Abstract
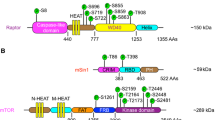
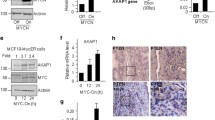
Survivin is a dual regulator of cell proliferation and cell viability overexpressed in most human tumors. Although strategies to lower survivin levels have been pursued for rational cancer therapy, the molecular circuitries controlling survivin expression in tumors have not been completely elucidated. Here, we show that stimulation with insulin-like growth factor-1 (IGF-1) results in increased survivin expression in prostate cancer cells. This response is independent of de novo gene transcription, changes in mRNA expression or modifications of survivin protein stability. Instead, IGF-1 induced persistence and translation of a pool of survivin mRNA, in a reaction abolished by the mTOR (mammalian target of rapamycin) inhibitor, rapamycin. Forced expression of the mTOR target p70S6K1 reproduced the increase in survivin expression in prostate cancer cells, whereas acute ablation of endogenous p70S6K1 by small interfering RNA downregulated survivin levels. Rapamycin, alone or in combination with suboptimal concentrations of taxol reduced survivin protein levels, and decreased viability of prostate cancer cells. Therefore, IGF-1/mTOR signaling elevates survivin in prostate cancer cells via rapid changes in mRNA translation. Antagonists of this pathway may be beneficial to lower an antiapoptotic threshold maintained by survivin in prostate cancer.






Similar content being viewed by others
References
Altieri DC . (2003). Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene 22: 8581–8589.
Altieri DC . (2006). Targeted therapy by disabling crossroad signaling networks: the survivin paradigm. Mol Cancer Ther 5: 478–482.
Amaravadi R, Thompson CB . (2005). The survival kinases Akt and Pim as potential pharmacological targets. J Clin Invest 115: 2618–2624.
Ambrosini G, Adida C, Altieri DC . (1997). A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 3: 917–921.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. (1999). Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96: 857–868.
Cory S, Adams JM . (2002). The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer 2: 647–656.
Dan HC, Jiang K, Coppola D, Hamilton A, Nicosia SV, Sebti SM et al. (2004). Phosphatidylinositol-3-OH kinase/AKT and survivin pathways as critical targets for geranylgeranyltransferase I inhibitor-induced apoptosis. Oncogene 23: 706–715.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y et al. (1997). Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91: 231–241.
Decker T, Hipp S, Ringshausen I, Bogner C, Oelsner M, Schneller F et al. (2003). Rapamycin-induced G1 arrest in cycling B-CLL cells is associated with reduced expression of cyclin D3, cyclin E, cyclin A, and survivin. Blood 101: 278–285.
Dohi T, Beltrami E, Wall NR, Plescia J, Altieri DC . (2004). Mitochondrial survivin inhibits apoptosis and promotes tumorigenesis. J Clin Invest 114: 1117–1127.
Fornaro M, Plescia J, Chheang S, Tallini G, Zhu YM, King M et al. (2003). Fibronectin protects prostate cancer cells from tumor necrosis factor. J Biol Chem 278: 50402–50411.
Goswami A, Burikhanov R, de Thonel A, Fujita N, Goswami M, Zhao Y et al. (2005). Binding and phosphorylation of par-4 by akt is essential for cancer cell survival. Mol Cell 20: 33–44.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hay N . (2005). The Akt-mTOR tango and its relevance to cancer. Cancer Cell 8: 179–183.
Hengartner MO . (2000). The biochemistry of apoptosis. Nature 407: 770–776.
Hoffman WH, Biade S, Zilfou JT, Chen J, Murphy M . (2002). Transcriptional repression of the anti-apoptotic survivin gene by wild type p53. J Biol Chem 277: 3247–3257.
Holcik M, Yeh C, Korneluk RG, Chow T . (2000). Translational upregulation of X-linked inhibitor of apoptosis (XIAP) increases resistance to radiation induced cell death. Oncogene 19: 4174–4177.
Hopfner M, Huether A, Sutter AP, Baradari V, Schuppan D, Scherubl H . (2006). Blockade of IGF-1 receptor tyrosine kinase has antineoplastic effects in hepatocellular carcinoma cells. Biochem Pharmacol 71: 1435–1448.
Kane LP, Mollenauer MN, Xu Z, Turck CW, Weiss A . (2002). Akt-dependent phosphorylation specifically regulates Cot induction of NF-kappa B-dependent transcription. Mol Cell Biol 22: 5962–5974.
Kane LP, Shapiro VS, Stokoe D, Weiss A . (1999). Induction of NF-kappaB by the Akt/PKB kinase. Curr Biol 9: 601–604.
Krajewska M, Krajewski S, Banares S, Huang X, Turner B, Bubendorf L et al. (2003). Elevated expression of inhibitor of apoptosis proteins in prostate cancer. Clin Cancer Res 9: 4914–4925.
