuclear (ZnII/PtII) Macrocycle with Multimodal Anticancer Potentialities, Inorg. Chem., 2013, 52, 321–328.
M. P. Donzello, D. Vittori, D. Futur, Z. Fu, C. Ercolani and K. M. Kadish, Tetra-2,3-pyrazinoporphyrazines with externally appended pyridine rings. 14. UV-visible spectral and electrochemical behavior of homo/heterobinuclear neutral and hexacationic macrocycles, J. Porphyrins Phthalocyanines, 2013, 17, 896–904.
M. P. Donzello, F. Gigante, F. Sciscione, E. Viola and K. M. Kadish, Tetra-2,3-pyrazinoporphyrazines with externally appended pyridine rings. 18. Physicochemical properties and photochemical behavior of new uncharged water soluble low-symmetry macrocycles [{Pd(OAc)2}3(PtCl2)LM] (M = MgII(H2O), ZnII, PdII), J. Porphyrins Phthalocyanines, 2017, 21, 334–344.
A.-M. Florea and D. Büsselberg, Cisplatin as an Anti-Tumor Drug: Cellular Mechanisms of Activity, Drug Resistance and Induced Side Effects, Cancers, 2011, 3, 1351–1371.
Y. Jung and S. J. Lippard, Direct cellular responses to platinum-induced DNA damage, Chem. Rev., 2007, 107, 1387–1407.
O. Vrana, V. Novohradsky, Z. Medrikova, J. Burdikova, O. Stuchlikova, J. Kasparkova and V. Brabec, Internalization of Ineffective Platinum Complex in Nanocapsules Renders It Cytotoxic, Chem. – Eur. J., 2016, 22, 2728–2735.
W. S. Szulbinski and J. R. Kincaid, Synthesis and spectroscopic characterization of zinc tetra(3,4-pyridine)porphyrazine entrapped within the supercages of Y-zeolite, Inorg. Chem., 1998, 37, 5014–5020.
C. Martí, S. Nonell, M. Nicolau and T. Torres, Photophysical properties of neutral and cationic tetrapyridinoporphyrazines, Photochem. Photobiol., 2000, 71, 53–59.
E. A. Dupouy, D. Lazzeri and E. N. Durantini, Photodynamic activity of cationic and non-charged Zn(II) tetrapyridinoporphyrazine derivatives: biological consequences in human erythrocytes and Escherichia coli, Photochem. Photobiol. Sci., 2004, 3, 992–998.
C. Ramirez, C. Antonacci, J. Ferreira and R. D. Sheardy, The facile synthesis and characterization of novel cationic metallated and nonmetallated tetrapyridino porphyrazines having different metal centers, Synth. Commun., 2004, 34, 3373–3379.
H. Tomoda, S. Saito, S. Ogawa and S. Shiraishi, Synthesis of Phthalocyanines from Phthalonitrile with Organic Strong Bases, Chem. Lett., 1980, 9, 1277–1280.
M. Obata, S. Hirohara, R. Tanaka, I. Kinoshita, K. Ohkubo, S. Fukuzumi, M. Tanihara and S. Yano, In Vitro Heavy- Atom Effect of Palladium(II) and Platinum(II) Complexes of Pyrrolidine-Fused Chlorin in Photodynamic Therapy, J. Med. Chem., 2009, 52, 2747–2753.
S. Fukuzumi, K. Ohkubo, X. Zheng, Y. Chen, R. K. Pandey, R. Zhan and K. M. Kadish, Metal Bacteriochlorins Which Act as Dual Singlet Oxygen and Superoxide Generators, J. Phys. Chem. B, 2008, 112, 2738–2746.
M. Bregnhøj, M. Westberg, F. Jensen and P. R. Ogilby, Solvent-dependent singlet oxygen lifetimes: temperature effects implicate tunneling and charge-transfer interactions, Phys. Chem. Chem. Phys., 2016, 18, 22946–22961.
