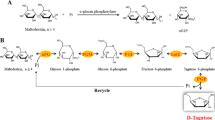
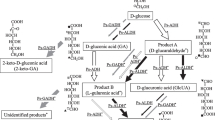
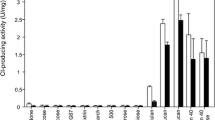
For the production of α-D-glucose-1-phosphate (G-1-P), α-1,4-D-glucan phosphorylase from Thermus caldophilus GK24 was partially purified to a specific activity of 13 U mg−1 and an enzyme recovery of 15%. The amount of G-1-P reached maximum (18%) when soluble starch was used as substrate, and the smallest substrate for G-1-P formation was maltotriose. The structure of purified G-1-P was confirmed by comparison to 13C-NMR data for an authentic sample. In addition to G-1-P, glucose-6-phosphate (12%) was simultaneously produced when 10 mM maltoheptaose was used as substrate. Journal of Industrial Microbiology & Biotechnology (2000) 24, 89–93.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 12 May 1999/ Accepted in revised form 29 August 1999
Rights and permissions
About this article
Cite this article
Shin, HJ., Shin, Y. & Lee, DS. Formation of α-D-glucose-1-phosphate by thermophilic α-1,4-D-glucan phosphorylase. J Ind Microbiol Biotech 24, 89–93 (2000). https://doi.org/10.1038/sj.jim.2900757
Issue Date:
DOI: https://doi.org/10.1038/sj.jim.2900757