Levine AJ, Feng Z, Mak TW, You H, Jin S . (2006). Coordination and communication between the p53 and IGF-1-AKT-TOR signal transduction pathways. Genes Dev 20: 267–275.
Li F, Ackermann EJ, Bennett CF, Rothermel AL, Plescia J, Tognin S et al. (1999). Pleiotropic cell-division defects and apoptosis induced by interference with survivin function. Nat Cell Biol 1: 461–466.
Li F, Altieri DC . (1999). Transcriptional analysis of human survivin gene expression. Biochem J 344 (Part 2): 305–311.
Li S, Resnicoff M, Baserga R . (1996). Effect of mutations at serines 1280-1283 on the mitogenic and transforming activities of the insulin-like growth factor I receptor. J Biol Chem 271: 12254–12260.
Majumder PK, Sellers WR . (2005). Akt-regulated pathways in prostate cancer. Oncogene 24: 7465–7474.
Mirza A, McGuirk M, Hockenberry TN, Wu Q, Ashar H, Black S et al. (2002). Human survivin is negatively regulated by wild-type p53 and participates in p53-dependent apoptotic pathway. Oncogene 21: 2613–2622.
Nam S, Buettner R, Turkson J, Kim D, Cheng JQ, Muehlbeyer S et al. (2005). Indirubin derivatives inhibit Stat3 signaling and induce apoptosis in human cancer cells. Proc Natl Acad Sci USA 102: 5998–6003.
O'Connor DS, Grossman D, Plescia J, Li F, Zhang H, Villa A et al. (2000). Regulation of apoptosis at cell division by p34cdc2 phosphorylation of survivin. Proc Natl Acad Sci USA 97: 13103–13107.
Reed JC . (2003). Apoptosis-targeted therapies for cancer. Cancer Cell 3: 17–22.
Romano G, Prisco M, Zanocco-Marani T, Peruzzi F, Valentinis B, Baserga R . (1999). Dissociation between resistance to apoptosis and the transformed phenotype in IGF-I receptor signaling. J Cell Biochem 72: 294–310.
Salvesen GS, Duckett CS . (2002). Apoptosis: IAP proteins: blocking the road to death's door. Nat Rev Mol Cell Biol 3: 401–410.
Sell C, Dumenil G, Deveaud C, Miura M, Coppola D, DeAngelis T et al. (1994). Effect of a null mutation of the insulin-like growth factor I receptor gene on growth and transformation of mouse embryo fibroblasts. Mol Cell Biol 14: 3604–3612.
Stromberg T, Ekman S, Girnita L, Dimberg LY, Larsson O, Axelson M et al. (2006). IGF-1 receptor tyrosine kinase inhibition by the cyclolignan PPP induces G2/M-phase accumulation and apoptosis in multiple myeloma cells. Blood 107: 669–678.
Vong QP, Cao K, Li HY, Iglesias PA, Zheng Y . (2005). Chromosome alignment and segregation regulated by ubiquitination of survivin. Science 310: 1499–1504.
Wullschleger S, Loewith R, Hall MN . (2006). TOR signaling in growth and metabolism. Cell 124: 471–484.
Yang S, Lim M, Pham LK, Kendall SE, Reddi AH, Altieri DC et al. (2006). Bone morphogenetic protein 7 protects prostate cancer cells from stress-induced apoptosis via both Smad and c-Jun NH2-terminal kinase pathways. Cancer Res 66: 4285–4290.
Zhang M, Latham DE, Delaney MA, Chakravarti A . (2005). Survivin mediates resistance to antiandrogen therapy in prostate cancer. Oncogene 24: 2474–2482.
Acknowledgements
We thank Drs J Blenis for providing p70S6K cDNA, and R Baserga for cells expressing wild type and mutant IGF-IR. This work was supported by NIH grants CA109874 (LRL), CA90917, CA78810 and HL54131 (DCA) and DOD-PCRP DAMD PC040221 (LRL).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vaira, V., Lee, C., Goel, H. et al. Regulation of survivin expression by IGF-1/mTOR signaling. Oncogene 26, 2678–2684 (2007). https://doi.org/10.1038/sj.onc.1210094
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210094
- Springer Nature Limited
Keywords
This article is cited by
-
Insulin-like growth factor receptor signaling in tumorigenesis and drug resistance: a challenge for cancer therapy
Journal of Hematology & Oncology (2020)
-
Insulin-like growth factor 1-induced enolase 2 deacetylation by HDAC3 promotes metastasis of pancreatic cancer
Signal Transduction and Targeted Therapy (2020)
-
Emerging Importance of Survivin in Stem Cells and Cancer: the Development of New Cancer Therapeutics
Stem Cell Reviews and Reports (2020)
-
Hexavalent chromium-induced autophagic death of WRL-68 cells is mitigated by aqueous extract of Cuminum cyminum L. seeds
3 Biotech (2020)
-
Early Cellular Responses of Prostate Carcinoma Cells to Sepantronium Bromide (YM155) Involve Suppression of mTORC1 by AMPK
Scientific Reports (2019)