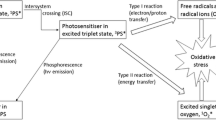
Abstract
Photochemotherapy, in which ultraviolet radiation (UVR: 280–400 nm) or visible light is combined with a photosensitizing drug to produce a therapeutic effect that neither drug or radiation can achieve alone, is a proven therapeutic strategy for a number of non-malignant hyperproliferative skin conditions and various cancers. Examples are psoralen plus UVA (320–400 nm) radiation (PUVA) and photodynamic therapy (PDT). All existing photochemotherapies have drawbacks–for example the association of PUVA with the development of skin cancer, and pain that is often associated with PDT treatment of skin lesions. There is a clear need to develop alternative approaches that involve lower radiation doses and/or improved selectivity for target cells. In this review, we explore the possibility to address this need by exploiting thionucleoside-mediated DNA photosensitisation to low, non toxic doses of UVA radiation.
Similar content being viewed by others
References
R. S. Stern, E. J. Lunder, Risk of squamous cell carcinoma and methoxsalen (psoralen) and UV-A radiation (PUVA) - A meta-analysis, Arch. Dermatol., 1998, 134, 1582–1585.
R. S. Stern, P. F. U. Study, The risk of melanoma in association with long-term exposure to PUVA, J. Am. Acad. Dermatol., 2001, 44, 755–761.
R. S. Stern, Psoralen and ultraviolet a light therapy for psoriasis, N. Engl. J. Med., 2007, 357, 682–690.
T. P. Singh, B. Huettner, H. Koefeler, G. Mayer, I. Bambach, K. Wallbrecht, M. P. Schon, P. Wolf, Platelet-Activating Factor Blockade Inhibits the T-Helper Type 17 Cell Pathway and Suppresses Psoriasis-Like Skin Disease in K5.hTGF-beta 1 Transgenic Mice, Am. J. Pathol., 2011, 178, 699–708.
D. Papadopoulo, F. Sagliocco, D. Averbeck, Mutagenic Effects of 3-Carbethoxypsoralen and 8-Methoxypsoralen Plus 365-nm Irradiation in Mammalian-Cells, Mutat. Res., Genet. Toxicol., 1983, 124, 287–297.
A. R. Young, I. A. Magnus, A. C. Davies, N. P. Smith, A Comparison of the Phototumorigenic Potential of 8-MOP and 5-MOP in Hairless Albino Mice Exposed to Solar Simulated Radiation, Br. J. Dermatol., 1983, 108, 507–518.
R. Knobler, M. L. Barr, D. R. Couriel, J. L. M. Ferrara, L. E. French, P. Jaksch, W. Reinisch, A. H. Rook, T. Schwarz, H. Greinix, Extracorporeal photopheresis: Past, present, and future, J. Am. Acad. Dermatol., 2009, 61, 652–665.
P. Babilas, S. Schreml, M. Landthaler, R. M. Szeimies, Photodynamic therapy in dermatology: state-of-the-art, Photodermatol., Photoimmunol. Photomed., 2010, 26, 118–132.
D. Fayter, M. Corbett, M. Heirs, D. Fox, A. Eastwood, A systematic review of photodynamic therapy in the treatment of pre-cancerous skin conditions, Barrett’s oesophagus and cancers of the biliary tract, brain, head and neck, lung, oesophagus and skin, Health Technol. Assess., 2010, 14, 1–288.
J. H. Pinthus, A. Bogaards, R. Weersink, B. C. Wilson, J. Trachtenberg, Photodynamic therapy for urological malignancies: Past to current approaches, J. Urol., 2006, 175, 1201–1207.
S. K. Attili, R. Dawe, S. Ibbotson, A review of pain experienced during topical photodynamic therapy–Our experience in Dundee, Photodiagn. Photodyn. Ther., 2011, 8, 53–57.
