Abstract
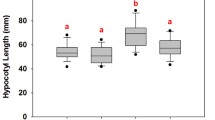
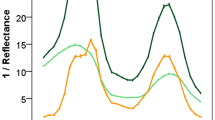
Blue light and UV radiation shape a plant’s morphology and development, but accession-dependent responses under natural conditions are unclear. Here we tested the hypothesis that two faba bean (Vicia faba L.) accessions adapted to different latitudes and altitudes vary in their responses to solar blue and UV light. We measured growth, physiological traits, phenolic profiles and expression of associated genes in a factorial experiment combining two accessions (Aurora, a Swedish cultivar adapted to high latitude and low altitude; ILB938, from the Andean region of Colombia and Ecuador, adapted to low latitude and high altitude) and four filter treatments created with plastic sheets: 1. transparent as control; 2. attenuated short UV (290–350 nm); 3. attenuated UV (290–400 nm); 4. attenuated blue and UV light. In both accessions, the exclusion of blue and UV light increased plant height and leaf area, and decreased transcript abundance of ELONGATED HYPOCOTYL 5 (HY5) and TYROSINE AMINOTRANSFERASE 3 (TAT3). Blue light and short UV induced the accumulation of epidermal and whole-leaf flavonoids, mainly quercetins, and the responses in the two accessions were through different glycosides. Filter treatments did not affect kaempferol concentration, but there were more tri-glycosides in Aurora and di-glycosides in ILB938. Furthermore, fewer quercetin glycosides were identified in ILB938. The transcript abundance was consistently higher in Aurora than in ILB938 for all seven investigated genes: HY5, TAT3, CHALCONE SYNTHASE (CHS), CHALCONE ISOMERASE (CHI), DON-GLUCOSYLTRANSFERASE 1 (DOGT1), ABA INSENSITIVE 2 (ABI2), AUXIN-INDUCIBLE 2–27 (IAA5). The two largest differences in transcript abundance between the two accessions across treatments were 132-fold in CHS and 30-fold in DOGT1 which may explain the accession-dependent glycosylation patterns. Our findings suggest that agronomic selection for adaptation to high altitude may favour phenotypes with particular adaptations to the light environment, including solar UV and blue light.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Chen, J. Chory and C. Fankhauser, Light Signal Transduction in Higher Plants, Annu. Rev. Genet., 2004, 38(1), 87–117, DOI: 10.1146/annurev.genet.38.072902.092259.
H. Smith, Phytochromes and light signal perception by plants—an emergingsynthesis, Nature, 2000, 407(6804), 585–591, DOI: 10.1038/35036500.
C. Lin, Plant blue-light receptors, Trends Plant Sci., 2000, 5(8), 337–342, DOI: 10.1016/S1360–1385(00)01687-3.
L. Rizzini, J. J. Favory, C. Cloix, et al., Perception of UV-B by the, Arabidopsis UVR8 protein, Science, 2011, 332(6025), 103–106, DOI: 10.1126/science.1200660.
J. J. Casal, Phytochromes, Cryptochromes, Phototropin: Photoreceptor Interactions in Plants, Photochem. Photobiol., 2007, 71(1), 1–11, DOI: 10.1562/0031-8655(2000) 0710001PCPPII2.0.CO2.
L. Ma, J. Li, L. Qu, et al., Light Control of, Arabidopsis Development Entails Coordinated Regulation of Genome Expression and Cellular Pathways, Plant Cell, 2001, 13, 2589–2607, DOI: 10.1105/tpc.010229.
A. Sancar, Structure and Function of DNA Photolyase and Cryptochrome Blue-Light Photoreceptors, Chem. Rev., 2003, 103(6), 2203–2237, DOI: 10.1021/cr0204348.
M. M. Caldwell and S. D. Flint, Stratospheric ozone reduction, solar UV-B radiation and terrestrial ecosystems, Clim. Change, 1994, 28(4), 375–394, DOI: 10.1007/BF01104080.
