Abstract
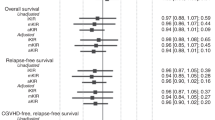
The reactivity of natural killer cells and some T-cell populations is regulated by killer immunoglobulin-like receptors (KIR) interactions with target cell HLA class I molecules. Such interactions have been suggested to influence outcomes after allogeneic hematopoietic stem cell transplantation, particularly for myeloid malignancies and with T-cell depletion. Donor KIR genotypes and recipient HLA KIR ligands were analyzed in 60 AML patients receiving T-cell replete, HLA-matched-related donor allogeneic bone marrow transplants. Patients were categorized according to their HLA inhibitory KIR ligand groups by determining whether or not they expressed: HLA-A3 or -A11; HLA-Bw4 and HLA-Cw groups (homozygous C1, homozygous C2 or heterozygous C1/C2). Heterozygous C1/C2 patients had significantly worse survival than those homozygous for C1 or C2 (5.8 vs 43.5 months, respectively, P=0.018) and the C1/C2 group had a higher relapse rate (47 vs 31%, respectively, P=0.048). Multivariate analysis found C1/C2 status to be an independent predictor for mortality (P=0.007, HR 2.54, confidence interval 1.29–5.00). C1/C2 heterozygosity was also associated with a delayed time to platelet engraftment, particularly for those with concurrent HLA-Bw4 expression (P=0.003). Since C1/C2 heterozygotes have a greater opportunity to engage inhibitory KIRs than do C1 or C2 homozygotes, they may more effectively inhibit KIR-positive NK- and T-cell populations involved in graft vs leukemia responses.



Similar content being viewed by others
References
Zittoun RA, Mandelli F, Willemze R, de Witte T, Labar B, Resegotti L et al. Autologous or allogeneic bone marrow transplantation compared with intensive chemotherapy in acute myelogenous leukemia. N Engl J Med 1995; 332: 217–223.
Kernan NA, Bartsch G, Ash RC, Beatty PG, Champlin R, Filipovich A et al. Analysis of 462 transplantations from unrelated donors facilitated by The National Marrow Donor Program. N Engl J Med 1993; 328: 593–602.
Molldrem JJ, Lee PP, Wang C, Felio K, Kantarjian HM, Champlin RE et al. Evidence that specific T lymphocytes may participate in the elimination of chronic myelogenous leukemia. Nat Med 2000; 6: 1018–1023.
Hercend T, Takvorian T, Nowill A, Tantravahi R, Moingeon P, Anderson KC et al. Characterization of natural killer cells with antileukemia activity following allogeneic bone marrow transplantation. Blood 1986; 67: 722–728.
Ruggeri L, Capanni M, Urbani E, Perruccio K, Shlomchik WD, Tosti A et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science 2002; 295: 2097–2100.
Farag S, Fehniger T, Ruggeri L, Velardi A, Caligiuri MA . Natural killer cell receptors: new biology and insights into the graft-versus-leukemia effect. Blood 2002; 100: 1935–1947.
Lanier LL, Corliss BC, Wu J, Leong C, Phillips JH . Immunoreceptor DAP12 bearing a tyrosine-based activation motif is involved in activating NK cells. Nature 1998; 391: 703–707.
Unkeless JC, Jin J . Inhibitory receptors, ITIM sequences and phosphatases. Curr Opin Immunol 1997; 9: 338–343.
Vales-Gomez M, Reyburn HT, Mandelboim M, Strominger JL . Kinetics of interaction of HLA-C ligands with natural killer cell inhibitory receptors. Immunity 1998; 9: 337–344.
Boyington JC, Sun PD . A structural perspective on MHC class I recognition by killer immunoglobulin-like receptors. Mol Immunol 2002; 38: 1007–1021.
Gumperz JE, Litwin V, Phillips JH, Lanier LL, Parham P . The Bw4 public epitope of HLA-B molecules confers reactivity with natural killer cell clones that express NKB1, a putative HLA receptor. J Exp Med 1995; 181: 1133–1144.
Ruggeri L, Capanni M, Casucci M, Volpi I, Tosti A, Perruccio K et al. Role of natural killer cell alloreactivity in HLA-mismatched hematopoietic stem cell transplantation. Blood 1999; 94: 333–339.
Giebel S, Locatelli FW, Lamparelli T, Velardi A, Davies S, Frumento G et al. Survival advantage with KIR ligand incompatibility in hematopoietic stem cell transplantation from unrelated donors. Blood 2003; 102: 814–819.
Cook MA, Milligan DW, Fegan CD, Darbyshire PJ, Mahendra P, Craddock CF et al. The impact of donor KIR and patient HLA-C genotypes on outcome following HLA-identical sibling hematopoietic stem cell transplantation for myeloid leukemia. Blood 2004; 103: 1521–1526.
Hsu KC, Keever-Taylor CA, Wilton A, Pinto C, Heller G, Arkun K et al. Improved outcome in HLA-identical sibling hematopoietic stem cell transplantation for acute myelogenous leukemia (AML) predicted by KIR and HLA genotypes. Blood 2005; 105: 4878–4884.
Paul P, Apgar J, Ball EJ . HLA-DRB1* intron-primed sequencing for haploid genotyping. Clin Chem 2003; 49: 692–694.
