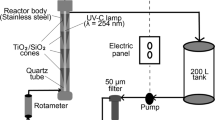
Abstract
In this work, the disinfection efficiency of water and secondary treated wastewater by means of UV-A, UV-C and solar irradiation in the presence or absence of TiO2, using a reference strain of Enterococcus faecalis as faecal indicator, was evaluated. Operating parameters such as TiO2 loading (0-1500 mg L−1), initial bacterial concentration (2 × 102-108 CFU mL−1) and treatment time (up to 120 min) were assessed concerning their impact on disinfection. E. faecalis inactivation was monitored by the conventional culture method and real-time PCR. Regarding photocatalytic treatment, disinfection efficiency was improved by increasing TiO2 concentration and bacterial inactivation took place in relatively short treatment times. Comparing the three disinfection methods, it was observed that UV-C irradiation yielded a better efficiency during water treatment than UV-A and solar irradiation. Furthermore, UV-A was more efficient than solar irradiation in the presence of the same loading of TiO2. Regarding real wastewater, it was observed that only UV-C irradiation was capable of totally inactivating E. faecalis population in a short time. Screening the results obtained from both applied techniques (culture method and real-time PCR), there was a discrepancy, regarding the recorded time periods of total bacterial inactivation. Real-time PCR data revealed that longer periods are needed for 100% bacterial reduction during the treatments tested compared to the estimated time by culture method. This is probably attributed to the phenomenon of “viable but not culturable bacteria”, caused by stressed conditions induced during disinfection experiments. Taking into account the contrast of results and in order to perform a thorough evaluation of disinfection techniques, conventional culture method should be accompanied by a DNA-based method. According to our findings, real-time PCR proved to be a reliable and accurate molecular tool for the identification and quantification of bacterial indicators, like E. faecalis, in aquatic samples after disinfection treatment.
Similar content being viewed by others
References
S. Malato, P. Fernandez-Ibanez, M. I. Maldonado, J. Blanco and W. Gernjak, Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends, Catal. Today, 2009, 147, 1–59.
M. N. Chong, B. Jin, C. W. K. Chow and C. Saint, Recent developments in photocatalytic water treatment technology: A review, Water Res., 2010, 44, 2997–3027.
C. M. Davies, D. J. Roser, A. J. Feitz and N. J. Ashbolt, Solar radiation disinfection of drinking water at temperate latitudes: Inactivation rates for an optimised reactor configuration, Water Res., 2009, 43, 643–652.
R. M. Maier, I. L. Pepper and C. P. Gerba, in Environmental Microbiology, Academic Press, Elsevier, 2nd edn, 2009, ch. 26, pp. 539–545.
T. Westrell, O. Bergstedt, T. A. Stenstrom and N. J. Ashbolt, A theoretical approach to assess microbial risks due to failures in drinking water systems, Int. J. Environ. Health Res., 2003, 13, 181–197.
J. Koivunen and H. Heinonen-Tanski, Inactivation of enteric microorganisms with chemical disinfectants, UV irradiation and combined chemical/UV treatments, Water Res., 2005, 39, 1519–1526.
N. Lydakis-Simantiris, D. Riga, E. Katsivela, D. Mantzavinos and N. P. Xekoukoulotakis, Disinfection of spring water and secondary treated municipal wastewater by TiO2 photocatalysis, Desalination, 2010, 250, 351–355.
C. Jungfer, T. Schwartz and U. Obst, UV-induced dark repair mechanisms in bacteria associated with drinking water, Water Res., 2007, 41, 188–196.
M. Guo, H. Hu, J. R. Bolton and M. G. El-Din, Comparison of low- and medium-pressure ultraviolet lamps: Photoreactivation of Escherichia coli and total coliforms in secondary effluents of municipal wastewater treatment plants, Water Res., 2009, 43, 815–821.
R. P. Sinha and D. P. Häder, UV-induced DNA damage and repair: a review, Photochem. Photobiol. Sci., 2002, 1, 225–236.
A. G. Rincón and C. Pulgarin, Field solar E. coli inactivation in the absence and presence of TiO2: Is UV solar dose an appropriate parameter for standardization of water solar disinfection?, Sol. Energy, 2004, 77, 635–648.
A. K. Benabbou, Z. Derriche, C. Felix, P. Lejeune and C. Guillard, Photocatalytic inactivation of Escherischia coli. Effect of concentration of TiO2 and microorganism, nature, and intensity of UV irradiation, Appl. Catal., B, 2007, 76, 257–263.
A. Paleologou, H. Marakas, N. P. Xekoukoulotakis, A. Moya, Y. Vergara, N. Kalogerakis, P. Gikas and D. Mantzavinos, Disinfection of water and wastewater by TiO2 photocatalysis, sonolysis and UV-C irradiation, Catal. Today, 2007, 129, 136–142.
L. Heijnen and G. Medema, Method for rapid detection of viable Escherichia coli in water using real-time NASBA, Water Res., 2009, 43, 3124–3132.
R. V. Grieken, J. Marugán, C. Pablos, L. Furones and A. López, Comparison between the photocatalytic inactivation of Gram-positive E. faecalis and Gram-negative E. coli faecal contamination indicator microorganisms, Appl. Catal., B, 2010, 100, 212–220.