C. L. Ballaré, M. C. Rousseau, P. S. Searles, et al., Impacts of solar ultraviolet-B radiation on terrestrial ecosystems of Tierra del Fuego (southern Argentina). An overview of recent progress, J. Photochem. Photobiol., B, 2001, 62(1–2), 67–77, DOI: 10.1016/s1011-1344(01)00152-x.
N. D. Paul and D. Gwynn-Jones, Ecological roles of solar UV radiation: towards an integrated approach, Trends Ecol. Evol., 2003, 18(1), 48–55, DOI: 10.1016/S0169-5347(02)00014-9.
G. I. Jenkins, Signal transduction in responses to UV-B radiation, Annu. Rev. Plant Biol., 2009, 60, 407–431, DOI: 10.1146/annurev.arplant.59.032607.092953.
G. Fuglevand, J. A. Jackson and G. I. Jenkins, UV-B, UV-A, and Blue Light Signal Transduction Pathways Interact Synergistically to Regulate Chalcone Synthase Gene Expression in, Arabidopsis, Plant Cell Online, 1996, 8(12), 2347–2357, DOI: 10.1105/tpc.8.12.2347.
G. Agati and M. Tattini, Multiple functional roles of flavonoids in photoprotection, New Phytol., 2010, 186(4), 786–793, DOI: 10.1111/j.1469-8137.2010.03269.x.
J. B. Harborne and C. A. Williams, Advances in flavonoid research since 1992, Phytochemistry, 2000, 55(6), 481–504, DOI: 10.1016/S0031-9422(00)00235-1.
J.-J. Favory, A. Stec, H. Gruber, et al., Interaction of COP1 and UVR8 regulates UV-B-induced photomorphogenesis and stress acclimation in, Arabidopsis, EMBO J., 2009, 28(5), 591–601, DOI: 10.1038/emboj.2009.4.
H. K. Wade, T. N. Bibikova, W. J. Valentine and G. I. Jenkins, Interactions within a network of phytochrome, cryptochrome and UV-B phototransduction pathways regulate chalcone synthase gene expression in, Arabidopsis leaf tissue, Plant J., 2002, 25(6), 675–685, DOI: 10.1046/j.1365-313x.2001.01001.x.
S. N. Gangappa and J. F. Botto, The Multifaceted Roles of HY5 in Plant Growth and Development, Mol. Plant, 2016, 9(10), 1353–1365, DOI: 10.1016/J.MOLP.2016.07.002.
M. Zuk and M. Travisano, Models on the runway: how do we make replicas of the world?, Am. Nat., 2018, 192(1), 1–9, DOI: 10.1086/697508.
V. Caracuta, O. Barzilai, H. Khalaily, et al., The onset of faba bean farming in the Southern Levant, Sci. Rep., 2015, 5(1), 14370, DOI: 10.1038/srep14370.
D. A. Lawes, D. A. Bond and M. H. Poulsen, Classification, origin, breeding methods and objectives. Faba Bean 1983, pp. 23–76.
W. Link, T. J. Hocking and F. L. Stoddard, Evaluation of physiological traits for improving drought tolerance in faba bean (Vicia faba L.), Plant Soil, 2007, 292(1–2), 205–217.
J. Diamond and N. Y. London, Guns, Germs And Steel The Fates Of Human Societies. http://www.cloverport.kyschools.us/userfiles/3/Classes/308/Jared Diamond - Guns Germs and Steel.pdf.
H. Khazaei, W. Link, K. Street and F. L. Stoddard, Short Communication ILB 938, a valuable faba bean (Vicia faba L.), accession. 2018. DOI: 10.18730/60FD2.
S. Madronich, S. Flocke, J. Zeng, I. Petropavlovskikh and J. Lee-Taylor, Tropospheric Ultraviolet-Visible Model (TUV), 2002.
