Abstract
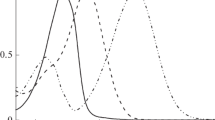
all-trans-1,4-Diindanylidenyl-2-butene (ttt-stiff-5-DPH), a torsionally constrained analogue of all-trans-1,6-diphenyl-1,3,5-hexatriene (ttt-DPH), was synthesized and studied in order to evaluate the role of phenyl–vinyl torsional motions in the photophysical and photochemical responses of the DPH chromophore. Spectroscopic and photoisomerization measurements reveal that the behavior of the rigid DPH analogue is very similar to that of the parent DPH. This similarity is obtained despite the fact that the alkyl substitution from the five-membered rings selectively lowers the energy of the 1 1Bu* state, leading to inversion of the order of the 1 1Bu* and 2 1Ag* energy levels in hydrocarbon solvents. In stiff-5-DPH, as in DPH, an increase in solvent polarity enhances terminal over central bond photoisomerization. Analyses of fluorescence and photoisomerization quantum yields show that, as in DPH, the torsional relaxation channel on the singlet excited state manifold is inefficient, falling far short of accounting for all radiationless decay. Significant (∼50 and 80% of all singlet decay in Bz and AN, respectively), photochemically unproductive, radiationless decay channels exist in both molecules. Competing one bond photoisomerizations give the two major photoproducts: tct-stiff-5-DPH and ctt-stiff-5-DPH. They were isolated in pure form and were spectroscopically characterized. Biacetyl-sensitization was used to study the behavior of the stiff-5-DPH triplet state. As in the parent DPH, stiff-5-DPH triplets undergo relatively efficient concentration dependent geometric photoisomerization.
Similar content being viewed by others
References
For a review, see J. Saltiel and Y.-P. Sun, Cis-trans isomerization of C,C double bonds, in Photochromism, Molecules and Systems, ed. H. Dürr and H. Bouas-Laurent, Elsevier, Amsterdam, 1990, pp. 64–164.
B. S. Hudson, B. E. Kohler, Linear polyene electronic structure and spectroscopy, Annu. Rev. Phys. Chem., 1974, 25, 437–460.
B. S. Hudson, B. E. Kohler and K. Schulten, Linear polyene electronic structure and potential surfaces, in Excited States, ed. E. C. Lim, Academic Press, New York, 1982, vol. 6, pp. 1–95.
E. F. Hilinski, W. M. McGowan, D. F. Sears, Jr., J. Saltiel, Evolutions of singlet excited state absorption and fluorescence of all-trans-1,6-diphenyl-1,3,5-hexatriene in the picosecond time domain, J. Phys. Chem., 1996, 100, 3308–3311.
W. A. Yee, R. H. O’Neil, J. W. Lewis, J. Z. Zhang, D. S. Kliger, Femtosecond transient absorption studies of diphenylpolyenes. Direct detection of S2 → S1 radiationless conversion in diphenylhexatriene and diphenyloctatetraene, Chem. Phys. Lett., 1997, 276, 430–434.
S. Hogiu, W. Werneke, M. Pfeiffer, A. Lau, T. Steinke, Picosecond time-resolved CARS spectroscopy of a mixed excited singlet state of diphenylhexatriene, Chem. Phys. Lett., 1998, 287, 8–16.
J. R. Andrews, B. S. Hudson, Environmental effects on radiative rate constants with applications to linear polyenes, J. Chem. Phys., 1978, 68, 4587–4594.
J. B. Birks, G. N. R. Tripathi, M. D. Lumb, The fluorescence of all-trans-diphenylpolyenes, Chem. Phys., 1978, 33, 185–194.
