Abstract
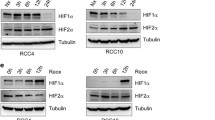
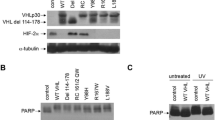
Loss of the von Hippel-Lindau (VHL) tumor suppressor gene contributes to proliferative disorders including renal cell carcinoma. The consequence of VHL loss is increased levels of hypoxia-inducible factor-α (HIFα), which is targeted for proteolytic degradation by the VHL gene product pVHL. HIF is a transcription factor that increases the expression of factors critical for tumorigenesis in renal cell carcinoma. We report here another regulatory component of HIFα expression in renal cancer cells. Phospholipase D (PLD), which is commonly elevated in renal and other cancers, is required for elevated levels of both HIF1α and HIF2α in VHL-deficient renal cancer cells. The induction of both HIF1α and HIF2α by hypoxic mimetic conditions was also dependent on PLD in renal cancer cells with restored pVHL expression. The effect of PLD activity upon HIFα expression was at the level of translation. PLD activity also provides a survival signal that suppresses apoptosis induced by serum deprivation in the renal cancer cells. Suppression of HIF2α has been shown to reverse tumorigenesis with renal cancer cells. The finding here that HIF2α expression is dependent on PLD in renal cancer cells suggests that targeting PLD signals may represent an alternative therapeutic strategy for targeting HIF2α in renal cancers where HIF2α is critical for tumorigenesis and elevated PLD activity is common.






Similar content being viewed by others
References
Bernardi R, Guernah I, Jin D, Grisendi S, Alimonti A, Teruya-Feldstein J et al. (2006). PML inhibits HIF-1α translation and neoangiogenesis through repression of mTOR. Nature 442: 779–785.
Chen Y, Rodrik V, Foster DA . (2005). Alternative phospholipase D/mTOR survival signal in human breast cancer cells. Oncogene 24: 672–679.
Chen Y, Zheng Y, Foster DA . (2003). Phospholipase D confers rapamycin resistance in human breast cancer cells. Oncogene 22: 3937–3942.
Colley WC, Sung TC, Roll R, Jenco J, Hammond SM, Altshuller Y et al. (1997). Phospholipase D2, a distinct phospholipase D isoform with novel regulatory properties that provokes cytoskeletal reorganization. Curr Biol 7: 191–201.
Fang Y, Vilella-Bach M, Bachmann R, Flanigan A, Chen J . (2001). Phosphatidic acid-mediated mitogenic activation of mTOR signaling. Science 294: 1942–1945.
Foster DA . (2004). Targeting mTOR-mediated survival signals in anticancer therapeutic strategies. Exp Rev Anticancer Ther 4: 691–701.
Foster DA . (2006). Phospholipase D survival signals as a therapeutic target in cancer. Curr Sig Trans Ther 1: 295–303.
Foster DA . (2007). Regulation of mTOR by phosphatidic acid? Cancer Res 67: 1–4.
Foster DA, Xu L . (2003). Phospholipase D in cell proliferation and cancer. Mol Cancer Res 1: 789–800.
Gatenby RA, Gillies RJ . (2004). Why do cancers have high aerobic glycolysis? Nat Rev Cancer 4: 891–899.
Gordan JD, Simon MC . (2007). Hypoxia-inducible factors: central regulators of the tumor phenotype. Curr Opin Genet Dev 17: 71–77.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hickey MM, Simon MC . (2006). Regulation of angiogenesis by hypoxia and hypoxia-inducible factors. Curr Top Dev Biol 76: 217–257.
Hui L, Abbas T, Pielak R, Joseph T, Bargonetti J, Foster DA . (2004). Phospholipase D elevates the level of MDM2 and suppresses DNA damage-induced increases in p53. Mol Cell Biol 24: 5677–5688.
Hui L, Rodrik V, Pielak RM, Zheng Y, Foster DA . (2005). mTOR-dependent suppression of protein phosphatase 2A is critical for phospholipase D survival signals in human breast cancer cells. J Biol Chem 280: 35829–35835.
Kaelin Jr WG . (2005). The von Hippel-Lindau protein, HIF hydroxylation, and oxygen sensing. Biochem Biophys Res Commun 338: 627–638.
Kaelin Jr WG . (2007). The von Hippel-Lindau tumor suppressor protein and clear cell renal carcinoma. Clin Cancer Res 13: 680s–684s.
Kondo K, Kim WY, Lechpammer M, Kaelin Jr WG . (2003). Inhibition of HIF2α is sufficient to suppress pVHL-defective tumor growth. PLoS Biol 1: 439–444.
Kondo K, Klco J, Nakamura E, Lechpammer M, Kaelin Jr WG . (2002). Inhibition of HIF is necessary for tumor suppression by the von Hippel-Lindau protein. Cancer Cell 1: 237–246.
Lonergan KM, Iliopoulos O, Ohh M, Kamura T, Conaway RC, Conaway JW et al. (1998). Regulation of hypoxia-inducible mRNAs by the von Hippel-Lindau tumor suppressor protein requires binding to complexes containing elongins B/C and Cul2. Mol Cell Biol 18: 732–741.
