Abstract
Capsaicin-sensitive afferents have complex regulatory functions in the joints orchestrated via neuropeptides. This study aimed to determine their role in the collagen-antibody induced rheumatoid arthritis model. Capsaicin-sensitive nerves were defunctionalized by the capsaicin receptor agonist resiniferatoxin in C57Bl/6 mice. Arthritis was induced by the ArithroMab antibody cocktail and adjuvant. Arthritis was monitored by measuring body weight, joint edema by plethysmometry, arthritis severity by clinical scoring, mechanonociceptive threshold by plantar esthesiometry, thermonociceptive threshold by hot plate, cold tolerance by paw withdrawal latency from 0 °C water. Grasping ability was determined by the wire-grid grip test. Bone structure was evaluated by in vivo micro-CT and histology. Arthritic animals developed a modest joint edema, mechanical and cold hyperalgesia, weight loss, and a diminished grasping function, while thermal hyperalgesia is absent in the model. Desensitised mice displayed reduced arthritis severity, edema, and mechanical hyperalgesia, however, cold hyperalgesia was significantly greater in this group. Arthritic controls displayed a transient decrease of bone volume and an increased porosity, while bone density and trabecularity increased in desensitised mice. The activation of capsaicin-sensitive afferents increases joint inflammation and mechanical hyperalgesia, but decreases cold allodynia. It also affects inflammatory bone structural changes by promoting bone resorption.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a chronic autouimmune inflammatory disease of the joints, constituting a global burden. RA is a public health problem due to its high prevalence, affecting 0.1–1% of the world population with significant regional differences1. In the last two decades the introduction of novel drugs, primarily biologics, improved the treatment of the immune-component of RA2. However, the analgesic therapeutic regime underwent only minimal change. The evidence of the complex interplay of neural and immune components in the development of RA has been proven by numerous clinical and experimental studies, but the mechanism of these interactions is still incompletely understood. Early experimental results3, and occassional clinical observations4 showed that local denervation is protective against joint inflammation. This highlighted the critical importance of innervation in the induction of arthritis.
Capsaicin-sensitive sensory afferents densely innervate the articular capsule and the synovium, hence their involvement in arthritic pain has been proposed relatively early on5. A hallmark feature of these nerve terminals is their dual nature: as classic afferents they participate in pain signaling towards the central nervous system and they also modulate the inflammatory reaction by acting as efferents by the release of sensory neuropeptides. A key feature of these nerve terminals is the expression of the Transient Receptor Potential Vanilloid 1 (TRPV1) capsaicin receptor, which is a non-selective cation channel. It is activated and sensitized not only by a plethora of exogenous irritants, but also by endogenous inflammatory mediators also upregulated during RA6. This includes noxious heat, protons, prostanoids, bradkyinin, TNF-α, and free radicals, but also phytochemicals like capsaicin, the pungent ingredient of chilli pepper, and its even more potent analogue resiniferatoxin (RTX)5,7. TRPV1 receptor activation results in the release of peptide mediators, some of which are proinflammatory, such as tachykinins, calcitonin gene-releated peptide. These in turn induce local hyperemia and the recruitment of inflammatory cells, which is called neurogenic inflammation8. However, simultaneously, anti-inflammatory mediators, like somatostatin are also released. The clinical importance of such peptide mediators is well established, and numerous studies have shown that RA and also osteoarthritis patients display altered levels of sensory neuropeptides in the synovial fluid and/or serum. Furthermore, recent studies also identified single nucleotide polymorphisms (SNP) responsible for this increased expression9,10,11,12,13,14,15. Large scale human studies have also discovered that certain SNPs of neuropeptide receptors increase the likelihood of symptomatic arthritis16. Sporadic clinical evidence also shows that anti-inflammatory neuropeptide mediators are able to delay the onset of RA, demonstrated by cases of rapidly developing RA immediately after the successful treatment of somatostatin-producing endocrine tumors17. Prior work has provided compelling, albeit in several cases conflicting evidence of the importance of both capsaicin-sensitive afferents, TRPV1 ion channels, and the divergent pro- and antiinflammatory role of their mediators using diverse models of joint inflammation mimicking different aspects of RA18,19,20,21.
