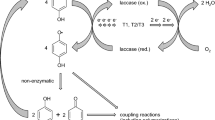
We are studying the enzymatic modification of polycyclic aromatic hydrocarbons (PAHs) by the laccase from Coriolopsis gallica UAMH 8260. The enzyme was produced during growth in a stirred tank reactor to 15 units ml−1, among the highest levels described for a wild-type fungus; the enzyme was the major protein produced under these conditions. After purification, it exhibited characteristics typical of a white rot fungal laccase. Fifteen azo and phenolic compounds at 1 mM concentration were tested as mediators in the laccase oxidation of anthracene. Higher anthracene oxidation was obtained with the mediator combination of ABTS and HBT, showing a correlation between the oxidation rate and the mediator concentration. Reactions with substituted phenols and anilines, conventional laccase substrates, and PAHs were compared using the native laccase and enzyme preparations chemically modified with 5000 MW-poly(ethylene glycol). Chemically modified laccase oxidized a similar range of substituted phenols as the native enzyme but with a higher catalytic efficiency. The k cat increase by the chemical modification may be as great as 1300 times for syringaldazine oxidation. No effect was found of chemical modification on mediated PAH oxidation. Both unmodified and PEG-modified laccases increased PAH oxidation up to 1000 times in the presence of radical mediators. Thus, a change of the protein surface improves the mediator oxidation efficiency, but does not affect non-enzymatic PAH oxidation by oxidized mediators.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 10 December 2001/ Accepted in revised form 20 July 2002
Rights and permissions
About this article
Cite this article
Vandertol-Vanier, H., Vazquez-Duhalt, R., Tinoco, R. et al. Enhanced activity by poly(ethylene glycol) modification of Coriolopsis gallica laccase. J Ind Microbiol Biotech 29, 214–220 (2002). https://doi.org/10.1038/sj.jim.7000308
Issue Date:
DOI: https://doi.org/10.1038/sj.jim.7000308