M. Pfeiffer, W. Werncke, S. Hogiu, A. Kummrow, A. Lau, Strong vibronic coupling in the first excited singlet state of diphenylhexatriene by an asymmetric low-frequency mode, Chem. Phys. Lett., 1998, 295, 56–62
S. Hogiu, W. Werncke, M. Pfeiffer, A. Lau, Evidence of strong vibronic coupling in the first excited singlet state of diphenylhexatriene by picosecond CARS spectroscopy, Chem. Phys. Lett., 1999, 303, 218–222
W. Werncke, S. Hogiu, M. Pfeiffer, Strong S1–S2 vibronic coupling and enhanced third order hyperpolarizability in the first excited singlet state of diphenylhexatriene studied by time-resolved CARS, A. Lau, and A. Kummrow, J. Phys. Chem. A, 2000, 104, 4211–4217.
P. C. Alford, T. F. Palmer, Fluorescence of DPH derivatives—evidence for emission from S2 and S1 excited-states, Chem. Phys. Lett., 1982, 86, 248–253.
P. C. Alford, T. F. Palmer, Photophysics of derivatives of all-trans-1,6-diphenyl-1,3,5-hexatriene (DPH). 1. Model involving fluorescence from S2 and S1 excited-states, J. Chem. Soc., Faraday Trans. 2, 1983, 79, 433–447.
T. Itoh, B. E. Kohler, Dual fluorescence of diphenylpolyenes, J. Phys. Chem., 1987, 91, 1760–1764.
J. Saltiel, D. F. Sears, Jr., Y.-P. Sun, J.-O. Choi, Evidence for ground state s-cis-conformers in the fluorescence spectra of all-trans-1,6-diphenyl-1,3,5-hexatriene, J. Am. Chem. Soc., 1992, 114, 3607–3612.
A. M. Turek, G. Krishnamoorthy, D. F. Sears, Jr., I. Garcia, O. Dmitrenko, J. Saltiel, Resolution of three fluorescence components in the spectra of all-trans-1,6-diphenyl-1,3,5-hexatriene under isopolarizability conditions, J. Phys. Chem. A, 2005, 109, 293–303.
J. Saltiel, S. Wang, D.-H. Ko, D. A. Gormin, Cis–trans photoisomerization of the 1,6-diphenyl-1,3,5-hexatrienes in the triplet state. The quantum chain mechanism and the structure of the triplet state, J. Phys. Chem. A, 1998, 102, 5383–5392
J. Saltiel, J. M. Crowder, S. Wang, Mapping the potential energy surfaces of the 1,6-diphenyl-1,3,5-hexatriene ground and triplet states, J. Am. Chem. Soc., 1999, 121, 895–902, 5352.
J. Saltiel, D.-H. Ko, S. A. Fleming, Differential medium effects on the trans to cis photoisomerization of all-trans-1,6-diphenyl-1,3,5-hexatriene. Competing diradicaloid vs zwitterionic pathways, J. Am. Chem. Soc., 1994, 116, 4099–4100
J. Saltiel, S. Wang, L. P. Watkins, D.-H. Ko, Direct photoisomerization of the 1,6-diphenyl-1,3,5-hexatrienes. Medium effect on triplet and singlet contributions, J. Phys. Chem. A, 2000, 104, 11443–11450.
J. Saltiel, G. Krishnamoorthy, Z. Huang, D.-H. Ko, S. Wang, The photoisomerization of all-trans-1,6-diphenyl-1,3,5-hexatriene. Temperature and deuterium isotope effects, J. Phys. Chem. A, 2003, 107, 3178–3186.
J. Saltiel, G. Krishnamoorthy, Z. Huang, D.-H. Ko, S. Wang, Stereoselective O2-induced photoisomerization of all-trans-1,6-diphenyl-1,3,5-hexatriene, Can. J. Chem., 2003, 81, 673–679.
J. B. Birks, D. J. S. Birch, The fluorescence of diphenyl- and retinol-polyenes, Chem. Phys. Lett., 1975, 31, 608–610
J. B. Birks, Horizontal radiationless transitions, Chem. Phys. Lett., 1978, 54, 430–434.
G. Orlandi, W. Siebrand, Model for the direct photoisomerization of stilbene, Chem. Phys. Lett., 1975, 30, 352–354.
J. Saltiel, Perdeuteriostilbene. The role of phantom states in the cis-trans photoisomerization of stilbenes, J. Am. Chem. Soc., 1967, 89, 1036–1037
J. Saltiel, Perdeuteriostilbene. The triplet and singlet paths for stilbene photoisomerization, J. Am. Chem. Soc., 1968, 90, 6394–6400.
