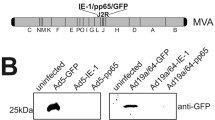
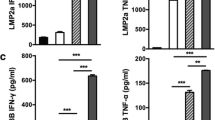
Abstract
Dendritic cell (DC) therapies using DC presenting tumor antigen/s can induce CD8+ CTL that mediate tumor eradication, nonetheless many patients remain unresponsive. Thus, cytokine gene vectors applied to DC may amplify these responses. Herein, we examined the responses that monocyte-derived DC (at different maturational stages) make when infected with a vaccinia virus-interleukin-2 (VV-IL-2) vector in vitro. VV-IL-2-infected DC secreted significant levels of bioactive IL-2 and maintained their antigen presentation function. However, we show that DC are exquisitely sensitive to their local antigenic microenvironment, and that responses generated by one antigen can be altered by another. VV-IL-2 infection of immature DC led to DC activation (upregulation of CD80, CD86 and class II surface molecules) when the virus was propagated through xenogeneic, but not syngeneic, mammalian cells; these DC secreted IL-10 and tumor necrosis factor-α (TNF-α), but not IL-12. In contrast, after VV-IL-2 infection (regardless of their mammalian cellular context), IFNγ/LPS-matured DC inevitably downregulated their antigen presenting machinery. In conclusion, immunostimulatory DC can be generated by VV-IL-2, but this depends upon (i) infecting immature DC only, (ii) the mammalian cells through which the virus is prepared and (iii) individual donors; hence donors must be screened to assess their specific responses.








Similar content being viewed by others
References
Finke J, Ferrone S, Frey A, et al. Where have all the T-cells gone? Mechanisms of immune evasion by tumors. Immunol Today. 1999;20:158–160.
Marzo AL, Lake RA, Lo D, et al. Tumor antigens are constitutively presented in the draining lymph nodes. J Immunol. 1999;162:5838–5845.
Nelson DJ, Mukherjee S, Bundell C, et al. Tumor progression despite efficient tumor antigen cross-presentation and effective “arming” of tumor antigen-specific CTL. J Immunol. 2001;166:5557–5566.
Mukherjee S, Haenel T, Himbeck R, et al. Replication-restricted vaccinia as a cytokine gene therapy vector in cancer: persistent transgene expression despite antibody generation. Cancer Gene Ther. 2000;7:663–670.
Bernsen MR, Tang JW, Everse LA, et al. Interleukin 2 (IL-2) therapy: potential advantages of locoregional versus systemic administration. Cancer Treat Rev. 1999; 25:73–82.
Pantuck AJ, Belldegrun AS . Phase I clinical trial of interleukin 2 (IL-2) gene therapy for prostate cancer. Curr Urol Rep. 2001;2:33.
Tartour E, Mehtali M, Sastre-Garau X, et al. Phase I clinical trial with IL-2-transfected xenogeneic cells administered in subcutaneous metastatic tumours: clinical and immunological findings. Br J Cancer. 2000;83:1454–1461.
Dorval T, Mathiot C, Chosidow O, et al. IL-2 phase II trial in metastatic melanoma: analysis of clinical and immunological parameters. Biotechnol Ther. 1992;3:63–79.
Castagneto B, Zai S, Mutti L, et al. Palliative and therapeutic activity of IL-2 immunotherapy in unresectable malignant pleural mesothelioma with pleural effusion: results of a phase II study on 31 consecutive patients. Lung Cancer. 2001;31:303–310.
Krastev Z, Koltchakov V, Vladov N, et al. A mesothelioma that is sensitive to locally applied IL-2. Cancer Immunol Immunother. 2001;50:226–227.
Steinman RM, Inaba K, Turley S, et al. Antigen capture, processing, and presentation by dendritic cells: recent cell biological studies. Hum Immunol. 1999;60:562–567.
Huang AY, Golumbek P, Ahmadzadeh M, et al. Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens. Science. 1994;264:961–965.
Bennett SR, Carbone FR, Karamalis F, et al. Induction of a CD8+ cytotoxic T lymphocyte response by cross-priming requires cognate CD4+ T-cell help. J Exp Med. 1997;186:65–70.
Marzo AL, Lake RA, Robinson BWS, et al. T-cell receptor transgenic analysis of tumor-specific CD8 and CD4 responses in the eradication of solid tumors. Cancer Res. 1999;59:1071–1079.
Steinman RM, Dhodapkar M . Active immunization against cancer with dendritic cells: the near future. Int J Cancer. 2001;94:459–473.
