Abstract
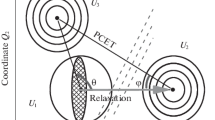
Excited state intramolecular proton transfer (ESIPT) time-constants of 4′-N, N-dimethylamino-3-hydroxyflavone (DMA3HF) in high n-alcohols - 1-butanol, 1-hexanol and 1-decanol - were measured to be 90 ps, 130 ps and 190 ps, respectively, which are unusually slow. At the same time, the solvation time-constants of the DMA3HF enol in the same set of solvents were measured as 100 ps, 150 ps and >300 ps, respectively. Thus, both the ESIPT and enol solvation time-constants in high n-alcohols increase monotonically with the alkyl chain-length of the solvent, although the increase is not strictly proportional. It appears that the H-bonding capacity of the solvent is the single major factor influencing both processes, causing them to become closely correlated. Solvation causes a drastic change in the solvent molecular configuration around the excited enol, E*, inducing the breakage of DMA3HF?solvent inter-molecular H-bonding, which in turn promotes ESIPT. Following previously reported theoretical work on ESIPT, a qualitative description of the S1 potential energy surface can be formulated, where the involvement of solvent relaxation with the ESIPT process is explained.
Similar content being viewed by others
References
T. C. Swinney, D. F. Kelley, Proton transfer dynamics in substituted 3-hydroxyflavones: Solvent polarization effects, J. Chem. Phys., 1993, 99, 211–221.
P.-T. Chou, M. L. Martinez, J. H. Clements, Reversal of excitation behaviour of proton-transfer vs. charge-transfer by dielectric perturbation of electronic manifolds, J. Phys. Chem., 1993, 97, 2618–2622.
S. M. Ormson, R. G. Brown, F. Vollmer, W. Rettig, Switching between charge- and proton-transfer emission in the excited state of a substituted 3-Hydroxyflavone, J. Photochem. Photobiol., A, 1994, 81, 65–72.
A. S. Klymchenko, A. P. Demchenko, Multiparametric probing of intermolecular interactions with fluorescent dye exhibiting excited state intramolecular proton transfer, Phys. Chem. Chem. Phys., 2003, 5, 461–468.
A. D. Roshal, J. A. Organero, A. Douhal, Tuning the mechanism of proton-transfer in a Hydroxyflavone derivative, Chem. Phys. Lett., 2003, 379, 53–59.
A. S. Klymchenko, V. G. Pivovarenkoand, A. P. Demchenko, Elimination of the hydrogen bonding effect on the solvatochromism of 3-hydroxyflavones, J. Phys. Chem. A, 2003, 107, 4211–4216.
A. Douhal, M. Sanz, M. A. Carranza, J. A. Organero, L. Santos, Femtosecond observation of intramolecular charge- and proton-transfer reactions in a hydroxyflavone derivative, Chem. Phys. Lett., 2004, 394, 54–60.
S. Ameer-Beg, S. M. Ormson, X. Poteau, R. G. Brown, P. Foggi, L. Bussotti, F. V. R. Neuwahl, Ultrafast measurements of charge and excited-state intramolecular proton transfer in solutions of 4′-(N,N-dimethylamino) derivatives of 3-Hydroxyflavone, J. Phys. Chem. A, 2004, 108, 6938–6943.
V. G. Pivovarenko, A. Wroblewska, J. Blazejowski, The effect of hydrogen bonding interactions between 2-[4-(dimethylamino) phenyl]-3-hydroxy-4H-chromene-4-one in the ground and excited states and dimethylsulfoxide or methanol on electronic absorption and emission transitions, J. Mol. Struct., 2004, 708, 175–181.
P.-T. Chou, S.-C. Pu, Y.-M. Cheng, W.-S. Yu, Y.-C. Yu, F.-T. Hung, W.-P. Hu, Femtosecond dynamics on excited-state proton/charge-transfer reaction in 4′-N, N-diethylamino-3-hydroxyflavone. The role of dipolar vectors in constructing a rational mechanism, J. Phys. Chem. A, 2005, 109, 3777–3787.
A. S. Klymchenko, C. A. Kenfack, G. Duportail, Y. Mély, Effects of polar protic solvents on dual emissions of 3-Hydroxychromones, J. Chem. Sci., 2007, 119, 83–89.
V. V. Shynkar, Y. Mély, G. Duportail, E. Piémont, A. S. Klymchenko, A. P. Demchenko, Picosecond time-resolved fluorescence studies are consistent with reversible excited-state intramolecular proton transfer in 4′-(dialkylamino)-3-hydroxyflavones, J. Phys. Chem. A, 2003, 107, 9522–9529.
V. V. Shynkar, A. S. Klymchenko, E. Piémont, A. P. Demchenko, Y. Mély, Dynamics of intermolecular hydrogen bonds in the excited states of 4′-dialkylamino-3-hydroxyflavones. On the pathway to an ideal fluorescent hydrogen bonding sensor, J. Phys. Chem. A, 2004, 108, 8151–8159.
J. A. Organero, L. Tormo, M. Sanz, A. Roshal, A. Douhal, Complexation effect of λ-cyclodextrin on a hydroxyflavone derivative: Formation of excluded and included anions, J. Photochem. Photobiol., A, 2007, 188, 74–82.