C. Rullière, A. Declémy, Picosecond photophysics of diphenylpolyenes: evidence for the influence of excited-state conformational changes on the energy gap δE(Bu*-Ag*), Chem. Phys. Lett., 1987, 135, 213–218.
J. Saltiel, S. Wang, Absorption and fluorescence spectra of a rigid analogue of all-trans-1,6-diphenyl-1,3,5-hexatriene. Solvent controlled order inversion of 2 1Ag and 1 1Bu energy levels, J. Am. Chem. Soc., 1995, 117, 10761–10762.
G. Wittig, A. Hesse, Directed aldol condensation: β-phenylcinnamaldehyde, Org. Synth., 1970, 50, 66–72.
J. E. McMurry, M. P. Fleming, New method for the reductive coupling of carbonyls to olefins. Synthesis of.beta.-carotene, J. Am. Chem. Soc., 1974, 96, 4708–4709
J. E. McMurry, Organic chemistry of low-valent titanium, Acc. Chem. Res., 1974, 7, 281.
J. Saltiel, D. F. Sears, Jr., D. -H Ko and K.-M. Park, Cis-trans isomerization of alkenes, in Handbook of Organic Photochemistry and Photobiology, ed. W. M. Horspool and P.-O. Song, CRC Press, London, 1995, Section 1, pp. 3–15.
F. G. Moses, R. S. H. Liu, B. M. Monroe, The ‘merry-go-round’ quantum yield apparatus, Mol. Photochem., 1969, 1, 245–249.
H. A. Hammond, D. E. DeMeyer, J. L. R. Williams, Quantum yields for the sensitized photoisomerization of cis- and trans-stilbene, J. Am. Chem. Soc., 1969, 91, 5180–5181
D. Valentine, Jr., G. S. Hammond, Energy wastage in photosensitized isomerizations of the stilbenes, J. Am. Chem. Soc., 1972, 94, 3449–3454.
A. A. Lamola, G. S. Hammond, Intersystem crossing efficiencies, J. Chem. Phys., 1965, 43, 2129–2135
J. Saltiel, A. Marinari, D. W.-L. Chang, J. C. Mitchener, E. D. Megarity, Trans-cis photoisomerization of the stilbenes and a reexamination of the positional dependence of the heavy-atom effect, J. Am. Chem. Soc., 1979, 101, 2982–2996.
J. Saltiel, D. F. Sears, Jr., J.-O. Choi, Y.-P. Sun, D. W. Eaker, The fluorescence, fluorescence-excitation and UV absorption spectra of trans-1-(2-naphthyl)-2-phenylethene conformers, J. Phys. Chem., 1994, 98, 35–46.
A. Marinari, J. Saltiel, A fluorescence technique for the separation of radiative from non-radiative energy transfer. Its application in the trans-stilbene/azulene system, Mol. Photochem., 1976, 7, 225–249.
For figures of the 1H NMR spectra see S. Wang, The photoisomerization of 1,6-diphenylhexatrienes Ph. D. Dissertation, 1998, Florida State University, Tallahassee, FL.
K. Lunde, L. Zechmeister, Cis–trans isomeric 1,6-diphenylhexatrienes, J. Am. Chem. Soc., 1954, 76, 2308–2313.
IUPAC Analytical Chemistry Division, Commission on Solubility Data, Oxygen and ozone, in Solubility Data Series, ed. R. Battino, Pergamon, Oxford, vol. 7, 1981.
W. D. K. Clark, C. Steele, Photochemistry of 2,3-diazabicyclo[2.2.2]oct-2-ene, J. Am. Chem. Soc., 1971, 93, 6347–6355.
J. Saltiel, J. T. D’Agostino, Separation of viscosity and temperature effects on the singlet pathway to stilbene photoisomerization, J. Am. Chem. Soc., 1972, 94, 6445–6457.
