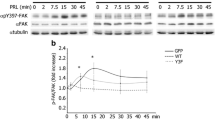
Abstract
Prolactin (PRL) stimulates the cytoskeletal re-organization and motility of breast cancer cells. During PRL receptor signaling, Vav2 becomes phosphorylated and activated, an event regulated by the serine/threonine kinase Nek3. Given the regulatory role of Vav2, the function of Nek3 in PRL-mediated motility and invasion was examined. Overexpression of Nek3 in Chinese hamster ovary transfectants potentiated cytoskeletal re-organization in response to PRL. In contrast, downregulation of Nek3 expression by small-interfering RNA (siRNA) attenuated PRL-mediated cytoskeletal reorganization, activation of GTPase Rac1, cell migration and invasion of T47D cells. In addition, PRL stimulation induced an interaction between Nek3 and paxillin and significantly increased paxillin serine phosphorylation, whereas Nek3 siRNA-transfected cells showed a marked reduction in paxillin phosphorylation. Analysis of breast tissue microarrays also demonstrated a significant up-regulation of Nek3 expression in malignant versus normal specimens. These data suggest that Nek3 contributes to PRL-mediated breast cancer motility through mechanisms involving Rac1 activation and paxillin phosphorylation.







Similar content being viewed by others
References
Abe K, Rossman KL, Liu B, Ritola KD, Chiang D, Campbell SL et al. (2000). Vav2 is an activator of Cdc42, Rac1, and RhoA. J Biol Chem 275: 10141–10149.
Acosta JJ, Munoz RM, Gonzalez L, Subtil-Rodriguez A, Dominguez-Caceres MA, Garcia-Martinez JM et al. (2003). Src mediates prolactin-dependent proliferation of T47D and MCF7 cells via the activation of focal adhesion kinase/Erk1/2 and phosphatidylinositol 3-kinase pathways. Mol Endocrinol 17: 2268–2282.
Beningo KA, Dembo M, Kaverina I, Small JV, Wang YL . (2001). Nascent focal adhesions are responsible for the generation of strong propulsive forces in migrating fibroblasts. J Cell Biol 153: 881–888.
Bhatavdekar JM, Shah NG, Balar DB, Patel DD, Bhaduri A, Trivedi SN et al. (1990). Plasma prolactin as an indicator of disease progression in advanced breast cancer. Cancer 65: 2028–2032.
Bouzahzah B, Albanese C, Ahmed F, Pixley F, Lisanti MP, Segall JD et al. (2001). Rho family GTPases regulate mammary epithelium cell growth and metastasis through distinguishable pathways. Mol Med 7: 816–830.
Brakebusch C, Fassler R . (2003). The integrin–actin connection, an eternal love affair. EMBO J 22: 2324–2333.
Brockman JL, Schroeder MD, Schuler LA . (2002). PRL activates the cyclin D1 promoter via the Jak2/Stat pathway. Mol Endocrinol 16: 774–784.
Brown MC, Turner CE . (2004). Paxillin: adapting to change. Physiol Rev 84: 1315–1339.
Burbelo PD, Drechsel D, Hall A . (1995). A conserved binding motif defines numerous candidate target proteins for both Cdc42 and Rac GTPases. J Biol Chem 270: 29071–29074.
Canbay E, Norman M, Kilic E, Goffin V, Zachary I . (1997). Prolactin stimulates the JAK2 and focal adhesion kinase pathways in human breast carcinoma T47-D cells. Biochem J 324: 231–236.
Clevenger CV, Furth PA, Hankinson SE, Schuler LA . (2003). The role of prolactin in mammary carcinoma. Endocr Rev 24: 1–27.
Clevenger CV, Russell DH, Appasamy PM, Prystowsky MB . (1990). Regulation of IL2-driven T-lymphocyte proliferation by prolactin. Proc Natl Acad Sci USA 87: 6460–6464.
del Pozo MA, Price LS, Alderson NB, Ren X-D, Schwartz MA . (2000). Adhesion to the extracellular matrix regulates the coupling of the small GTPase Rac to its effector PAK. EMBO J 19: 2008–2014.
DeMali KA, Wennerberg K, Burridge K . (2003). Integrin signaling to the actin cytoskeleton. Curr Opin Cell Biol 15: 572–582.