S. H. Ibbotson, Adverse effects of topical photodynamic therapy, Photodermatol., Photoimmunol. Photomed., 2011, 27, 116–130.
A. M. Bugaj, Targeted photodynamic therapy - a promising strategy of tumor treatment, Photochem. Photobiol. Sci., 2011, 10, 1097–1109.
M. Verhille, P. Couleaud, R. Vanderesse, D. Brault, M. Barberi-Heyob, C. Frochot, Modulation of Photosensitization Processes for an Improved Targeted Photodynamic Therapy, Curr. Med. Chem., 2010, 17, 3925–3943.
A. R. Young, C. A. Chadwick, G. I. Harrison, O. Nikaido, J. Ramsden, C. S. Potten, The similarity of action spectra for thymine dimers in human epidermis and erythema suggests that DNA is the chromophore for erythema, J. Invest. Dermatol., 1998, 111, 982–988.
A. Massey, Y.-Z. Xu, P. Karran, Photoactivation of DNA thiobases as a potential novel therapeutic option, Curr. Biol., 2001, 11, 1142–1146.
A. Massey, Y. Z. Xu, P. Karran, Ambiguous coding is required for the lethal interaction between methylated DNA bases and DNA mismatch repair, DNA Repair, 2002, 1, 275–286.
S. W. Pridgeon, R. Heer, G. A. Taylor, D. R. Newell, K. O’Toole, M. Robinson, Y. Z. Xu, P. Karran, A. V. Boddy, Thiothymidine combined with UVA as a potential novel therapy for bladder cancer, Br. J. Cancer, 2011, 104, 1869–1876.
O. Reelfs, P. MacPherson, X. Ren, Y.-Z. Xu, P. Karran, A. Young, Identification of potentially cytotoxic lesions induced by UVA photoactivation of DNA 4-thiothymidine in human cells, Nucleic Acids Res., 2011 10.1093/nar/gkr674.
D. Cunningham, J. R. Zalcberg, U. Rath, I. Olver, E. Van Cutsem, C. Svensson, J. F. Seitz, P. Harper, D. Kerr, G. Perez-Manga et al., ‘Tomudex’ (ZD1694): results of a randomised trial in advanced colorectal cancer demonstrate efficacy and reduced mucositis and leucopenia. The ‘Tomudex’ Colorectal Cancer Study Group, Eur. J. Cancer, 1995, 31A, 1945–1954.
P. F. Swann, T. R. Waters, D. C. Moulton, Y. Z. Xu, Q. Zheng, M. Edwards, R. Mace, Role of postreplicative DNA mismatch repair in the cytotoxic action of thioguanine, Science, 1996, 273, 1109–1111.
O Reelfs, Y. Z. Xu, A. Massey, P. Karran, A. Storey, Thiothymidine plus low-dose UVA kills hyperproliferative human skin cells independently of their human papilloma virus status, Mol. Cancer Ther., 2007, 6, 2487–2495.
J. L. Sherley, T. J. Kelly, Regulation of human thymidine kinase during the cell cycle, J. Biol. Chem., 1988, 263, 8350–8358.
M. Hengstschlager, M. Pfeilstocker, E. Wawra, Thymidine kinase expression. A marker for malignant cells, Adv. Exp. Med. Biol., 1998, 431, 455–460.
J. Cadet, T. Douki, J. L. Ravanat, P. Di Mascio, Sensitized formation of oxidatively generated damage to cellular DNA by UVA radiation, Photochem. Photobiol. Sci., 2009, 8, 903–911.
P. O’Donovan, C. M. Perrett, X. Zhang, B. Montaner, Y. Z. Xu, C. A. Harwood, J. M. McGregor, S. L. Walker, F. Hanaoka, P. Karran, Azathioprine and UVA light generate mutagenic oxidative DNA damage, Science, 2005, 309, 1871–1874.