The collagen-induced arthritis (CIA) is widely known as a gold standard murine model of RA, but due to its variable incidence, disease severity, and limited susceptibility of standard laboratory mouse strains using this model is often not feasible, and comparisons remain a challenge. The CIA model is characterized by a high titer of anti-type II collagen IgG autoantibodies, which is also a hallmark feature of RA. Passive transfer of the antibodies of CIA mice into healthy recipients can induce a more rapid, albeit transient and mild arthritis with 100% penetration in many mouse strains not readily susceptible to CIA, hence it is termed collagen-antibody-induced arthritis (CAIA). Unlike the original model, CAIA is mediated mainly via macrophages and neutrophils, without any involvement of the adaptive immunity22,23.
In the present study we aimed to characterize the role of capsaicin-sensitive afferents in the CAIA model of RA, which offers an unique way to interrogate the involvement of these nerve endings in anti-type II collagen antibody-driven joint inflammation. Furthermore, we also evaluated the performance of the model in the C57Bl/6 strain, which is widely used for interrogating mechanisms of pain and inflammation, but is not readily susceptible to other collagen-induced arthritis models.
Results
Decreased edema and arthritis severity in desensitized arthritic mice
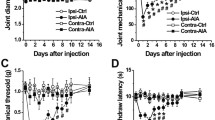
Hindpaw edema and clinical arthritis severity scores remained unchanged in both groups until day 3, when the lipopolysaccharide booster injection was administered. In non-desensitized mice a modest, but significant 20% increase of hindlimb volume was observed, which peaked on day 6 (Fig. 1A). Arthritis severity scores reached their maximum on day 9 having the maximal values of 3–4 in non-pretreated mice, indicating moderate, but significant arthritic changes (Fig. 1B). In desensitized mice no significant edema formation, or clinical severity score increase could be observed.
Diminished mechanical hyperalgesia, but increased cold allodynia in desensitized mice
The baseline touch sensitivity threshold of the non-desensitized and RTX-desensitized groups were not significantly different. In non-desensitized mice a 45% of mechanonociceptive threshold drop was observed, peaking on day 8. In RTX-pretreated mice inflammatory mechanical hyperalgesia peaked at 30%, and remained significantly lower thereafter (Fig. 2A). Paw withdrawal latency from ice-cold water gradually decreased during the study in both groups, being significantly lower in desensitized animals on days 4 and 11 showing cold hyperalgesia in the model (Fig. 2B). Thermal hyperalgesia did not develop in the model in either group (Fig. 2C).
Changes of (A) mechanical hyperalgesia, (B) cold allodynia, (C) thermal hyperalgesia. Data points represent mean ± SEM. (n = 5–6/group, *p < 0.05, **p < 0.01, ***p < 0.001 vs. initial self-control, #p < 0.05, ##p < 0.01, ###p < 0.001 vs. non-pretreated, two-way repeated measures ANOVA followed by Sidak’s post-test).
Absent difference in weight loss and grasping ability in arthritic mice after sensory desensitization
A 20% weight loss developed rapidly upon arthritis induction in both desensitized and non-pretreated mice, peaking on days 6–7 of the study (Fig. 3A). Wire grid performance decreased steadily during the first half of study and by day 5 about 50% of the mice in both groups became unable to maintain their position on the wire mesh grid for the test period, however no significant difference could be observed (Fig. 3B). The grasping ability returned to normal in both groups after day 12 (Supplementary Fig. S1).
Absent bone resorption, increased bone volume, but diminished bone pore formation in desensitized arthritic animals
No visually apparent bone erosions or osteophytes developed around the ankle joints during the study, reflecting the transient nature of the model (Fig. 4A). RTX-desensitization alone did not influence bone mass. However a significant 15% increase of the bone volume/total volume ratio could be observed in desensitized mice by day 7, while non-pretreated animals displayed a 12% decrease (Fig. 4B). The percentage of open pores increased significantly in non-pretreated, but not in desensitized mice by day 7, but this difference was not maintained for the rest of the study (Fig. 4C). On the other hand, bone trabecular thickness was found to be approximately 20% greater in RTX-pretreated mice, a difference which was consistently maintained throughout the study (Fig. 4D). Trabecular separation increased remarkably by 15% in non-pretreated mice, while decreasing by 10% after desensitization by day 7 (Fig. 4E).