Nouri-Shirazi M, Banchereau J, Fay J, et al. Dendritic cell based tumor vaccines. Immunol Lett. 2000;74:5–10.
Celluzzi CM, Mayordomo JI, Storkus WJ, et al. Peptide-pulsed dendritic cells induce antigen-specific CTL-mediated protective tumor immunity. J Exp Med. 1996;183:283–287.
Nestle FO, Alijagic S, Gilliet M, et al. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med. 1998;4:328–332.
Zhou Y, Bosch ML, Salgaller ML . Current methods for loading dendritic cells with tumor antigen for the induction of antitumor immunity. J Immunother. 2002;25:289–303.
Tirapu I, Rodriguez-Calvillo M, Qian C, et al. Cytokine gene transfer into dendritic cells for cancer treatment. Curr Gene Ther. 2002;2:79–89.
Berard F, Blanco P, Davoust J, et al. Cross-priming of naive CD8 T-cells against melanoma antigens using dendritic cells loaded with killed allogeneic melanoma cells. J Exp Med. 2000;192:1535–1544.
Dhodapkar MV, Steinman RM, Krasovsky J, et al. Antigen-specific inhibition of effector T-cell function in humans after injection of immature dendritic cells. J Exp Med. 2001;193:233–238.
Drillien R, Spehner D, Bohbot A, et al. Vaccinia virus-related events and phenotypic changes after infection of dendritic cells derived from human monocytes. Virology 2000;268:471–481.
Jenne L, Hauser C, Arrighi JF, et al. Poxvirus as a vector to transduce human dendritic cells for immunotherapy: abortive infection but reduced APC function. Gene Therapy. 2000;7:1575–1583.
Engelmayer J, Larsson M, Subklewe M, et al. Vaccinia virus inhibits the maturation of human dendritic cells: a novel mechanism of immune evasion. J Immunol. 1999;163:6762–6768.
Drexler I, Antunes E, Schmitz M, et al. Modified vaccinia virus ankara for delivery of human tyrosinase as melanoma-associated antigen: induction of tyrosinase- and melanoma-specific human leukocyte antigen a*0201-restricted cytotoxic T-cells in vitro and in vivo. Cancer Res. 1999;59:4955–4963.
Kim CJ, Cormier J, Roden M, et al. Use of recombinant poxviruses to stimulate anti-melanoma T-cell reactivity. Ann Surg Oncol. 1998;5:64–76.
Yang S, Kittlesen D, Slingluff Jr. CL, et al. Dendritic cells infected with a vaccinia vector carrying the human gp100 gene simultaneously present multiple specificities and elicit high-affinity T cells reactive to multiple epitopes and restricted by HLA-α2 and -α3. J Immunol. 2000;164:4204–4211.
Kedl RM, Rees WA, Hildeman DA, et al. T-cells compete for access to antigen-bearing antigen-presenting cells. J Exp Med. 2000;192:1105–1113.
Norbury CC, Malide D, Gibbs JS, et al. Visualizing priming of virus-specific CD8+ T-cells by infected dendritic cells in vivo. Nat Immunol. 2002;3:265.
Nossal GJ . Tolerance and ways to break it. Ann NY Acad Sci. 1993;690:34–41.
Tham EL, Shrikant P, Mescher MF . Activation-induced nonresponsiveness: a Th-dependent regulatory checkpoint in the CTL response. J Immunol. 2002;168:1190–1197.
Romani N, Gruner S, Brang D, et al. Proliferating dendritic cell progenitors in human blood. J Exp Med. 1994;180:83–93.
Ramshaw IA, Andrew ME, Phillips SM, et al. Recovery of immunodeficient mice from a vaccinia virus/IL-2 recombinant infection. Nature. 1987;329:545–546.
Warren HS, Kinnear BF, Skipsey LJ . Human natural killer (NK) cells: requirements for cell proliferation and expansion of phenotypically novel subpopulations. Immunol Cell Biol. 1993;71:87–97.
Brossart P, Goldrath AW, Butz EA, et al. Virus-mediated delivery of antigenic epitopes into dendritic cells as a means to induce CTL. J Immunol. 1997;158:3270–3276.
Di Nicola M, Siena S, Bregni M, et al. Gene transfer into human dendritic antigen-presenting cells by vaccinia virus and adenovirus vectors. Cancer Gene Ther. 1998; 5:350–356.
