Abstract
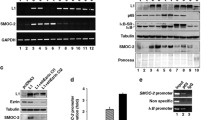
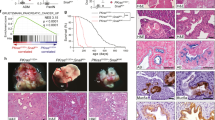
Inactivation of the transforming growth factor-β (TGF-β) pathway occurs often in malignancies of the gastrointestinal (GI) system. However, only a fraction of sporadic GI tumors exhibit inactivating mutations in early stages of cancer formation, suggesting that other mechanisms play a critical role in the inactivation of this pathway. Here, we show a wide range of GI tumors, including those of the stomach, liver and colon in elf+/− and elf+/−/Smad4+/− mutant mice. We found that embryonic liver fodrin (ELF), a β-Spectrin originally identified in endodermal stem/progenitor cells committed to foregut lineage, possesses potent antioncogenic activity and is frequently inactivated in GI cancers. Specifically, E-cadherin accumulation at cell–cell contacts and E-cadherin-β-catenin-dependent epithelial cell–cell adhesion is disrupted in elf+/−/Smad4+/− mutant gastric epithelial cells, and could be rescued by ectopic expression of full-length elf, but not Smad3 or Smad4. Subcellular fractionation revealed that E-cadherin is expressed mainly at the cell membrane after TGF-β stimulation. In contrast, elf+/−/Smad4+/− mutant tissues showed abnormal distribution of E-cadherin that could be rescued by overexpression of ELF but not Smad3 or Smad4. Our results identify a group of common lethal malignancies in which inactivation of TGF-β signaling, which is essential for tumor suppression, is disrupted by inactivation of the ELF adaptor protein.










Similar content being viewed by others
Change history
19 April 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41388-020-01632-1
Abbreviations
- ELF:
-
embryonic liver fodrin
- TGF-β:
-
transforming growth factor-β
- TβRII:
-
transforming growth factor-β receptor II
- TβRI:
-
transforming growth factor-β receptor I
- MEFs:
-
mouse embryonic fibroblasts
- HNPCC:
-
hereditary nonpolyposis colorectal cancer
- BrdU:
-
5-bromo-2′-deoxyuridine
- TUNEL:
-
TdT-mediated dUTP-biotin nick-end labeling
References
Attisano L, Wrana JL . (2000). Curr Opin Cell Biol 12: 235–243.
Bennett V, Baines AJ . (2001). Physiol Rev 81: 1353–1392.
Brooks-Wilson AR, Kaurah P, Suriano G, Leach S, Senz J, Grehan N et al. (2004). J Med Genet 41: 508–517.
Derynck R, Zhang YE . (2003). Nature 425: 577–584.
Dong C, Li Z, Alvarez Jr R, Feng XH, Goldschmidt-Clermont PJ . (2000). Mol Cell 5: 27–34.
Ehrlich JS, Hansen MD, Nelson WJ . (2002). Dev Cell 3: 259–270.
Fath KR, Trimbur GM, Burgess DR . (1997). J Cell Biol 139: 1169–1181.
Guilford P, Hopkins J, Harraway J, McLeod M, McLeod N, Harawira P et al. (1998). Nature 392: 402–405.
Hadzija MP, Radosevic S, Kovacevic D, Lukac J, Hadzija M, Spaventi R et al. (2004). Mutat Res 548: 61–73.
Kim JB, Islam S, Kim YJ, Prudoff RS, Sass KM, Wheelock MJ et al. (2000). J Cell Biol 151: 1193–1206.
Kretzschmar M, Doody J, Timokhina I, Massague J . (1999). Genes Dev 13: 804–816.
Li QL, Ito K, Sakakura C, Fukamachi H, Inoue K, Chi XZ et al. (2002). Cell 109: 113–124.
Li W, Qiao W, Chen L, Xu X, Yang X, Li D et al. (2003). Development 130: 6143–6153.
Mishra L, Cai T, Levine A, Weng D, Mezey E, Mishra B et al. (1998). Int J Dev Biol 42: 221–224.
Mishra L, Cai T, Yu P, Monga SP, Mishra B . (1999). Oncogene 18: 353–364.
Miyaki M, Iijima T, Kimura J, Yasuno M, Mori T, Hayashi Y et al. (1999). Cancer Res 59: 4506–4509.
Muller N, Reinacher-Schick A, Baldus S, van Hengel J, Berx G, Baar A et al. (2002). Oncogene 21: 6049–6058.
Nelson WJ, Hammerton RW . (1989). J Cell Biol 108: 893–902.
Nelson WJ, Nusse R . (2004). Science 303: 1483–1487.
Peinado H, Quintanilla M, Cano A . (2003). J Biol Chem 278: 21113–21123.
Piepenhagen PA, Nelson WJ . (1998). Mol Biol Cell 9: 3161–3177.
Pinder JC, Baines AJ . (2000). Nature 406: 253–254.
Roberts AB, Sporn MB . (1990). The Transforming Growth Factor-βs. Springer-Verlag: Heildelberg.
Rosivatz E, Becker I, Specht K, Fricke E, Luber B, Busch R et al. (2002). Am J Pathol 161: 1881–1891.
Scartozzi M, Galizia E, Freddari F, Berardi R, Cellerino R, Cascinu S . (2004). Cancer Treat Rev 30: 451–459.
Shi Y, Massague J . (2003). Cell 113: 685–700.
Siegel PM, Massague J . (2003). Nat Rev Cancer 3: 807–821.
Sormunen RT, Leong AS, Vaaraniemi JP, Fernando SS, Eskelinen SM . (1999). J Pathol 187: 416–423.
Tang B, Vu M, Booker T, Santner SJ, Miller FR, Anver MR et al. (2003). J Clin Invest 112: 1116–1124.
Tang Y, Katuri V, Dillner A, Mishra B, Deng CX, Mishra L . (2003). Science 299: 574–577.
Tang Y, Katuri V, Srinivasan R, Fogt F, Redman R, Anand G et al. (2005). Cancer Res 65: 4228–4237.
Tsukazaki T, Chiang TA, Davison AF, Attisano L, Wrana JL . (1998). Cell 95: 779–791.
Waite KA, Eng C . (2003). Nat Rev Genet 4: 763–773.
Xu X, Brodie SG, Yang X, Im YH, Parks WT, Chen L et al. (2000). Oncogene 19: 1868–1874.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. (2004). Cell 117: 927–939.
Acknowledgements
We thank Dr M Zasloff, Dr R Schlegel and Dr S Baker for critical review of the manuscript, and Dr Eugene A Volpe and Anan H Said for their technical support.
This work was supported by NIH Grants RO1 CA106614A (LM), RO1 DK56111 (LM), RO1 DK58637 (BM), VA Merit Award (LM) and R Robert and Sally D Funderburg Research Scholar (LM).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Katuri, V., Tang, Y., Li, C. et al. Critical interactions between TGF-β signaling/ELF, and E-cadherin/β-catenin mediated tumor suppression. Oncogene 25, 1871–1886 (2006). https://doi.org/10.1038/sj.onc.1209211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209211
- Springer Nature Limited
Keywords
This article is cited by
-
Genomic landscape of gastric cancer: molecular classification and potential targets
Science China Life Sciences (2017)
-
Stromal upregulation of lateral epithelial adhesions: Gene expression analysis of signalling pathways in prostate epithelium
Journal of Biomedical Science (2011)
-
Runx3 expression in gastrointestinal tract epithelium: resolving the controversy
Oncogene (2009)