Abstract
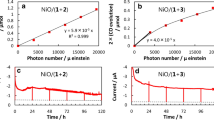
We report a novel system of visible-light-driven CO2 reduction to CO in an aqueous solution, in which DPPC vesicles dispersed in the solution act as a photocatalyst using ascorbate (HAsc−) as an electron source. In the vesicles metal complexes [Ru(dtb)(bpy)2]2+ and Re(dtb)(CO)3Cl (dtb = 4,4′-ditridecyl-2,2′-bipyridyl) are incorporated, which act as a photosensitizer and a catalyst for CO2 reduction, respectively. The reaction is initiated with the reductive quenching of the 3MLCT excited state of the Ru complex with HAsc−, followed by an electron transfer from the reduced Ru complex to the Re complex to give a one-electron reduced Re species having catalytic ability for CO2 reduction. In order to search for optimum conditions for the CO production, the dependence of the initial rate of CO formation, vi, on the concentration of the metal complexes and HAsc− in the vesicle solution was examined. Consequently, we obtained ∼3.5 μmol h−1 and 190 for vi and the turnover number for CO formation with respect to the Re catalyst, respectively. On the basis of the dependence of vi on the incident light intensity, we have concluded that the photocatalytic reduction of CO2 to CO with HAsc− in this system requires only one photon, and propose that HAsc− donates an electron not only to the excited state of the Ru complex, but also to the Re–CO2 adduct involved in the catalytic cycle for CO2 reduction.
Similar content being viewed by others
References
J.-M. Lehn, and R. Ziessel, Photochemical generation of carbon monoxide and hydrogen by reduction of carbon dioxide and water under visible light irradiation, Proc. Natl. Acad. Sci. U. S. A., 1982, 79, 701–704.
H. Ishida, T. Terada, K. Tanaka, and T. Tanaka, Photochemical CO2 reduction catalyzed by [Ru(bpy)2(CO)2]2+ using triethanolamine and 1-benzyl-1,4-dihydronicotinamide as an electron donor, Inorg. Chem., 1990, 29, 905–911.
E. Kimura, X. Bu, M. Shionoya, S. Wada, and S. Maruyama, A new nickel(ii) cyclam (cyclam=1,4,8,11-tetraazacyclotetradecane) complex covalently attached to Ru(phen)32+ (phen=1,10-phenanthroline). A new candidate for the catalytic photoreduction of carbon dioxide, Inorg. Chem., 1992, 31, 4542–4546.
J. Grodkowski, and P. Neta, Ferrous ions as catalysts for photochemical reduction of CO in homogeneous solutions, J. Phys. Chem. A, 2000, 104, 4475–4479.
J. Hawecker, J.-M. Lehn, and R. Ziessel, Photochemical and electrochemical reduction of carbon dioxide to carbon monoxide mediated by (2,2′-bipyridine)tricarbonylchlororhenium(i) and related complexes as homogeneous catalysts, Helv. Chim. Acta, 1986, 69, 1990–2012.
H. Takeda, and O. Ishitani, Development of efficient photocatalytic systems for CO2 reduction using mononuclear and multinuclear metal complexes based on mechanistic studies, Coord. Chem. Rev., 2010, 254, 346–354.
H. Hori, F. P. A. Johnson, K. Koike, O. Ishitani, and T. Ibusuki, Efficient photocatalytic CO2 reduction using [Re(bpy)(CO)3P(OEt)3]+, J. Photochem. Photobiol., A, 1996, 96, 171–174.
K. Koike, S. Naito, S. Sato, Y. Tamaki, and O. Ishitani, Architecture of supramolecular metal complexes for photocatalytic CO2 reduction III: Effects of length of alkyl chain connecting photosensitizer to catalyst, J. Photochem. Photobiol., A, 2009, 207, 109–114.
A. D. Bangham, Membrane models with phospholipids, Prog. Biophys. Mol. Biol., 1968, 18, 29–95.
K. Watanabe, S. Takizawa, and S. Murata, Hydrogen generation using a photoinduced electron-transport system with a molecular catalyst in vesicles, Chem. Lett., 2011, 40, 345–347.
T. Toyota, K. Takakura, Y. Kageyama, K. Kurihara, N. Maru, K. Ohnuma, K. Kaneko, and T. Sugawara, Population study of size and components of self-reproducing giant multilamellar vesicles, Langmuir, 2008, 24, 3037–3044.
J. N. Robinson, D. J. Cole-Hamilton, Electron transfer across vesicle bilayers, Chem. Soc. Rev., 1991, 20, 49–94.
T. Mizushima, A. Yoshida, A. Harada, Y. Yoneda, T. Minatani, and S. Murata, Pyrene-sensitized electron transport across vesicle bilayers: dependence of transport efficiency on pyrene substituents, Org. Biomol. Chem., 2006, 4, 4336–4344.
D. Njus, and P. M. Kelley, The secretory-vesicle ascorbate-regenerating system: a chain of concerted H+/e−-transfer reactions, Biochim. Biophys. Acta, 1993, 1144, 235–248.
B. Shan, T. Baine, X. A. N. Ma, X. Zhao, and R. H. Schmehl, Mechanistic details for cobalt catalyzed photochemical hydrogen production in aqueous solution: Efficiencies of the photochemical and non-photochemical steps, Inorg. Chem., 2013, 52, 4853–4859.
G. M. Brown, B. S. Brunschwig, C. Creutz, J. F. Endicott, and N. Sutin, Homogeneous catalysis of the photoreduction of water by visible light. Mediation by a tris(2,2-bipyridine)ruthenium(ii)-cobalt(ii) macrocycle system, J. Am. Chem. Soc., 1979, 101, 1298–1300.