The changes of bone structural parameters throughout the study. (A) representative micro-CT images of the ankle joints. Changes of (B) bone mass, (C) percent volume of open pores, (D) mean thickness, (E) mean separation of bone trabecules. (n = 5–6/group, *p < 0.05, **p < 0.01, ***p < 0.001 vs. initial self control, #p < 0.05, ##p < 0.01, ###p < 0.001 vs. non-pretreated, two-way repeated measures ANOVA followed by Sidak’s post-test).
Retained integrity of the articular cartilage
Examination of the histological slides made from the ankle joints obtained on day 21 demonstrated mild synovial proliferation in the tibiotarsal joints of both arthritic groups, but the articular cartilage remained intact (Fig. 5A,B).
Discussion
Our experiment provides the first evidence for the involvement of capsaicin-sensitive sensory nerves in the CAIA model of autoantibody-induced joint inflammation. Desensitization of these afferents results in decreased edema-formation and clinical arthritis severity, along with diminished mechanical hyperalgesia and bone resorption, but leads to increased cold allodynia.
Since there is no completely relevant disease model of RA that could mimic all mechanisms and phases of the human condition, it is important to compare the results obtained in models of different arthritis-related mechanisms to conclude on the role of the capsaicin-sensitive peptidergic sensory nerves.
In previous studies using the CFA-induced arthritis model and the K/BxN serum-transfer model systemic or topical RTX-pretreatment increased arthritis severity, but diminished mechanical hyperalgesia18,19,24. In the more transient mast cell tryptase-induced knee inflammation model edema was similar, while spontaneous pain was diminished after desensitization21. The decreased mechanical hyperalgesia is in good agreement with these previous results, but in the CAIA-model the observed decreased inflammation severity and edema seem to contradict our earlier results. This likely reflects the different inflammatory pathways involved in these models. Unlike the CAIA model, the K/BxN model mediated by different autoantibodies (anti-glucose-6-phosphate isomerase) eliciting a rapidly occurring vascular leakage dependent on mast cells, neutrophils, and the Fc-gamma receptor III (FcγRIII)22,25. Therefore, the exact inflammatory signaling cascade occurs via distinct pathways, leading to different overall effects of the desensitization of capsaicin-sensitive afferents. The CFA arthritis differs even more, as it involves not only myeloid but T cells as well. Furthermore, it is induced by the administration of exogenous mycobacterial antigens (the disease-inducing antigen being a bacterial heat shock protein cross-reactive with cartilage proteoglycan), thereby not resembling the autoinflammatory nature of RA26. However, similar to our findings, in the carrageenan-induced joint inflammation of the rat intraarticular administration of RTX or capsaicin not only induced a long-lasting analgesia, but also significantly reduced edema formation27,28.
Development of cold allodynia that remains persistent even after the decline of the acute inflammation was previously reported in the CAIA model, similarly to our results29. Furthermore, we observed a moderate, but significant facilitating effect of RTX-pretreatment on this phenomenon being more prominent in the early phase of the experiment. While seemingly perplexing, such diverging effects of systemic RTX-pretreatment on thermal and mechanical allodynia have been observed in previous studies, and were attributed mainly to the damage of myelinated afferent fibers, and consquent abnormal sprouting30. However, the possible confounding effect of behavioral changes due to RTX-pretreatment also cannot be ruled out.
While presence of thermal hyperalgesia in CAIA was shown by others by using the Hargreaves method23, our study using the increasing temperature hot-plate test could not corroborate this, and we did not find any significant difference between desensitized and non-pretreated animals. This is however, in agreement with our earlier results with the K/BxN serum-transfer arthritis, where the noxious heat threshold of the plantar surface of the paw was also unaffected by the inflammation of the joints19.
The moderate bone structural changes observed in our experiment reflect on the self-limiting and transient nature of the CAIA model. Naïve mice demonstrated a slight decrease of bone volume and increased trabecular separation, in good agreement with earlier results showing that CAIA is mainly characterized by trabecular bone loss due to increased osteoclast activity31,32. The increased bone surface porosity to the best of our knowledge has not been reported previously, however it is a logical consequence of increased bone resorption. RTX-pretreated mice display a slight increase of bone mass immediately upon the induction of arthritis, whilst formation of surface bone pores, a hallmark feature of arthritic bone resorption is diminished. Previously in the K/BxN serum-transfer model we have also observed a paradoxical bone neoformation in RTX desensitised animals, and also knockout mouse strains deficient in the neuropeptide pituitary adenylate-cyclase activating polypeptide (PACAP)19,33. The present results thus corroborate that defunctionalization of capsaicin-sensitive afferents leads to altered bone metabolic response during joint inflammation, shifting from bone resorption towards increased bone neoformation.