B. Montaner, P. O’Donovan, O. Reelfs, C. M. Perrett, X. Zhang, Y. Z. Xu, X. Ren, P. Macpherson, D. Frith, P. Karran, Reactive oxygen-mediated damage to a human DNA replication and repair protein, EMBO Rep., 2007, 8, 1074–1079.
X. Zhang, G. Jeffs, X. Ren, P. O’Donovan, B. Montaner, C. M. Perrett, P. Karran, Y. Z. Xu, Novel DNA lesions generated by the interaction between therapeutic thiopurines and UVA light, DNA Repair, 2007, 6, 344–354.
Y. Harada, T. Suzuki, T. Ichimura, Y. Z. Xu, Triplet formation of 4-thiothymidine and its photosensitization to oxygen studied by time-resolved thermal lensing technique, J. Phys. Chem. B, 2007, 111, 5518–5524.
M. A. Warren, J. B. Murray, B. A. Connolly, Synthesis and characterisation of oligodeoxynucleotides containing thio analogues of (6-4) pyrimidine-pyrimidinone photo-dimers, J. Mol. Biol., 1998, 279, 89–100.
C. Kielbassa, B. Epe, DNA damage induced by ultraviolet and visible light and its wavelength dependence, Methods Enzymol., 2000, 319, 436–445.
T. Douki, A. Reynaud-Angelin, J. Cadet, E. Sage, Bipyrimidine photoproducts rather than oxidative lesions are the main type of DNA damage involved in the genotoxic effect of solar UVA radiation, Biochemistry, 2003, 42, 9221–9226.
J. E. Cleaver, F. Cortes, L. H. Lutze, W. F. Morgan, A. N. Player, D. L. Mitchell, Unique DNA repair properties of a xeroderma pigmentosum revertant, Mol. Cell Biol., 1987, 7, 3353–3357.
A. Favre, C. Saintome, J. L. Fourrey, P. Clivio, P. Laugaa, Thionucleobases as intrinsic photoaffinity probes of nucleic acid structure and nucleic acid-protein interactions, J. Photochem. Photobiol., B, 1998, 42, 109–124.
R. O. Rahn, J. L. Hosszu, Photochemical Studies of Thymine in Ice, Photochem. Photobiol., 1969, 10, 131–137.
P. Clivio, J. L. Fourrey, J. Gasche, A. Favre, DNA Photodamage Mechanistic Studies - Characterization of a Thietane Intermediate in a Model Reaction Relevant to 6-4 Lesions, J. Am. Chem. Soc., 1991, 113, 5481–5483.
P. Clivio, J. L. Fourrey, J. Gasche, A. Favre, Novel Insight into the Stereochemical Pathway Leading to (6-4) Pyrimidine-Pyrimidone Photoproducts in DNA, Tetrahedron Lett., 1992, 33, 1615–1618.
D. Perdiz, P. Grof, M. Mezzina, O. Nikaido, E. Moustacchi, E. Sage, Distribution and repair of bipyrimidine photoproducts in solar UV-irradiated mammalian cells. Possible role of Dewar photoproducts in solar mutagenesis, J. Biol. Chem., 2000, 275, 26732–26742.
B. S. Rosenstein, D. L. Mitchell, The repair of DNA damages induced in normal human skin fibroblasts exposed to simulated sunlight, Radiat. Res., 1991, 126, 338–342.
S. Courdavault, C. Baudouin, M. Charveron, B. Canguilhem, A. Favier, J. Cadet, T. Douki, Repair of the three main types of bipyrimidine DNA photoproducts in human keratinocytes exposed to UVB and UVA radiations, DNA Repair, 2005, 4, 836–844.
B. Bartholomew, B. R. Braun, G. A. Kassavetis, E. P. Geiduschek, Probing close DNA contacts of RNA polymerase III transcription complexes with the photoactive nucleoside 4-thiodeoxythymidine, J. Biol. Chem., 1994, 269, 18090–18095.