Fincham VJ, Wyke JA, Frame MC . (1995). v-Src-induced degradation of focal adhesion kinase during morphological transformation of chicken embryo fibroblasts. Oncogene 10: 2247–2252.
Fritz G, Just I, Kaina B . (1999). Rho GTPases are over-expressed in human tumors. Int J Cancer 81: 682–687.
Gao J, Horseman ND . (1999). Prolactin-independent modulation of the beta-casein response element by Erk2 MAP kinase. Cell Signal 11: 205–210.
Gordon LA, Mulligan KT, Maxwell-Jones H, Adams M, Walker RA, Jones JL . (2003). Breast cell invasive potential relates to the myoepithelial phenotype. Int J Cancer 106: 8–16.
Hall A . (1998). Rho GTPases and the actin cytoskeleton. Science 279: 509–514.
Holtkamp W, Nagel GA, Wander HE, Rauschecker HF, von Heyden D . (1984). Hyperprolactinemia is an indicator of progressive disease and poor prognosis in advanced breast cancer. Int J Cancer 34: 323–328.
Howard TH, Oresajo CO . (1985). A method for quantifying F-actin in chemotactic peptide activated neutrophils: study of the effect of tBOC peptide. Cell Motil 5: 545–557.
Huang C, Rajfur Z, Borchers C, Schaller MD, Jacobson K . (2003). JNK phosphorylates paxillin and regulates cell migration. Nature 424: 219–223.
Ilic D, Furuta Y, Kanazawa S, Takeda N, Sobue K, Nakatsuji N et al. (1995). Reduced cell motility and enhanced focal adhesion contact formation in cells from FAK-deficient mice. Nature 377: 539–544.
Inoue T, Cavanaugh PG, Steck PA, Brunner N, Nicolson GL . (1993). Differences in transferrin response and numbers of transferrin receptors in rat and human mammary carcinoma lines of different metastatic potentials. J Cell Physiol 156: 212–217.
Kline JB, Moore DJ, Clevenger CV . (2001). Activation and association of the Tec tyrosine kinase with the human prolactin receptor: mapping of a Tec/Vav1-receptor binding site. Mol Endocrinol 15: 832–841.
Kondo A, Hashimoto S, Yano H, Nagayama K, Mazaki Y, Sabe H . (2000). A new paxillin-binding protein, PAG3/Papalpha/KIAA0400, bearing an ADP-ribosylation factor GTPase-activating protein activity, is involved in paxillin recruitment to focal adhesions and cell migration. Mol Biol Cell 11: 1315–1327.
Lissoni P, Barni S, Cazzaniga M, Ardizzoia A, Rovelli F, Tancini G et al. (1995). Prediction of recurrence in operable breast cancer by postoperative changes in prolactin secretion. Oncology 52: 439–442.
Liu BP, Burridge K . (2000). Vav2 activates Rac1, Cdc42, and RhoA downstream from growth factor receptors but not b1 integrins. Mol Cell Biol 20: 7160–7169.
Marignani PA, Carpenter CL . (2001). Vav2 is required for cell spreading. J Cell Biol 154: 177–186.
Maus MV, Reilly SC, Clevenger CV . (1999). Prolactin as a chemoattractant for human breast carcinoma. Endocrinology 140: 5447–5450.
Miller SL, DeMaria JE, Freier DO, Riegel AM, Clevenger CV . (2005). Novel association of Vav2 and Nek3 modulates signaling through the human prolactin receptor. Mol Endocrinol 19: 939–949.
Montes dO, Macotela Y, Nava G, Lopez-Barrera F, de la Escalera GM, Clapp C . (2005). Prolactin stimulates integrin-mediated adhesion of circulating mononuclear cells to endothelial cells. Lab Invest 85: 633–642.
Nawrocki RB, Polette M, Gilles C, Clavel C, Strumane K, Matos M et al. (2001). Quantitative cell dispersion analysis: new test to measure tumor cell aggressiveness. Int J Cancer 93: 644–652.
Raftopoulou M, Hall A . (2004). Cell migration: Rho GTPases lead the way. Dev Biol 265: 23–32.
Ren XD, Kiosses WB, Sieg DJ, Otey CA, Schlaepfer DD, Schwartz MA . (2000). Focal adhesion kinase suppresses Rho activity to promote focal adhesion turnover. J Cell Sci 113 (Part 20): 3673–3678.