Histopathological examination of the tibiotarsal joint did not show any major bone and cartilage alterations neither in response to arthritis nor RTX desensitization in our model. These data clearly suggest that micro-CT is more sensitive and sophisticated technique to evaluate arthritis-related microarchitectural bone changes.
In summary, capsaicin sensitive afferents are pivotal factors in the development of autoantibody-induced joint inflammation, but the overall effect of their defunctionalization is greatly dependent on the pathophysiological mechanisms involved in the model. While their importance in facilitating inflammatory mechanical hyperalgesia has been supported by all prior experiments, their deactivation in the CAIA model leads to diminished arthritis severity and an absence of inflammatory bone loss. Thus, the importance of peptidergic sensory afferents in arthritis goes well beyond their already established importance in arthritic pain, representing a common denominator through which inflammatory and structural changes of RA could be influenced. The observed significant difference between different RA models highlights the importance of careful selection, and if possible cross-comparison of the different models. This is crucial for making conclusions about the role of sensory afferents and their mediators in immune-mediated arthritides. This is particularly important, as most models are narrowed down to only on a single signaling pathway, thereby not resembling the heterogenicity of the human disease.
Methods
Ethics
All experimental procedures were carried out according to the 1998/XXVIII Act of the Hungarian Parliament on Animal Protection and Consideration Decree of Scientific Procedures of Animal Experiments (243/1988) and complied with the recommendations of the International Association for the Study of Pain. The studies were approved by the Ethics Committee on Animal Research of University of Pécs according to the Ethical Codex of Animal Experiments and licence was given (licence No.: BA 02/2000-2/2012).
Animals
Experiments were performed on 10–12-week-old male C57Bl/6 mice weighing 25–30 g. The original breeding pairs were purchased from Charles-River Hungary. The animals were bred and kept in the Laboratory Animal House of the Department of Pharmacology and Pharmacotherapy of the University of Pécs at 24–25 °C, provided with standard mouse chow and water ad libitum and maintained under a 12-h light-dark cycle.
Resiniferatoxin pretreatment
Mice were pretreated with increasing doses (30, 70, 100 μg/kg s.c. on 3 consecutive days) of resiniferatoxin (RTX, Sigma-Aldrich), a potent TRPV1 receptor agonist, inducing a permanent depletion of capsaicin-sensitive afferents (desensitization or sensory neuronal deletion)19,34,35. To diminish the acute effects of RTX (bronchoconstriction and mucus secretion) the animals were simultaneously treated with a solution of 4% theophylline-ethylendiamine (Gedeon Richter Plc., Hungary), 4% terbutaline-sulphate (AstraZeneca Ltd., Hungary) and 2% atropine-sulphate (Egis Pharmaceuticals Plc., Hungary)21. The success of the desensitization was controlled by the lack of eye wiping response upon application of capsaicin eye drops (50 μl, 0.1%) 14 days thereafter18. Mice showing nocifensive response were excluded from the study.
Induction of arthritis
The ArithroMab™ (MD Biosciences) antibody cocktail (4 mg/mouse) was injected into the tail vein two weeks after the desensitization under ketamine-xylazine anesthesia (ketamine 100 mg/kg; xylazine 5 mg/kg body weight i.p.). Lipopolysaccharide (100 μg/mouse) was i.p. injected three days thereafter. Sham-treated animals served as controls36.
Arthritis severity and paw volume assessment
Hind paw volume was evaluated using plethysmometry (Ugo Basile 7140) at 2–3 days intervals, while clinical severity was semiquantitatively scored daily using a 0–10 grading system as described previously (0–0.5: normal, 10: maximal inflammation) based on the hallmark features of edema and hyperemia37.
