Abstract
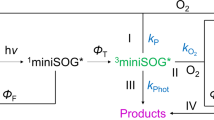
The fluorescent protein KillerRed generates reactive oxygen species through the CALI effect. This property paves the way for the design of genetically encoded photosensitizers for use in cell killing and cancer photodynamic therapy. In this article, we have investigated the diffusion pathways of di-oxygen and the superoxide radical in KillerRed, using molecular dynamics simulations. Our results suggest that, by comparison to the Ser-65-Thr mutant of GFP, diffusion of molecular oxygen (and singlet oxygen) is greatly facilitated in KillerRed, mostly due to the presence of a unique water-filled channel. In contrast, due to their negative charge, superoxide radical ions putatively produced inside the chromophore pocket are unable to escape the protein. These results are consistent with the hypothesis that superoxide generation, if it occurs, proceeds via light-induced photoreduction of the chromophore followed by long-range electron transfer, a mechanism in which the long hydrogen bond network through the channel could play a key role. Alternatively, the facilitated diffusion of di-oxygen through the channel suggests that singlet di-oxygen could be the principal cause of specific CALI of fused proteins. The entry of di-oxygen through the channel probably also accounts for the high susceptibility of KillerRed to photobleaching.
Similar content being viewed by others
Abbreviations
- GFP:
-
Green Fluorescent Protein
- EGFP:
-
Enhanced Green Fluorescent Protein
- LES:
-
Locally Enhanced Sampling
- CALI:
-
Chromophore-Assisted Light Inactivation
- PDB:
-
Protein Data Bank
- ROS:
-
Reactive Oxygen Species
- RMSD:
-
Root Mean Square Deviation
References
M. E. Bulina, D. M. Chudakov, O. V. Britanova, Y. G. Yanushevich, D. B. Staroverov, T. V. Chepurnykh, E. M. Merzlyak, M. A. Shkrob, S. Lukyanov, K. A. Lukyanov, Nat. Biotechnol., 2006, 24, 95–99.
K. Jacobson, Z. Rajfur, E. Vitriol, K. Hahn, Trends Cell Biol., 2008, 18, 443–450.
B. A. Griffin, S. R. Adams, R. Y. Tsien, Science, 1998, 281, 269–272.
S. R. Adams, R. E. Campbell, L. A. Gross, B. R. Martin, G. K. Walkup, Y. Yao, J. Llopis, R. Y. Tsien, J. Am. Chem. Soc., 2002, 124, 6063–6076.
M. E. Bulina, K. A. Lukyanov, O. V. Britanova, D. Onichtchouk, S. Lukyanov, D. M. Chudakov, Nat. Protoc., 2006, 1, 947–953.
E. O. Serebrovskaya, E. F. Edelweiss, O. A. Stremovskiy, K. A. Lukyanov, D. M. Chudakov, S. M. Deyev, Proc. Natl. Acad. Sci. U. S. A., 2009, 106, 9221–9225.
E. Horstkotte, T. Schroder, J. Niewohner, E. Thiel, D. G. Jay, S. W. Henning, Photochem. Photobiol., 2005, 81, 358–366.
A. Jimenez-Banzo, S. Nonell, J. Hofkens, C. Flors, Biophys. J., 2008, 94, 168–172.
D. Yarbrough, R. M. Wachter, K. Kallio, M. V. Matz, S. J. Remington, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 462–467.
S. Pletnev, N. G. Gurskaya, N. V. Pletneva, K. A. Lukyanov, D. M. Chudakov, V. I. Martynov, V. O. Popov, M. V. Kovalchuk, A. Wlodawer, Z. Dauter, V. Pletnev, J. Biol. Chem., 2009, 284, 32028–32039.
V. Adam, P. Carpentier, S. Violot, M. Lelimousin, C. Darnault, G. U. Nienhaus, D. Bourgeois, J. Am. Chem. Soc., 2009, 131, 18063–18065.
F. V. Subach, V. N. Malashkevich, W. D. Zencheck, H. Xiao, G. S. Filonov, S. C. Almo, V. V. Verkhusha, Proc. Natl. Acad. Sci. U. S. A., 2009, 106, 21097–21102.
A. M. Bogdanov, A. S. Mishin, I. V. Yampolsky, V. V. Belousov, D. M. Chudakov, F. V. Subach, V. V. Verkhusha, S. Lukyanov, K. A. Lukyanov, Nat. Chem. Biol., 2009, 5, 459–461.