Kim CJ, Prevette T, Cormier J, et al. Dendritic cells infected with poxviruses encoding MART-1/melan A sensitize T lymphocytes in vitro. J Immunother. 1997;20:276–286.
Bronte V, Carroll MW, Goletz TJ, et al. Antigen expression by dendritic cells correlates with the therapeutic effectiveness of a model recombinant poxvirus tumor vaccine. Proc Natl Acad Sci USA. 1997;94:3183–3188.
Akiyama Y, Watanabe M, Maruyama K, et al. Enhancement of antitumor immunity against B16 melanoma tumor using genetically modified dendritic cells to produce cytokines. Gene Therapy. 2000;7:2113–2121.
Shimizu K, Fields RC, Giedlin M, et al. Systemic administration of interleukin 2 enhances the therapeutic efficacy of dendritic cell-based tumor vaccines. Proc Natl Acad Sci USA. 1999;96:2268–2273.
Shimizu K, Fields RC, Redman BG, et al. Potentiation of immunologic responsiveness to dendritic cell-based tumor vaccines by recombinant interleukin-2. Cancer J Sci Am. 2000;6(Suppl 1):S67–S75.
Eggert AO, Becker JC, Ammon M, et al. Specific peptide-mediated immunity against established melanoma tumors with dendritic cells requires IL-2 and fetal calf serum-free cell culture. Eur J Immunol. 2002;32:122–127.
Faulkner L, Buchan G, Lockhart E, et al. IL-2 linked to a peptide from influenza hemagglutinin enhances T-cell activation by affecting the antigen-presentation function of bone marrow-derived dendritic cells. Int Immunol. 2001;13:713–721.
Granucci F, Vizzardelli C, Pavelka N, et al. Inducible IL-2 production by dendritic cells revealed by global gene expression analysis. Nat Immunol. 2001;2:882–888.
Sauter B, Albert ML, Francisco L, et al. Consequences of cell death: exposure to necrotic tumor cells, but not primary tissue cells or apoptotic cells, induces the maturation of immunostimulatory dendritic cells. J Exp Med. 2000;191:423–434.
Ferlazzo G, Semino C, Spaggiari GM, et al. Dendritic cells efficiently cross-prime HLA class I-restricted cytolytic T lymphocytes when pulsed with both apoptotic and necrotic cells but not with soluble cell-derived lysates. Int Immunol. 2000;12:1741–1747.
Kotera Y, Shimizu K, Mule JJ . Comparative analysis of necrotic and apoptotic tumor cells as a source of antigen(s) in dendritic cell-based immunization. Cancer Res. 2001;61:8105–8109.
Fernando GJP, Stewart TJ, Tindle RW, et al. Th2-type CD4+ cells neither enhance nor suppress antitumor CTL activity in a mouse tumor model. J Immunol. 1998;161:2421–2427.
Nishimura T, Iwakabe K, Sekimoto M, et al. Distinct role of antigen-specific T helper type 1 (Th1) and Th2 cells in tumor eradication in vivo. J Exp Med. 1999;190:617–627.
Kuniyoshi JS, Kuniyoshi CJ, Lim AM, et al. Dendritic cell secretion of IL-15 is induced by recombinant huCD40Lt and augments the stimulation of antigen-specific cytolytic T-cells. Cell Immunol. 1999;193:48–58.
Acknowledgements
This work was supported by grants from the NH&MRC, SGIC and ALF. We thank Jane Allan for her advice and supply of the control vaccinia virus construct, as well as Dr Matthew Wikstrom and the Lotteries Commission WA Flow Cytometry Unit for assistance with FACs analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mukherjee, S., Upham, J., Ramshaw, I. et al. Dendritic cells infected with a vaccinia virus interleukin-2 vector secrete high levels of IL-2 and can become efficient antigen presenting cells that secrete high levels of the immunostimulatory cytokine IL-12. Cancer Gene Ther 10, 591–602 (2003). https://doi.org/10.1038/sj.cgt.7700604
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700604
- Springer Nature America, Inc.
Keywords
This article is cited by
-
Evaluating vaccinia virus cytokine co‐expression in TLR GKO mice
Immunology & Cell Biology (2011)
-
Cytokine-armed vaccinia virus infects the mesothelioma tumor microenvironment to overcome immune tolerance and mediate tumor resolution
Cancer Gene Therapy (2010)
-
Immune properties of recombinant vaccinia virus encoding CD154 (CD40L) are determined by expression of virally encoded CD40L and the presence of CD40L protein in viral particles
Cancer Gene Therapy (2004)