Evaluation of mechanical and thermal hyperalgesia and cold allodynia
The mechanonociceptive threshold of the hindlimb was assessed by dynamic plantar esthesiometry (Ugo Basile 37400), before and following arthritis induction at 2–3 days intervals. Mechanical hyperalgesia was expressed as % of initial control thresholds. Thermal hyperalgesia was evaluated using an increasing temperature hot plate (IITC Life Sciences). The platform of the device was heated until nocifensive reactions were observed (jumping, shaking, lifting or licking either limb), or until the preset maximum was attained (53 °C). The nociceptive threshold was expressed in °C33. Cold allodynia was determined by measuring the hind paw withdrawal latency from 0 °C water in seconds.
Determination of joint function and body weight
To evaluate grasping ability the mice were placed onto a horizontal wire mesh grid which was turned over and maintained in this position for 30s or until the animal fell. This simple test has been previously successfully employed as a rapid screening tool for functional disability due to joint inflammation37. Body weight in grams was measured daily prior to all other tests and was expressed in % compared to an average of three initial self controls38.
In vivo micro-computed tomography (micro-CT) of the ankle joint
The right tibiotarsal joints were imaged repeatedly (days 0, 7, 14, 21) by in vivo micro-CT (SkyScan 1176, Bruker) with a voxel size of 17.5 μm under ketamine-xylazine anesthesia as described earlier (average anesthesia duration: 30–40 min, scan duration 7–10 min). Bone structural changes were evaluated using the CT Analyser® software by applying regions of interests (ROI) of identical size around the ankle joints33. ROIs were applied by first realigning the scanned distal part of the tibia so that it is perpendicular to the axial imaging plane, after which the last slice still including a part of the tibia was identified, and used as an anatomical reference point to establish a 3D geometric ROI. Within the ROIs the structural parameters such as bone volume (μm3), open pore volume (μm3) were expressed as percentage of the total ROI volume. Bone trabecularity was characterized by calculating average trabecular thickness and separation (μm).
Histological sectioning and examination of the tibiotarsal joint
Tibiotarsal joints were obtained on day 21, at the end of the experiment, when the inflammatory phase is over as shown by our functional data and also the literature29,39,40. After fixation, decalcification and dehydration samples were embedded in paraffin, 3–5 µm sections were made and stained with hematoxylin-eosin18 in order to assess synovial proliferation, bone erosion and cartilage distruction and to compare the microscopical bone structure changes with the micro-CT data.
Statistical analysis
Statistical evaluation was performed using GraphPad Prism®. All data points represent mean ± SEM. Functional and micro-CT results were assessed by two-way repeated measures ANOVA followed by Sidak’s post-test for multiple comparisons. Survival curves of grasping ability were analysed by the Mantel-Cox test. In all cases p < 0.05 was considered to be statistically significant.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Chaudhari, K., Rizvi, S. & Syed, B. A. Rheumatoid arthritis: current and future trends. Nat. Rev. Drug Discov. 15, 305–306 (2016).
Kourilovitch, M., Galarza-Maldonado, C. & Ortiz-Prado, E. Diagnosis and classification of rheumatoid arthritis. J. Autoimmun. 48–49, 26–30 (2014).
Courtright, L. J. & Kuzell, W. C. Sparing effect of neurological deficit and trauma on the course of adjuvant arthritis in the rat. Ann. Rheum. Dis. 24, 360–368 (1965).
Thompson, M. & Bywaters, E. G. L. Unilateral Rheumatoid Arthritis following Hemiplegia. Ann. Rheum. Dis. 21, 370–377 (1962).
Talbot, S., Foster, S. L. & Woolf, C. J. Neuroimmunity: Physiology and Pathology. Annu. Rev. Immunol. 34, 421–447 (2016).
Szallasi, A., Cortright, D. N., Blum, C. A. & Eid, S. R. The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nat. Rev. Drug Discov. 6, 357–372 (2007).
Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 29, 355–384 (2013).
Holzer, P. Neurogenic vasodilatation and plasma leakage in the skin. Gen. Pharmacol. 30, 5–11 (1998).
Anichini, M. et al. Substance P in the serum of patients with rheumatoid arthritis. Rev. Rhum. Engl. Ed 64, 18–21 (1997).
Arnalich, F. et al. Neuropeptides and interleukin-6 in human joint inflammation relationship between intraarticular substance P and interleukin-6 concentrations. Neurosci. Lett. 170, 251–254 (1994).