Ridley AJ, Paterson HF, Johnston CL, Diekmann D, Hall A . (1992). The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell 70: 401–410.
Roy S, Ruest PJ, Hanks SK . (2002). FAK regulates tyrosine phosphorylation of CAS, paxillin, and PYK2 in cells expressing v-Src, but is not a critical determinant of v-Src transformation. J Cell Biochem 84: 377–388.
Rycyzyn MA, Clevenger CV . (2002). The intranuclear prolactin/cyclophilin B complex as a transcriptional inducer. Proc Natl Acad Sci USA 99: 6790–6795.
Sahai E, Marshall CJ . (2002). Rho-GTPases and cancer. Nat Rev Cancer 2: 133–142.
Salcedo R, Martins-Green M, Gertz B, Oppenheim JJ, Murphy WJ . (2002). Combined administration of antibodies to human interleukin 8 and epidermal growth factor receptor results in increased antimetastatic effects on human breast carcinoma xenografts. Clin Cancer Res 8: 2655–2665.
Salcedo R, Ponce ML, Young HA, Wasserman K, Ward JM, Kleinman HK et al. (2000). Human endothelial cells express CCR2 and respond to MCP-1: direct role of MCP-1 in angiogenesis and tumor progression. Blood 96: 34–40.
Schmitz AA, Govek EE, Bottner B, Van Aelst L . (2000). Rho GTPases: signaling, migration, and invasion. Exp Cell Res 261: 1–12.
Schuebel KE, Bustelo XR, Nielsen DA, Song B-J, Barbacid M, Goldman D et al. (1996). Isolation and characterization of murine vav2, a member of the vav family of protooncogenes. Oncogene 13: 363–371.
Schuebel KE, Movilla N, Rosa JL, Bustelo XR . (1998). Phosphorylation-dependent and constitutive activation of Rho proteins by wild-type and oncogenic Vav-2. EMBO J 17: 6608–6621.
Tanaka K, Nigg EA . (1999). Cloning and characterization of the murine Nek3 protein kinase, a novel member of the NIMA family of putative cell cycle regulators. J Biol Chem 274: 13491–13497.
Turner CE, Brown MC, Perrotta JA, Riedy MC, Nikolopoulos SN, McDonald AR et al. (1999). Paxillin LD4 motif binds PAK and PIX through a novel 95-kD ankyrin repeat, ARF-GAP protein: a role in cytoskeletal remodeling. J Cell Biol 145: 851–863.
Turner CE, Miller JT . (1994). Primary sequence of paxillin contains putative SH2 and SH3 domain binding motifs and multiple LIM domains: identification of a vinculin and pp125Fak-binding region. J Cell Sci 107 (Part 6): 1583–1591.
Yoneda T, Sasaki A, Dunstan C, Williams PJ, Bauss F, Clerck YA et al. (1997). Inhibition of osteolytic bone metastasis of breast cancer by combined treatment with the bisphosphonate ibandronate and tissue inhibitor of the matrix metalloproteinase-2. J Clin Invest 99: 2509–2517.
Acknowledgements
We are grateful to Dr Michael E Hodsdon for receipt of recombinant PRL. This work was supported in part by grants from the NIH RO1CA69294 and RO1CA92265 (to CVC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miller, S., Antico, G., Raghunath, P. et al. Nek3 kinase regulates prolactin-mediated cytoskeletal reorganization and motility of breast cancer cells. Oncogene 26, 4668–4678 (2007). https://doi.org/10.1038/sj.onc.1210264
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210264
- Springer Nature Limited
Keywords
This article is cited by
-
Predictive and prognostic biomarkers of bone metastasis in breast cancer: current status and future directions
Cell & Bioscience (2023)
-
Analysis of the effect of NEKs on the prognosis of patients with non-small-cell lung carcinoma based on bioinformatics
Scientific Reports (2022)
-
Decidualized endometrial stromal cells present with altered androgen response in PCOS
Scientific Reports (2021)
-
Expression of the NEK family in normal and cancer tissue: an immunohistochemical study
BMC Cancer (2020)
-
Biallelic loss of function NEK3 mutations deacetylate α-tubulin and downregulate NUP205 that predispose individuals to cilia-related abnormal cardiac left–right patterning
Cell Death & Disease (2020)