A. S. Klymchenko, D. A. Yushchenko, Y. Mély, Tuning excited state intramolecular proton transfer in 3-hydroxyflavone derivative by reaction of its isothiocyanate group with an amine, J. Photochem. Photobiol., A, 2007, 192, 93–97.
M. Sanz, J. A. Organero, A. Douhal, Proton and charge transfer reactions dynamics of a hydroxyflavone derivative in a polar solvent and in a cyclodextrin nanocavity, Chem. Phys., 2007, 338, 135–142.
N. A. Nemkovich, J. V. Kruchenok, A. N. Rubinov, V. G. Pivovarenko, W. Baumann, Site selectivity in excited-state intramolecular proton transfer in flavonols, J. Photochem. Photobiol., A, 2001, 139, 53–62.
N. A. Nemkovich, W. Baumann, V. G. Pivovarenko, Dipole moments of 4′-aminoflavonols determined using electro-optical absorption measurements or molecular Stark-effect spectroscopy, J. Photochem. Photobiol., A, 2002, 153, 19–24.
N. A. Nemkovich, V. G. Pivovarenko, W. Baumann, A. N. Rubinov, A. N. Sobchuk, Dipole Moments of 4′-Aminoflavonol Fluorescent Probes in Different Solvents, J. Fluoresc., 2005, 15, 29–36.
A. P. Demchenko, K.-C. Tang, P.-T. Chou, Excited-state proton coupled charge transfer modulated by molecular structure and media polarization, Chem. Soc. Rev., 2013, 42, 1379–1408.
D. Ghosh, S. Batuta, S. Das, N. A. Begum, D. Mandal, Proton Transfer Dynamics of 4′-N,N-Dimethylamino-3-hydroxyflavone Observed in Hydrogen-Bonding Solvents and Aqueous Micelles, J. Phys. Chem. B, 2015, 119, 5650–5661.
M. L. Horng, J. A. Gardecki, A. Papazyan, M. Maroncelli, Subpicosecond Measurements of Polar Solvation Dynamics: Coumarin 153 Revisited, J. Phys. Chem., 1995, 99, 17311–17337.
M. H. Abraham, Scales of Solute Hydrogen-bonding: Their Construction and Application to Physicochemical and Biochemical Processes, Chem. Soc. Rev., 1993, 22, 73–83.
M. A. Smith, R. M. Neumann, R. A. Webb, A modification of the Alger-Flynn-Oyamada preparation of Flavonols, J. Heterocycl. Chem., 1968, 5, 425–426.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision B.01, Gaussian, Inc., Wallingford CT, 2009.
A. V. Marenich, C. J. Cramer, D. G. Truhlar, Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions, J. Phys. Chem. B, 2009, 113, 6378–6396.
C. A. Kenfack, A. S. Klymchenko, G. Duportail, A. Burgerc, Y. Mely, Ab initio study of the solvent H-bonding effect on ESIPT reaction and electronic transitions of 3-hydroxychromone derivatives, Phys. Chem. Chem. Phys., 2012, 14, 8910–8918.
C. Peng, H. B. Schlegel, Combining synchronous transit and quasi-Newton methods to find transition states, Isr. J. Chem., 1993, 33, 449–454.
K. Suda, M. Terazima, H. Sato, Y. Kimura, Excitation wavelength dependence of excited state intramolecular proton transfer reaction of 4′-N,N-diethylamino-3-hydroxyflavone in room temperature ionic liquids studied by optical kerr gate fluorescence measurement, J. Phys. Chem. B, 2013, 117, 12567–12582.
Y. Kimura, M. Fukuda, K. Suda, M. Terazima, Excited State Intramolecular Proton Transfer Reaction of 4′-N, N-Diethylamino-3-hydroxyflavone and Solvation Dynamics in Room Temperature Ionic Liquids Studied by Optical Kerr Gate Fluorescence Measurement, J. Phys. Chem. B, 2010, 114, 11847–11858.
S. Hayaki, Y. Kimura, H. Sato, Ab Initio Study on an Excited-State Intramolecular Proton-Transfer Reaction in Ionic Liquid, J. Phys. Chem. B, 2013, 117, 6759–6767.
M. Fukuda, M. Terazima, Y. Kimura, Study on the excited state intramolecular proton transfer of 4′-N,N-diethylamino-3-hydroxyflavone in imidazolium-based room temperature ionic liquids, Chem. Phys. Lett., 2008, 463, 364–368.
K. Suda, M. Terazima, Y. Kimura, Excitation wavelength dependence of photo-induced intramolecular proton transfer reaction of 4′-N,N-diethylamino-3-hydroxyflavone in various liquids, Chem. Phys. Lett., 2012, 531, 70–74.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c5pp00377f
Rights and permissions
About this article
Cite this article
Ghosh, D., Batuta, S., Begum, N.A. et al. Unusually slow intramolecular proton transfer dynamics of 4′-N,N-dimethylamino-3-hydroxyflavone in high n-alcohols: involvement of solvent relaxation. Photochem Photobiol Sci 15, 266–277 (2016). https://doi.org/10.1039/c5pp00377f
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c5pp00377f