T. T. Nikiforov, B. A. Connolly, Oligodeoxynucleotides containing 4-thiothymidine and 6-thiodeoxyguanosine as affinity labels for the Eco RV restriction endonuclease and modification methylase, Nucleic Acids Res., 1992, 20, 1209–1214.
J. T. Reardon, A. Sancar, Repair of DNA-polypeptide crosslinks by human excision nuclease, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 4056–4061.
T. Nakano, A. Katafuchi, M. Matsubara, H. Terato, T. Tsuboi, T. Masuda, T. Tatsumoto, S. P. Pack, K. Makino, D. L. Croteau, B. Van Houten, K. Iijima, H. Tauchi, H. Ide, Homologous recombination but not nucleotide excision repair plays a pivotal role in tolerance of DNA-protein cross-links in mammalian cells, J. Biol. Chem., 2009, 284, 27065–27076.
A. Ito, F. T. Robb, J. G. Peak, M. J. Peak, Base-specific damage induced by 4-thiouridine photosensitization with 334-nm radiation in M13 phage DNA, Photochem. Photobiol., 1988, 47, 231–240.
M. C. Wei, T. Lindsten, V. K. Mootha, S. Weiler, A. Gross, M. Ashiya, C. B. Thompson, S. J. Korsmeyer, tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c, Genes Dev., 2000, 14, 2060–2071.
D. E. Brash, J. A. Rudolph, J. A. Simon, A. Lin, G. J. McKenna, H. P. Baden, A. J. Halperin, J. Ponten, A role for sunlight in skin cancer: UV-induced p53 mutations in squamous cell carcinoma, Proc. Natl. Acad. Sci. U. S. A., 1991, 88, 10124–10128.
A. Ziegler, D. J. Leffell, S. Kunala, H. W. Sharma, M. Gailani, J. A. Simon, A. J. Halperin, H. P. Baden, P. E. Shapiro, A. E. Bale et al., Mutation hotspots due to sunlight in the p53 gene of nonmelanoma skin cancers, Proc. Natl. Acad. Sci. U. S. A., 1993, 90, 4216–4220.
E. Sage, E. A. Drobetsky, E. Moustacchi, 8-Methoxypsoralen induced mutations are highly targeted at crosslinkable sites of photoaddition on the non-transcribed strand of a mammalian chromosomal gene, EMBO J., 1993, 12, 397–402.
A. R. Young, I. A. Magnus, An Action Spectrum for 8-MOP Induced Sunburn Cells in Mammalian Epidermis, Br. J. Dermatol., 1981, 104, 541–548.
K. H. Kaidbey, An Action Spectrum for 8-Methoxypsoralen-Sensitized Inhibition of DNA-Synthesis In vivo, J. Invest. Dermatol., 1985, 85, 98–101.
R. M. Sayre, J. C. Dowdy, R. W. Gottschalk, Comparative effectiveness of clinically used light sources for cutaneous protoporphyrin IX-based photodynamic therapy, J. Cosmetic Laser Ther., 2011, 13, 63–68.
N. R. Attard, P. Karran, UVA photosensitization of thiopurines and skin cancer in organ transplant recipients, Photochem. Photobiol. Sci., 2012 10.1039/C1PP05194F.
A. Tewari, C. Lahmann, R. Sarkany, J. Bergemann, A. R. Young, Human erythema and matrix metalloproteinase-1 mRNA induction, in vivo, share an action spectrum which suggests common chromophores, Photochem. Photobiol. Sci., 2012 10.1039/C1PP05243H.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contribution to the themed issue on the biology of UVA.
Rights and permissions
About this article
Cite this article
Reelfs, O., Karran, P. & Young, A.R. 4-thiothymidine sensitization of DNA to UVA offers potential for a novel photochemotherapy. Photochem Photobiol Sci 11, 148–154 (2012). https://doi.org/10.1039/c1pp05188a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c1pp05188a