L. Vachova, M. Machacek, R. Kucera, J. Demuth, P. Cermak, K. Kopecky, M. Miletin, A. Jedlickova, T. Simunek, V. Novakova and P. Zimcik, Heteroatom-substituted tetra (3,4-pyrido)porphyrazines: a stride toward near-infrared-absorbing macrocycles, Org. Biomol. Chem., 2015, 13, 5608–5612.
J. Piskorz, K. Konopka, N. Düzgünes, Z. Gdaniec, J. Mielcarek and T. Goslinski, Diazepinoporphyrazines Containing Peripheral Styryl Substituents and Their Promising Nanomolar Photodynamic Activity against Oral Cancer Cells in Liposomal Formulations, ChemMedChem, 2014, 9, 1775–1782.
P. Skupin-Mrugalska, W. Szczolko, P. Gierlich, K. Konopka, T. Goslinski, J. Mielcarek and N. Düzgünes, Physicochemical properties of liposome-incorporated 2-(morpholin-4-yl)ethoxy phthalocyanines and their photodynamic activity against oral cancer cells, J. Photochem. Photobiol., A, 2018, 353, 445–457.
N. Y. Shilyagina, N. N. Peskova, S. A. Lermontova, A. A. Brilkina, V. A. Vodeneev, A. V. Yakimansky, L. G. Klapshina and I. V. Balalaeva, Effective delivery of porphyrazine photosensitizers to cancer cells by polymer brush nanocontainers, J. Biophotonics, 2017, 10, 1189–1197.
D. L. Boger, B. E. Fink, S. R. Brunette, W. C. Tse and M. P. Hedrick, A simple, high-resolution method for establishing DNA binding affinity and sequence selectivity, J. Am. Chem. Soc., 2001, 123, 5878–5891.
R. P. Rastogi, S. P. Singh, D.-P. Häder and R. P. Sinha, Detection of reactive oxygen species (ROS) by the oxidant-sensing probe 2′,7′-dichlorodihydrofluorescein diacetate in the cyanobacterium Anabaena variabilis PCC 7937, Biochem. Biophys. Res. Commun., 2010, 397, 603–607.
S. S. Bharate, V. Kumar and R. A. Vishwakarma, Determining Partition Coefficient (Log P), Distribution Coefficient (Log D) and Ionization Constant (pKa) in Early Drug Discovery, Comb. Chem. High Throughput Screening, 2016, 19, 461–469.
V. Pierroz, T. Joshi, A. Leonidova, C. Mari, J. Schur, I. Ott, L. Spiccia, S. Ferrari and G. Gasser, Molecular and Cellular Characterization of the Biological Effects of Ruthenium(II) Complexes Incorporating 2-Pyridyl-2-pyrimidine- 4-carboxylic Acid, J. Am. Chem. Soc., 2012, 134, 20376–20387.
H. K. Moon, M. Son, J. E. Park, S. M. Yoon, S. H. Lee and H. C. Choi, Significant increase in the water dispersibility of zinc phthalocyanine nanowires and applications in cancer phototherapy, NPG Asia Mater., 2012, 4, e12.
M. S. Meijer, V. S. Talens, M. F. Hilbers, R. E. Kieltyka, A. M. Brouwer, M. M. Natile and S. Bonnet, NIR-Light- Driven Generation of Reactive Oxygen Species Using Ru(II)- Decorated Lipid-Encapsulated Upconverting Nanoparticles, Langmuir, 2019, 35, 12079–12090.
U. Michelsen, H. Kliesch, G. Schnurpfeil, A. K. Sobbi and D. Wöhrle, Unsymmetrically substituted benzonaphthoporphyrazines: A new class of cationic photosensitizers for the photodynamic therapy of cancer, Photochem. Photobiol., 1996, 64, 694–701.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c9pp00336c
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Schneider, L., Larocca, M., Wu, W. et al. Exocyclically metallated tetrapyridinoporphyrazine as a potential photosensitizer for photodynamic therapy. Photochem Photobiol Sci 18, 2792–2803 (2019). https://doi.org/10.1039/c9pp00336c
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00336c