W. R. McNamara, Z. Han, P. J. Alperin, W. W. Brennessel, P. L. Holland, and R. Eisenberg, A cobalt-dithiolene complex for the photocatalytic and electrocatalytic reduction of protons, J. Am. Chem. Soc., 2011, 133, 15368–15371.
Very recently, MacDonnell and co-workers reported photochemical catalytic CO2 reduction to formate and methanol using ascorbic acid as an electron source: D. J. Boston, C. Xu, D. W. Armstrong, and F. M. MacDonnell, Photochemical reduction of carbon dioxide to methanol and formate in a homogeneous system with pyridinium catalysts, J. Am. Chem. Soc., 2013, 135, 16252–16255.
B. Gholamkhass, H. Mametsuka, K. Koike, T. Tanabe, M. Furue, and O. Ishitani, Architecture of supramolecular metal complexes for photocatalytic CO2 reduction: Ruthenium-rhenium bi- and tetranuclear complexes, Inorg. Chem., 2005, 44, 2326–2336.
S. Sato, K. Koike, H. Inoue, and O. Ishitani, Highly efficient supramolecular photocatalysts for CO2 reduction using visible light, Photochem. Photobiol. Sci., 2007, 6, 454–461.
L. Rodríguez, M. Ferrer, O. Rossell, F. J. S. Duarte, A. G. Santos, and J. C. Lima, Solvent effects on the absorption and emission of [Re(R2bpy)(CO)3X] complexes and their sensitivity to CO2 in solution, J. Photochem. Photobiol., A, 2009, 204, 174–182.
Z.-Y. Bian, S.-M. Chi, L. Li, and W. Fu, Conjugation effect of the bridging ligand on the CO2 reduction properties in difunctional photocatalysts, Dalton Trans., 2010, 39, 7884–7887.
C. Bruckmeier, M. W. Lehenmeier, R. Reithmeier, B. Rieger, J. Herranz, and C. Kavakli, Binuclear rhenium(i) complexes for the photocatalytic reduction of CO2, Dalton Trans., 2012, 41, 5026–5037.
K. Watanabe, K. Moriya, T. Kouyama, A. Onoda, T. Minatani, S. Takizawa, and S. Murata, Photoinduced transmembrane electron transport in DPPC vesicles: Mechanism and application to a hydrogen generation system, J. Photochem. Photobiol., A, 2011, 221, 113–122.
C. Creutz, N. Sutin, and B. S. Brunschwig, Excite-state photochemistry in the tris(2,2′-bipyridine)ruthenium(ii)-sulfite system, J. Am. Chem. Soc., 1979, 101, 1297–1298.
D. Rehm, and A. Weller, Kinetics of fluorescence quenching by electron and H-atom transfer, Isr. J. Chem., 1970, 8, 259–271.
For the calculation of the free-energy change for the photoinduced electron transfer, +0.09 V (vs. SCE) was employed for the oxidation potential of HAsc˙/HAsc− (pH 7.0).14 The reduction potential (−1.34 V vs. SCE) and the 3MLCT excited state energy (2.10 eV) of [Ru(dtb)(bpy)2]2+ were estimated by the observed redox potential and phosphorescence spectrum of [Ru(dtb)(bpy)2](PF6)2, respectively.
S. Nojavan, F. Khalilian, F. M. Kiaie, A. Rahimi, A. Arabanian, and S. Chalavi, Extraction and quantitative determination of ascorbic acid during different maturity state of Rosa canina L. fruit, J. Food Compos. Anal., 2008, 21, 300–305.
J. van Houten, and R. J. Watts, Photochemistry of tris(2,2′-bipyridyl)ruthenium(ii) in aqueous solutions, Inorg. Chem., 1978, 17, 3381–3385.
H. Takeda, K. Koike, H. Inoue, and O. Ishitani, Development of an efficient photocatalytic system for CO2 reduction using rhenium(i) complexes based on mechanistic studies, J. Am. Chem. Soc., 2008, 130, 2023–2031.
For a calculation of the free-energy change, −0.28 V and +0.40 V (vs. NHE) were employed for the standard redox potentials of CO2/CO32 and Asc/H2Asc,33,34 respectively.
J. P. Collin, and J. P. Sauvage, Electrochemical reduction of carbon dioxide mediated by molecular catalysts, Coord. Chem. Rev., 1989, 93, 245–268.
E. G. Ball, Studies on oxidation-reduction: XXIII. Ascorbic acid, J. Biol. Chem., 1937, 118, 219–239.
C. Creutz, The complexities of ascorbate as a reducing agent, Inorg. Chem., 1981, 20, 4449–4452.
G. Sprintschnik, H. W. Sprintschnik, P. P. Kirsch, and D. G. Whitten, Preparation and photochemical reactivity of surfactant ruthenium(ii) complexes in monolayer assemblies and at water-solid interfaces, J. Am. Chem. Soc., 1977, 99, 4947–4954.
J. M. Smieja, and C. P. Kubiak, Re(bipy-tBu)(CO)3Cl-improved catalytic activity for carbon dioxide: IR-spectroelectrochemical and mechanistic studies, Inorg. Chem., 2010, 49, 9283–9289.
D. Kiriya, H.-C. Chang, A. Kamata, and S. Kitagawa, Polytypic phase transition in alkyl chain-functionalized valence tautomeric complexes, Dalton Trans., 2006, 1377–1382.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c3pp50429h
Rights and permissions
About this article
Cite this article
Ikuta, N., Takizawa, Sy. & Murata, S. Photochemical reduction of CO2 with ascorbate in aqueous solution using vesicles acting as photocatalysts. Photochem Photobiol Sci 13, 691–702 (2014). https://doi.org/10.1039/c3pp50429h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c3pp50429h