P. J. Aphalo, The r4photobiology suite, UV4Plants Bulletin, 2015, 21–29, DOI: 10.19232/uv4pb.2015.1.14.
A. Lindfors, A. Heikkilä, J. Kaurola, T. Koskela and K. Lakkala, Reconstruction of Solar Spectral Surface UV Irradiances Using Radiative Transfer Simulations, Photochem. Photobiol., 2009, 85(5), 1233–1239, DOI: 10.1111/j.1751-1097.2009.00578.x.
P. J. Aphalo, A. Albert, L. O. Björn, A. R. Mcleod, T. M. Robson and E. Rosenqvist, Beyond the visible: A handbook of best practice in plant UV photobiology. https://helda.helsinki.fi/bitstream/handle/10138/37558/Handbook_BeyondtheVisible.pdf;sequence=1.
W. S. Rasband, ImageJ. Version 1.46 r, US National Institutes of Health, Bethesda, MD, 2012.
Z. G. Cerovic, G. Masdoumier, N. B. Ghozlen and G. Latouche, A new optical leaf-clip meter for simultaneous non-destructive assessment of leaf chlorophyll and epidermal flavonoids, Physiol. Plant., 2012, 146(3), 251–260, DOI: 10.1111/j.1399-3054.2012.01639.x.
S. Schmidt, M. Zietz, M. Schreiner, S. Rohn, L. W. Kroh and A. Krumbein, Genotypic and climatic influences on the concentration and composition of flavonoids in kale (Brassica oleracea var. sabellica), Food Chem., 2010, 119(4), 1293–1299, DOI: 10.1016/j.foodchem.2009.09.004.
S. Schmidt, M. Zietz, M. Schreiner, S. Rohn, L. W. Kroh and A. Krumbein, Identification of complex, naturally occurring flavonoid glycosides in kale (Brassica oleracea, var. sabellica) by high-performance liquid chromatography diode-array detection/electrospray ionization multi-stage mass spectrometry, Rapid Commun. Mass Spectrom., 2010, 24(14), 2009–2022, DOI: 10.1002/rcm.4605.
S. Neugart and M. Schreiner, Identification of complex, naturally occurring flavonoid glycosides in, Vicia faba and, Pisum sativum, leaves by HPLC-DAD-ESI-MSn and the genotypic effect on their flavonoid profile, Food Res. Int., 2015, 76, 114–121, DOI: 10.1016/J.FOODRES.2015.02.021.
A. Untergasser, I. Cutcutache, T. Koressaar, et al., Primer3-new capabilities and interfaces, Nucleic Acids Res., 2012, 40(15), e115, DOI: 10.1093/nar/gks596.
N. Gutierrez, M. J. Giménez, C. Palomino and C. M. Avila, Assessment of candidate reference genes for expression studies in, Vicia faba L. by real-time quantitative PCR, Mol. Breed., 2011, 28(1), 13–24, DOI: 10.1007/s11032-010-9456-7.
J. Hellemans, G. Mortier, A. De Paepe, F. Speleman and J. Vandesompele, qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data, Genome Biol., 2007, 8(2), R19, DOI: 10.1186/gb-2007-8-2-r19.
R Core Team, R: A Language and Environment for Statistical Computing, 2018, https://www.r-project.org/.
Linear Mixed-Effects Models: Basic Concepts and Examples, in, Mixed-Effects Models in S and S-PLUS, Springer-Verlag, New York, 2000, pp. 3–56. DOI: 10.1007/0-387-22747-4_1.
A. R. Gregory Warnes, B. Bolker and T. Lumley, Johnson Contributions from Randall C Johnson RC. Package “gmodels” Title Various R Programming Tools for Model Fitting, 2018, https://cran.r-project.org/web/packages/gmodels/gmodels.pdf.
Y. Tang, M. Horikoshi and W. Li, ggfortify: Unified Interface to Visualize Statistical Results of Popular R Packages, https://journal.r-project.org/archive/2016/RJ-2016–060/RJ-2016-060.pdf.