Dirmeier, M. et al. Lower density of synovial nerve fibres positive for calcitonin gene-related peptide relative to substance P in rheumatoid arthritis but not in osteoarthritis. Rheumatol. Oxf. Engl. 47, 36–40 (2008).
Grimsholm, O., Rantapää-Dahlqvist, S. & Forsgren, S. Levels of gastrin-releasing peptide and substance P in synovial fluid and serum correlate with levels of cytokines in rheumatoid arthritis. Arthritis Res. Ther. 7, R416–R426 (2005).
Martínez, C. et al. Serum Levels of Vasoactive Intestinal Peptide as a Prognostic Marker in Early Arthritis. PLoS ONE 9 (2014).
Origuchi, T. et al. Reduction in serum levels of substance P in patients with rheumatoid arthritis by etanercept, a tumor necrosis factor inhibitor. Mod. Rheumatol. 21, 244–250 (2011).
Seoane, I. V. et al. Vasoactive intestinal peptide gene polymorphisms, associated with its serum levels, predict treatment requirements in early rheumatoid arthritis. Sci. Rep. 8, 2035 (2018).
Warner, S. C. et al. Pain in knee osteoarthritis is associated with variation in the neurokinin 1/substance P receptor (TACR1)gene. Eur. J. Pain Lond. Engl. 21, 1277–1284 (2017).
Grasland, A., Pouchot, J., Vinceneux, P. & Ruszniewski, P. Onset of rheumatoid arthritis following curative treatment of a somatostatinoma. Arthritis Rheum. 46, 277–278 (2002).
Helyes, Z. et al. Antiinflammatory and analgesic effects of somatostatin released from capsaicin-sensitive sensory nerve terminals in a Freund’s adjuvant-induced chronic arthritis model in the rat. Arthritis Rheum. 50, 1677–1685 (2004).
Borbély, É. et al. Capsaicin-sensitive sensory nerves exert complex regulatory functions in the serum-transfer mouse model of autoimmune arthritis. Brain. Behav. Immun. 45, 50–59 (2015).
Steinhoff, M. S., von Mentzer, B., Geppetti, P., Pothoulakis, C. & Bunnett, N. W. Tachykinins and their receptors: contributions to physiological control and the mechanisms of disease. Physiol. Rev. 94, 265–301 (2014).
Borbély, É. et al. Role of capsaicin-sensitive nerves and tachykinins in mast cell tryptase-induced inflammation of murine knees. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. Al 65, 725–736 (2016).
Asquith, D. L., Miller, A. M., McInnes, I. B. & Liew, F. Y. Animal models of rheumatoid arthritis. Eur. J. Immunol. 39, 2040–2044 (2009).
Bas, D. B., Su, J., Wigerblad, G. & Svensson, C. I. Pain in rheumatoid arthritis: models and mechanisms. Pain Manag. 6, 265–284 (2016).
Bert, J. et al. The Effect of Treatment with Resiniferatoxin and Capsaicin on Dynamic Weight Bearing Measures and Evoked Pain Responses in a Chronic Inflammatory Arthritis Murine Model. Intern. Med. Rev. Wash. DC Online 2016 (2016).
Moudgil, K. D., Kim, P. & Brahn, E. Advances in Rheumatoid Arthritis Animal Models. Curr. Rheumatol. Rep. 13, 456–463 (2011).
Schopf, L. R., Anderson, K. & Jaffee, B. D. Rat models of arthritis: Similarities, differences, advantages, and disadvantages in the identification of novel therapeutics. In In Vivo Models of Inflammation (eds Stevenson, C. S., Marshall, L. A. & Morgan, D. W.) 1–34 (Birkhäuser Basel, 2006).
Kissin, E. Y., Freitas, C. F. & Kissin, I. Effects Of Intraarticular Resiniferatoxin In Experimental Knee-Joint Arthritis. Anesth. Analg. 101, 1433–1439 (2005).
Lam, F. Y. & Ferrell, W. R. Inhibition of carrageenan induced inflammation in the rat knee joint by substance P antagonist. Ann. Rheum. Dis. 48, 928–932 (1989).
Bas, D. B. et al. Collagen antibody-induced arthritis evokes persistent pain with spinal glial involvement and transient prostaglandin dependency. Arthritis Rheum. 64, 3886–3896 (2012).