Paul P, Thomas D, Kawczak P, Good D, Cook DJ, Ball EJ . Resolution of cis-trans ambiguities between HLA-DRB1 alleles using single-strand conformation polymorphisms and sequencing. Tissue Antigens 2001; 57: 300–307.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Vogelsang GB . How I treat chronic graft-versus-host disease. Blood 2001; 97: 1196–1201.
Leung W, Iyengar R, Turner V, Lang P, Bader P, Conn P et al. Determinants of antileukemia effects of allogeneic NK cells. J Immunol 2004; 172: 644–650.
Collins Jr RH, Shpilberg O, Drobyski WR, Porter DL, Giralt S, Champlin R et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Kolb HJ, Mittermuller J, Clemm C, Holler E, Ledderose G, Brehm G et al. Donor leukocyte transfusions for treatment of recurrent chronic myelogenous leukemia in marrow transplant patients. Blood 1990; 76: 2462–2465.
Porter DL, Collins Jr RH, Hardy C, Kernan NA, Drobyski WR, Giralt S et al. Treatment of relapsed leukemia after unrelated donor marrow transplantation with unrelated donor leukocyte infusions. Blood 2000; 95: 1214–1221.
Levine JE, Braun T, Penza SL, Beatty P, Cornetta K, Martino R et al. Prospective trial of chemotherapy and donor leukocyte infusions for relapse of advanced myeloid malignancies after allogeneic stem-cell transplantation. J Clin Oncol 2002; 20: 405–412.
Verheyden S, Schots R, Duquet W, Demanet C . A defined donor activating natural killer cell receptor genotype protects against leukemic relapse after HLA-identical hematopoietic stem cell transplantation. Leukemia 2005; 19: 1446–1451.
Xu H, Exner B, Cramer D, Tanner MK, Mueller YM, Ildstad ST . CD8+, αβ-TCR+, and γδ-TCR+ cells in the recipient hematopoietic environment mediate resistance to engraftment of allogeneic donor bone marrow. J Immunol 2002; 168: 1636–1643.
Tiberghien P, Longo D, Wine J, Alvord WG, Reynolds CW . Anti-asialo GM1 antiserum treatment of lethally irradiated recipients before bone marrow transplantation: Evidence that recipient natural killer depletion enhances survival, engraftment, and hematopoietic recovery. Blood 1990; 76: 1419–1430.
Vivier E, Anfossi N . Inhibitory NK-cell receptors on T cells: witness of the past, actors of the future. Nat Rev Immunol 2004; 4: 190–198.
Ferrini S, Cambiaggi A, Meazza R, Sforzini S, Marciano S, Mingari MC et al. T cell clones expressing the natural killer cell-related p58 receptor molecule display heterogeneity in phenotypic properties and p58 functions. Eur J Immunol 1994; 24: 2294–2298.
Leung W, Iyengar R, Triplett B, Turner V, Behm FG, Holladay MS et al. Comparison of killer Ig-like receptor genotyping and phenotyping for selection of allogeneic blood stem cell donors. J Immunol 2005; 174: 6540–6545.
Gardiner CM, Guethlein LA, Shilling HG, Pando M, Carr WH, Rajalingam R et al. Different NK cell surface phenotypes defined by the DX9 antibody are due to KIR3DL1 gene polymorphism. J Immunol 2001; 166: 2992–3001.
Santourlidis S, Trompeter HI, Weinhold S, Eisermann B, Meyer KL, Wernet P et al. Crucial role of DNA methylation in determination of clonally distributed killer cell Ig-like receptor expression patterns in NK cells. J Immunol 2002; 169: 4253–4261.
Shilling HG, Guethlein LA, Cheng CM, Gardiner CM, Rodriguez R, Tyan D et al. Allelic polymorphism synergizes with variable gene content to individualize human KIR genotype. J Immunol 2002; 168: 2307–2315.
Goodridge JP, Witt CS, Christiansen FT, Warren HS . KIR2DL4 (CD158d) genotype influences expression and function in NK cells. J Immunol 2003; 171: 1768–1774.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sobecks, R., Ball, E., Maciejewski, J. et al. Survival of AML patients receiving HLA-matched sibling donor allogeneic bone marrow transplantation correlates with HLA-Cw ligand groups for killer immunoglobulin-like receptors. Bone Marrow Transplant 39, 417–424 (2007). https://doi.org/10.1038/sj.bmt.1705609
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705609
- Springer Nature Limited
Keywords
This article is cited by
-
Leukemia relapse via genetic immune escape after allogeneic hematopoietic cell transplantation
Nature Communications (2023)
-
Association of killer cell immunoglobulin-like receptors and their cognate HLA class I ligands with susceptibility to acute myeloid leukemia in Iranian patients
Scientific Reports (2023)
-
Prognostic role of KIR genes and HLA-C after hematopoietic stem cell transplantation in a patient cohort with acute myeloid leukemia from a consanguineous community
Bone Marrow Transplantation (2018)
-
Donor-recipient killer immunoglobulin like receptor (KIR) genotype matching has a protective effect on chronic graft versus host disease and relapse incidence following HLA-identical sibling hematopoietic stem cell transplantation
Annals of Hematology (2018)
-
Bone marrow may be the preferable graft source in recipients homozygous for HLA-C group 2 ligands for inhibitory killer Ig-like receptors
Bone Marrow Transplantation (2012)