M. Lee, J. N. Haseltine, A. B. Smith, R. M. Hochstrasser, Isomerization processes of electronically excited stilbene and diphenylbutadiene in liquids. Are they one-dimensional?, J. Am. Chem. Soc., 1989, 111, 5044–5051.
S. E. Wallace-Williams, B. J. Schwartz, S. Møller, R. A. Goldbeck, W. A. Yee, M. A. El-Bayoumi, D. S. Kliger, Excited state spectra and dynamics of phenyl-substituted butadienes, J. Phys. Chem., 1994, 98, 60–67.
B. Jousselme, P. Blanchard, P. Frère, J. Roncali, Enhancement of the π-electron delocalization and fluorescence efficiency of 1,6-diphenyl-1,3,5-hexatriene by covalent rigidification, Tetrahedron Lett., 2000, 41, 5057–5061.
H. L. J. Bäckstrom, K. Sandros, Transfer of triplet state energy in fluid solutions. 1. Sensitized phosphorescence and its application to the determination of triplet state lifetimes, Acta Chem. Scand., 1960, 14, 48–62.
Parallel irradiations of degassed solutions of biacetyl and trans-stilbene (0.201 and 5.00 × 10−3 M, respectively) and benzophenone and trans-stilbene (0.0204 and 0.0101 M, respectively) in a merry-go-round at 366 and 370 nm gave ftrans → cis = 0.502 for biacetyl-sensitization of stilbene isomerization, indicating fis = 0.91, a somewhat lower value than that given in ref. 35.
E. D. Cehelnik, R. B. Cundall, J. R. Lockwood, T. J. Palmer, Solvent and temperature effects on the fluorescence of all-trans-1,6-diphenyl-1,3,5-hexatriene, J. Phys. Chem., 1975, 79, 1369–1380.
D. H. Ko, Photochemistry of all-trans 1,6-diphenyl-1,3,5-hexatriene Ph.D. Thesis, 1997, Florida State University, Tallahassee, FL, 32306-4390.
J. B. Birks, D. J. Dyson, Relations between fluorescence and absorption properties of organic molecules, Proc. R. Soc. London, Ser. A, 1963, 275, 135–148.
S. J. Strickler, R. A. Berg, Relationship between absorption intensity and fluorescence lifetime of molecules, J. Chem. Phys., 1962, 37, 814–822.
B. S. Hudson, B. E. Kohler, Polyene spectroscopy: the lowest energy excited singlet state of diphenyloctatetraene and other linear polyenes, J. Chem. Phys., 1973, 59, 4984–5002.
We thank L. P. Watkins for this calculation.
J. Saltiel, A. S. Waller, D. F. Sears, Jr., C. Z. Garrett, Fluorescence quantum yields of trans-stilbene-d0 and -d2 in n-hexane and n-tetradecane. Medium and deuterium isotope effects on decay processes, J. Phys. Chem., 1993, 97, 2516–2522.
L. A. Sklar, B. S. Hudson, M. Petersen, J. Diamond, Conjugated polyene fatty acids on fluorescent probes: spectroscopic characterization, Biochemistry, 1977, 16, 813–818.
J. Timmermans, Physico-Chemical Constants of Pure Organic Compounds, Elsevier, New York, 1950, vol. 1 and vol. 2.
M. T. Allen, L. Miola, D. G. Whitten, Temperature effects on fluorescence in diphenylpolyene derivatives: structure- and substituent-dependent changes in mechanisms and rates for nonradiative decay, J. Phys. Chem., 1987, 91, 6099–6102.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper was published as part of the special issue in honour of the late Professor George S. Hammond.
Rights and permissions
About this article
Cite this article
Saltiel, J., Wang, S. The photochemistry and photophysics of all-trans-1,4-diindanylidenyl-2-butene, a rigid analogue of all-trans-1,6-diphenyl-1,3,5-hexatriene. Photochem Photobiol Sci 5, 883–895 (2006). https://doi.org/10.1039/b608065k
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/b608065k