P. Carpentier, S. Violot, L. Blanchoin, D. Bourgeois, FEBS Lett., 2009, 583, 2839–2842.
F. V. Subach, G. H. Patterson, M. Renz, J. Lippincott-Schwartz, V. V. Verkhusha, J. Am. Chem. Soc., 2010, 132, 6481–6491.
R. Heim, D. C. Prasher, R. Y. Tsien, Proc. Natl. Acad. Sci. U. S. A., 1994, 91, 12501–12504.
A. G. Evdokimov, M. E. Pokross, N. S. Egorov, A. G. Zaraisky, I. V. Yampolsky, E. M. Merzlyak, A. N. Shkoporov, I. Sander, K. A. Lukyanov, D. M. Chudakov, EMBO Rep., 2006, 7, 1006–1012.
K. Nienhaus, F. Renzi, B. Vallone, J. Wiedenmann, G. U. Nienhaus, Biochemistry, 2006, 45, 12942–12953.
R. M. Wachter, M. A. Elsliger, K. Kallio, G. T. Hanson, S. J. Remington, Structure, 1998, 6, 1267–1277.
R. Elber, M. Karplus, J. Am. Chem. Soc., 1990, 112, 9161–9175.
R. Heim, A. B. Cubitt, R. Y. Tsien, Nature, 1995, 373, 663–664.
J. C. Phillips, R. Braun, W. Wang, J. Gumbart, E. Tajkhorshid, E. Villa, C. Chipot, R. D. Skeel, L. Kale, K. Schulten, J. Comput. Chem., 2005, 26, 1781–1802.
M. Brunori, F. Cutruzzola, C. Savino, C. Travaglini-Allocatelli, B. Vallone, Q. H. Gibson, Biophys. J., 1999, 76, 1259–1269.
J. Cohen, A. Arkhipov, R. Braun, K. Schulten, Biophys. J., 2006, 91, 1844–1857.
P. Amara, P. Andreoletti, H. M. Jouve, M. J. Field, Protein Sci., 2001, 10, 1927–1935.
A. D. Mackerell Jr., D. Bashford, M. Bellott, R. L. Dunbrack Jr., J. D. Evanseck, M. J. Field, S. Fischer, J. Gao, H. Guo, S. Ha, J. Phys. Chem. B, 1998, 102, 3586–3616.
N. Reuter, H. Lin, W. Thiel, J. Phys. Chem. B, 2002, 106, 6310–6321.
W. Humphrey, A. Dalke, K. Schulten, J. Mol. Graphics, 1996, 14, 33–38.
M. Lelimousin, M. Noirclerc-Savoye, C. Lazareno-Saez, B. Paetzold, S. Le Vot, R. Chazal, P. Macheboeuf, M. J. Field, D. Bourgeois, A. Royant, Biochemistry, 2009, 48, 10038–10046.
G. J. Kleywegt, T. A. Jones, Acta Crystallogr., Sect. D: Biol. Crystallogr., 1994, 50, 178–185.
Collaborative Computational Project, Number 4, The CCP4 suite: programs for protein crystallography, Acta Crystallogr., Sect. D: Biol. Crystallogr., 1994, 50, 760–763.
R. Y. Tsien, Annu. Rev. Biochem., 1998, 67, 509–544.
M. Gardès-Albert, D. Bonnefont–Rousselot, Z. Abedinzadeh, D. Jore, Actual. Chim., 2003, 269–270, 91–96.
S. Luo, R. L. Levine, FASEB J., 2008, 23, 464–472.
F. Wilkinson, W. P. Helman, A. B. Ross, J. Phys. Chem. Ref. Data, 1995, 24, 663–680.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published as part of a themed issue on photofunctional proteins: from understanding to engineering.
‡ Electronic supplementary information (ESI) available: Distance, energy and RMSD analyses of the molecular dynamics simulations. See DOI: 10.1039/c0pp00141d
Rights and permissions
About this article
Cite this article
Roy, A., Carpentier, P., Bourgeois, D. et al. Diffusion pathways of oxygen species in the phototoxic fluorescent protein Killer Red. Photochem Photobiol Sci 9, 1342–1350 (2010). https://doi.org/10.1039/c0pp00141d
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c0pp00141d