Pan, H.-L., Khan, G. M., Alloway, K. D. & Chen, S.-R. Resiniferatoxin Induces Paradoxical Changes in Thermal and Mechanical Sensitivities in Rats: Mechanism of Action. J. Neurosci. 23, 2911–2919 (2003).
Grahnemo, L. et al. Trabecular bone loss in collagen antibody-induced arthritis. Arthritis Res. Ther. 17, 189 (2015).
Oestergaard, S. et al. Evaluation of cartilage and bone degradation in a murine collagen antibody-induced arthritis model. Scand. J. Immunol. 67, 304–312 (2008).
Botz, B. et al. Differential regulatory role of pituitary adenylate cyclase-activating polypeptide in the serum-transfer arthritis model. Arthritis Rheumatol. Hoboken NJ 66, 2739–2750 (2014).
Szolcsanyi, J., Szallasi, A., Szallasi, Z., Joo, F. & Blumberg, P. M. Resiniferatoxin: an ultrapotent selective modulator of capsaicin-sensitive primary afferent neurons. J. Pharmacol. Exp. Ther. 255, 923–928 (1990).
Olah, Z. et al. Ligand-induced dynamic membrane changes and cell deletion conferred by vanilloid receptor 1. J. Biol. Chem. 276, 11021–11030 (2001).
Khachigian, L. M. Collagen antibody-induced arthritis. Nat. Protoc. 1, 2512–2516 (2006).
Jakus, Z., Simon, E., Balázs, B. & Mócsai, A. Genetic deficiency of Syk protects mice from autoantibody-induced arthritis. Arthritis Rheum. 62, 1899–1910 (2010).
Horváth, Á. et al. Transient receptor potential ankyrin 1 (TRPA1) receptor is involved in chronic arthritis: in vivo study using TRPA1-deficient mice. Arthritis Res. Ther. 18, 6 (2016).
Hutamekalin, P. et al. Collagen antibody-induced arthritis in mice: development of a new arthritogenic 5-clone cocktail of monoclonal anti-type II collagen antibodies. J. Immunol. Methods 343, 49–55 (2009).
Nandakumar, K. S. & Holmdahl, R. Efficient promotion of collagen antibody induced arthritis (CAIA) using four monoclonal antibodies specific for the major epitopes recognized in both collagen induced arthritis and rheumatoid arthritis. J. Immunol. Methods 304, 126–136 (2005).
Acknowledgements
National Brain Research Program 20017-1.2.1-NKP-2017-00002, GINOP-2.3.2-15-2016-00050 (“PEPSYS: Complexity of peptidergic signalization and its role in systemic diseases”), EFOP-3.6.1- 16-2016- 00004 (“Comprehensive development for implementing smart specialization strategies at the University of Pécs”), and EFOP-3.6.2-16-2017-00008 (“The role of neuro-inflammation in neurodegeneration: from molecules to clinics”) supported this work. B. Botz was supported by the ÚNKP-17-4-I-PTE-96 New National Excellence Program of the Ministry of Human Capacities. The authors are grateful to Anikó Perkecz for her expert help in histological processing, to Dóra Ömböli for the assistance in functional measurements.
Author information
Authors and Affiliations
Contributions
É.B. and B.B. carried out all functional measurements, T.K. performed and analysed the in vivo micro CT scans, and assisted in writing the paper. K. Sz. performed the histological processing and evaluation. B.B., Z.H. and É.B. wrote the manuscript. B.B., Z.H., E.P. and J. Sz. designed the experiments. Z.H., E.P. and J. Sz. also assisted in data analysis and helped to write the manuscript. All authors read and approved the final manuscript, except K. Sz. whose authorship is posthumous.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Borbély, É., Kiss, T., Szabadfi, K. et al. Complex Role of Capsaicin-Sensitive Afferents in the Collagen Antibody-Induced Autoimmune Arthritis of the Mouse. Sci Rep 8, 15916 (2018). https://doi.org/10.1038/s41598-018-34005-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34005-6
- Springer Nature Limited
Keywords
This article is cited by
-
Alterations of voluntary behavior in the course of disease progress and pharmacotherapy in mice with collagen-induced arthritis
Arthritis Research & Therapy (2019)