E.-K. Lim, D. A. Ashford, B. Hou, R. G. Jackson and D. J. Bowles, Arabidopsis glycosyltransferases as biocatalysts in fermentation for regioselective synthesis of diverse quercetin glucosides, Biotechnol. Bioeng., 2004, 87(5), 623–631, DOI: 10.1002/bit.20154.
S. M. Siipola, T. Kotilainen, N. Sipari, et al., Epidermal UV-, A absorbance and whole-leaf flavonoid composition in pea respond more to solar blue light than to solar UV radiation, Plant, Cell Environ., 2015, 38(5), 941–952, DOI: 10.1111/pce.12403.
R. Gonzalez, A. R. Wellburn and N. D. Paul, Dose responses of two pea lines to ultraviolet-B radiation (280–315 nm), Physiol. Plant., 1998, 104(3), 373–378, DOI: 10.1034/j.1399-3054.1998.1040312.x.
L. O. Morales, R. Tegelberg, M. Brosché, M. Keinänen, A. Lindfors and P. J. Aphalo, Effects of solar UV-A and UV-B radiation on gene expression and phenolic accumulation in, Betula pendula leaves, Tree Physiol., 2010, 30(7), 923–934, DOI: 10.1093/treephys/tpq051.
L. O. Morales, M. Brosché, J. Vainonen, et al., Multiple roles for UV RESISTANCE LOCUS8 in regulating gene expression and metabolite accumulation in, Arabidopsis under solar ultraviolet radiation, Plant Physiol., 2013, 161(2), 744–759, DOI: 10.1104/pp.112.211375.
J. Le Roy, B. Huss, A. Creach, S. Hawkins and G. Neutelings, Glycosylation Is a Major Regulator of Phenylpropanoid Availability and Biological Activity in Plants, Front. Plant Sci., 2016, 7, 735, DOI: 10.3389/fpls.2016.00735.
G. I. Jenkins, Signal Transduction in Responses to UV-B Radiation, Annu. Rev. Plant Biol., 2009, 60, 407–431, DOI: 10.1146/annurev.arplant.59.032607.092953.
W. V. Yadav, C. Mallappa, S. N. Gangappa, S. Bhatia and S. Chattopadhyay, A Basic Helix-Loop-Helix Transcription Factor in Arabidopsis, MYC2, Acts as a Repressor of Blue Light–Mediated Photomorphogenic Growth, Plant Cell, 2005, 17(7), 1953–1966, DOI: 10.1105/tpc.105.032060.
M. A. Jansen, V. Gaba and B. M. Greenberg, Higher plants and UV-B radiation: balancing damage, repair and acclimation, Trends Plant Sci., 1998, 3(4), 131–135, DOI: 10.1016/S1360-1385(98)01215-1.
M. Salehin, R. Bagchi and M. Estelle, SCFTIR1/AFB-based auxin perception: mechanism and role in plant growth and development, Plant Cell, 2015, 27(1), 9–19, DOI: 10.1105/tpc.114.133744.
H. U. R. Khan, W. Link, T. J. Hocking and F. L. Stoddard, Evaluation of physiological traits for improving drought tolerance in faba bean (Vicia faba L.), Plant Soil, 2007, 292(1–2), 205–217, DOI: 10.1007/s11104-007-9217-5.
W. R. Briggs and E. Huala, Blue-Light Photoreceptors in Higher Plants, Annu. Rev. Cell Dev. Biol., 1999, 15(1), 33–62, DOI: 10.1146/annurev.cellbio.15.1.33.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: ESI Fig. 1–6 and Tables 1–3. See DOI: 10.1039/c8pp00567b
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Yan, Y., Stoddard, F.L., Neugart, S. et al. Responses of flavonoid profile and associated gene expression to solar blue and UV radiation in two accessions of Vicia faba L. from contrasting UV environments. Photochem Photobiol Sci 18, 434–447 (2019). https://doi.org/10.1039/c8pp00567b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c8pp00567b