Maxwell PH . (2005). The HIF pathway in cancer. Semin Cell Dev Biol 16: 523–530.
Maynard MA, Ohh M . (2004). Von Hippel-Lindau tumor suppressor protein and hypoxia-inducible factor in kidney cancer. Am J Nephrol 24: 1–13.
Ohh M . (2006). Ubiquitin pathway in VHL cancer syndrome. Neoplasia 8: 623–629.
Ohh M, Yauch RL, Lonergan K, Whaley JM, Stemmer-Rachamimov AO, Louis DN et al. (1998). The von Hippel-Lindau tumor suppressor protein is required for proper assembly of an extracellular fibronectin matrix. Mol Cell 1: 959–968.
Rizzo MA, Shome K, Vasudevan C, Stolz DB, Sung TC, Frohman MA et al. (1999). Phospholipase D and its product, phosphatidic acid, mediate agonist-dependent raf-1 translocation to the plasma membrane and the activation of the mitogen-activated protein kinase pathway. J Biol Chem 274: 1131–1139.
Rodrik V, Gomes E, Hui L, Rockwell P, Foster DA . (2006). Myc stabilization in response to estrogen and phospholipase D in MCF-7 breast cancer cells. FEBS Lett 580: 5647–5652.
Rodrik V, Zheng Y, Harrow F, Chen Y, Foster DA . (2005). Survival signals generated by estrogen and phospholipase D in MCF-7 breast cancer cells are dependent on Myc. Mol Cell Biol 25: 7917–7925.
Sawyers CL . (2003). Will mTOR inhibitors make it as cancer drugs? Cancer Cell 4: 343–348.
Semenza GL . (2001). HIF-1 and mechanisms of hypoxia sensing. Curr Opin Cell Biol 13: 167–171.
Shaw RJ . (2006). Glucose metabolism and cancer. Curr Opin Cell Biol 18: 598–608.
Shen Y, Xu L, Foster DA . (2001). Phospholipase D requirement for receptor-mediated endocytosis. Mol Cell Biol 21: 595–602.
Stickle NH, Chung J, Klco JM, Hill RP, Kaelin Jr WG, Ohh M . (2004). pVHL modification by NEDD8 is required for fibronectin matrix assembly and suppression of tumor development. Mol Cell Biol 24: 3251–3261.
Sung TC, Altshuller YM, Morris AJ, Frohman MA . (1999). Molecular analysis of mammalian phospholipase D2. J Biol Chem 274: 494–502.
Sung TC, Roper RL, Zhang Y, Rudge SA, Temel R, Hammond SM et al. (1997). Mutagenesis of phospholipase D defines a superfamily including a trans-Golgi viral protein required for poxvirus pathogenicity. EMBO J 16: 4519–4530.
Zhao Y, Ehara H, Akao Y, Shamoto M, Nakagawa Y, Banno Y et al. (2000). Increased activity and intranuclear expression of phospholipase D2 in human renal cancer. Biochem Biophys Res Commun 278: 140–143.
Zheng Y, Rodrik V, Toschi A, Shi M, Hui L, Shen Y et al. (2006). Phospholipase D couples survival and migration signals in response to stress in human breast cancer cells. J Biol Chem 281: 15862–15868.
Zhong M, Shen Y, Zheng Y, Joseph T, Jackson D, Beychenok S et al. (2003). Phospholipase D prevents apoptosis in v-Src-transformed rat fibroblasts and MDA-MB-231 breast cancer cells. Biochem Biophys Res Comm 302: 615–619.
Acknowledgements
This manuscript is dedicated to Dr Allan Stanley, whose extended battle with renal cancer was the inspiration for much of this study. We thank Michael Frohman (SUNY, Stony Brook) for the PLD genes used in this study. This work was supported by grants from the National Cancer Institute (CA46677) and a SCORE grant from the National Institutes of Health (GM60654) (DAF), and grants from the Canadian Cancer Society of the National Cancer Institute of Canada (MO). Research Centers in Minority Institutions award RR-03037 from the National Center for Research Resources of the National Institutes of Health, which supports infrastructure and instrumentation in the Biological Sciences Department at Hunter College, is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Toschi, A., Edelstein, J., Rockwell, P. et al. HIFα expression in VHL-deficient renal cancer cells is dependent on phospholipase D. Oncogene 27, 2746–2753 (2008). https://doi.org/10.1038/sj.onc.1210927
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210927
- Springer Nature Limited
Keywords
This article is cited by
-
NADH elevation during chronic hypoxia leads to VHL-mediated HIF-1α degradation via SIRT1 inhibition
Cell & Bioscience (2023)
-
Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions
Signal Transduction and Targeted Therapy (2023)
-
High Expression of PhospholipaseD2 Induced by Hypoxia Promotes Proliferation of Colon Cancer Cells through Activating NF- κ Bp65 Signaling Pathway
Pathology & Oncology Research (2020)
-
Emerging roles of lipid metabolism in cancer metastasis
Molecular Cancer (2017)
-
Essential role for SphK1/S1P signaling to regulate hypoxia-inducible factor 2α expression and activity in cancer
Oncogenesis (2016)