J. A. Herrera Melián, J. M. Doña Rodríguez, A. Viera Suárez, E. Tello Rendón, C. Valdés do Campo, J. Arana and J. Pérez Peña, The photocatalytic disinfection of urban waste waters, Chemosphere, 2000, 41, 323–327.
K. Backhaus, J. Marugán, R. V. Grieken and C. Sordo, Photocatalytic inactivation of E. faecalis in secondary wastewater plant effluents, Water Sci. Technol., 2010, 61, 2355–2361.
M. J. Figueras and J. J. Borrego, New perspectives in monitoring drinking water microbial quality, Int. J. Environ. Res. Public Health, 2010, 7, 4179–4202.
M. M. Lleo, B. Bonato, M. C. Tafi, C. Signoretto, C. Pruzzo and P. Canepari, Molecular vs. culture methods for the detection of bacterial faecal indicators in groundwater for human use, Lett. Appl. Microbiol., 2005, 40, 289–294.
I. Brettar and M. G. Höfle, Molecular assessment of bacterial pathogens - a contribution to drinking water safety, Curr. Opin. Biotechnol., 2008, 19, 274–280.
L. A. Bjergbæk and P. Roslev, Formation of nonculturable Escherichia coli in drinking water, J. Appl. Microbiol., 2005, 99, 1090–1098.
A. Muela, C. Seco, E. Camafeita, I. Arana, M. Orruño, J. A. López and I. Barcina, Changes in Escherichia coli outer membrane subproteome under environmental conditions inducing the viable but nonculturable state, FEMS Microbiol. Ecol., 2008, 64, 28–36.
E. Frahm and U. Obst, Application of the fluorogenic probe technique (TaqMan PCR) to the detection of Enterococcus spp. and Escherichia coli in water samples, J. Microbiol. Methods, 2003, 52, 123–131.
N. Wéry, C. Lhoutellier, F. Ducray, J. P. Delgenès and J. J. Godon, Behaviour of pathogenic and indicator bacteria during urban wastewater treatment and sludge composting, as revealed by quantitative PCR, Water Res., 2008, 42, 53–62.
APHA, Standard Methods for the Examination of Water and Wastewater, 20th edn, American Public Health Association, Washington, DC, 1999.
D. Y. Lee, H. Lauder, H. Cruwys, P. Falletta and L. A. Beaudette, Development and application of an oligonucleotide microarray and real-time quantitative PCR for detection of wastewater bacterial pathogens, Sci. Total Environ., 2008, 398, 203–211.
J. M. Williams, M. Trope, D. J. Caplan and D. C. Shugars, Detection and quantitation of E. faecalis by Real-time PCR (qPCR), Reverse Transcription-PCR (RT-PCR), and cultivation during endodontic treatment, Clin. Res., 2006, 32, 715–721.
M. A. Rauf, M. A. Meetani and S. Hisaindee, An overview on the photocatalytic degradation of azo dyes in the presence of TiO2 doped with selective transition metals, Desalination, 2011, 276, 13.
G. Gogniat, M. Thyssen, M. Denis, C. Pulgarin and S. Dukan, The bactericidal effect of TiO2 photocatalysis involves adsorption onto catalyst and the loss of membrane integrity, FEMS Microbiol. Lett., 2006, 258, 18–24.
M. Lebuhn, M. Effenberger, G. Garcés, A. Gronauer and P. A. Wilderer, Evaluating real-time PCR for the quantification of distinct pathogens and indicator organisms in environmental samples, Water Sci. Technol., 2004, 50, 263–270.
E. Viau and J. Peccia, Evaluation of the enterococci indicator in biosolids using culture-based and quantitative PCR assays, Water Res., 2009, 43, 4878–4887.
J. Süb, S. Volz, U. Obst and T. Schwartz, Application of a molecular biology concept for the detection of DNA damage and repair during UV disinfection, Water Res., 2009, 43, 3705–3716.
K. Rudi, I. Hagen, B. C. Johnsrud, G. Skjefstad and I. Tryland, Different Length (DL) qPCR for quantification of cell killing by UV-induced DNA damage, Int. J. Environ. Res. Public Health, 2010, 7, 3376–3381.
R. T. Noble, A. D. Blackwood, J. F. Griffith, C. D. McGee and S. B. Weisberg, Comparison of rapid quantitative PCR-based and conventional culture-based methods for enumeration of Enterococcus spp. and Escherichia coli in recreational waters, Appl. Environ. Microbiol., 2010, 76, 7437–7443.
I. Bertrand, C. Gantzer, T. Chesnot and J. Schwartzbrod, Improved specificity for Giardia lamblia cyst quantification in wastewater by development of a real-time PCR method, J. Microbiol. Methods, 2004, 57, 41–53.
C. Maya, N. Beltran, B. Jimenez and P. Bonilla, Evaluation of the UV disinfection process in bacteria and amphizoic amoeba inactivation, Water Sci. Technol.: Water Supply, 2003, 3, 285–291.
Y. Gilboa and E. Friedler, UV disinfection of RBC-treated light greywater effluent: kinetics, survival and regrowth of selected mi-croorgnisms, Water Res., 2008, 42, 1043–1050.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Venieri, D., Chatzisymeon, E., Gonzalo, M.S. et al. Inactivation of Enterococcus faecalis by TiO2-mediated UV and solar irradiation in water and wastewater: culture techniques never say the whole truth. Photochem Photobiol Sci 10, 1744–1750 (2011). https://doi.org/10.1039/c1pp05198a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c1pp05198a