Abstract
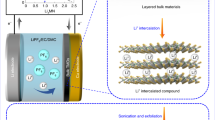
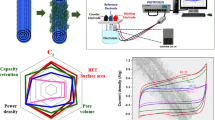
Transition metal tellurides (TMTs) have been ideal materials for exploring exotic properties in condensed-matter physics, chemistry and materials science1,2,3. Although TMT nanosheets have been produced by top-down exfoliation, their scale is below the gram level and requires a long processing time, restricting their effective application from laboratory to market4,5,6,7,8. We report the fast and scalable synthesis of a wide variety of MTe2 (M = Nb, Mo, W, Ta, Ti) nanosheets by the solid lithiation of bulk MTe2 within 10 min and their subsequent hydrolysis within seconds. Using NbTe2 as a representative, we produced more than a hundred grams (108 g) of NbTe2 nanosheets with 3.2 nm mean thickness, 6.2 µm mean lateral size and a high yield (>80%). Several interesting quantum phenomena, such as quantum oscillations and giant magnetoresistance, were observed that are generally restricted to highly crystalline MTe2 nanosheets. The TMT nanosheets also perform well as electrocatalysts for lithium–oxygen batteries and electrodes for microsupercapacitors (MSCs). Moreover, this synthesis method is efficient for preparing alloyed telluride, selenide and sulfide nanosheets. Our work opens new opportunities for the universal and scalable synthesis of TMT nanosheets for exploring new quantum phenomena, potential applications and commercialization.





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available in the paper and its Supplementary Information. Other raw data are available from the corresponding authors on request. Source data are provided with this paper.
References
Zhou, J. et al. A library of atomically thin metal chalcogenides. Nature 556, 355–359 (2018).
Fiori, G. et al. Electronics based on two-dimensional materials. Nat. Nanotechnol. 9, 768–779 (2014).
Pomerantseva, E. & Gogotsi, Y. Two-dimensional heterostructures for energy storage. Nat. Energy 2, 17089 (2017).
Choi, S. H. et al. Large-scale synthesis of graphene and other 2D materials towards industrialization. Nat. Commun. 13, 1484 (2022).
Editorial. Moving towards the market. Nat. Mater. 18, 519. https://doi.org/10.1038/s41563-019-0394-4 (2019).
Li, J. et al. Printable two-dimensional superconducting monolayers. Nat. Mater. 20, 181–187 (2021).
Zhang, L. et al. Solid phase exfoliation for producing dispersible transition metal dichalcogenides nanosheets. Adv. Funct. Mater. 30, 2004139 (2020).
Coleman, J. N. et al. Two-dimensional nanosheets produced by liquid exfoliation of layered materials. Science 331, 568–571 (2011).
Li, H. et al. Two-dimensional metal telluride atomic crystals: preparation, physical properties, and applications. Adv. Funct. Mater. 31, 2010901 (2021).
Keum, D. H. et al. Bandgap opening in few-layered monoclinic MoTe2. Nat. Phys. 11, 482–486 (2015).
Xu, X. et al. Seeded 2D epitaxy of large-area single-crystal films of the van der Waals semiconductor 2H MoTe2. Science 372, 195–200 (2021).
Ali, M. N. et al. Large, non-saturating magnetoresistance in WTe2. Nature 514, 205–208 (2014).
Wang, W. et al. Evidence for an edge supercurrent in the Weyl superconductor MoTe2. Science 368, 534–537 (2020).
Jindal, A. et al. Coupled ferroelectricity and superconductivity in bilayer Td-MoTe2. Nature 613, 48–52 (2023).
Zhao, X. et al. Selective electrochemical production of hydrogen peroxide at zigzag edges of exfoliated molybdenum telluride nanoflakes. Natl Sci. Rev. 7, 1360–1366 (2020).
McGlynn, J. C. et al. The rapid electrochemical activation of MoTe2 for the hydrogen evolution reaction. Nat. Commun. 10, 4916 (2019).
Nicolosi, V., Chhowalla, M., Kanatzidis, M. G., Strano, M. S. & Coleman, J. N. Liquid exfoliation of layered materials. Science 340, 1226419 (2013).
Yang, S., Zhang, P., Nia, A. S. & Feng, X. Emerging 2D materials produced via electrochemistry. Adv. Mater. 32, 1907857 (2020).
Yang, R. et al. Synthesis of atomically thin sheets by the intercalation-based exfoliation of layered materials. Nat. Synth. 2, 101–118 (2023).
Yang, R. et al. High-yield production of mono- or few-layer transition metal dichalcogenide nanosheets by an electrochemical lithium ion intercalation-based exfoliation method. Nat. Protoc. 17, 358–377 (2022).
Lin, Z. et al. Solution-processable 2D semiconductors for high-performance large-area electronics. Nature 562, 254–258 (2018).
Peng, J. et al. High phase purity of large-sized 1T′-MoS2 monolayers with 2D superconductivity. Adv. Mater. 31, 1900568 (2019).
Joensen, P., Frindt, R. F. & Morrison, S. R. Single-layer MoS2. Mater. Res. Bull. 21, 457–461 (1986).
Zheng, J. et al. High yield exfoliation of two-dimensional chalcogenides using sodium naphthalenide. Nat. Commun. 5, 2995 (2014).
Tsai, H.-L., Heising, J., Schindler, J. L., Kannewurf, C. R. & Kanatzidis, M. G. Exfoliated-restacked phase of WS2. Chem. Mater. 9, 879–882 (1997).
Kanatzidis, M. G. & Marks, T. J. Tetrahydroborate intercalation reagents. Convenient, straightforward routes to known and new types of layered intercalation compounds. Inorg. Chem. 26, 783–784 (1987).
Ono, M. et al. New lithium- and ethylenediamine-intercalated superconductors Lix(C2H8N2)yWTe2. J. Phys. Soc. Jpn. 90, 014706 (2020).
Peng, J. et al. Two-dimensional tellurium nanosheets exhibiting an anomalous switchable photoresponse with thickness dependence. Angew. Chem. Int. Ed. 57, 13533–13537 (2018).
Jawaid, A. et al. Redox exfoliation of layered transition metal dichalcogenides. ACS Nano 11, 635–646 (2017).
Zhang, C. et al. Mass production of 2D materials by intermediate-assisted grinding exfoliation. Natl Sci. Rev. 7, 324–332 (2020).
Hangyo, M., Nakashima, S.-I. & Mitsuishi, A. Raman spectroscopic studies of MX2-type layered compounds. Ferroelectrics 52, 151–159 (1983).
Li, J. et al. Synthesis of ultrathin metallic MTe2 (M = V, Nb, Ta) single-crystalline nanoplates. Adv. Mater. 30, 1801043 (2018).
Mleczko, M. J. et al. High current density and low thermal conductivity of atomically thin semimetallic WTe2. ACS Nano 10, 7507–7514 (2016).
Chia, X., Ambrosi, A., Lazar, P., Sofer, Z. & Pumera, M. Electrocatalysis of layered Group 5 metallic transition metal dichalcogenides (MX2, M = V, Nb, and Ta; X = S, Se, and Te). J. Mater. Chem. A 4, 14241–14253 (2016).
Huang, J.-H. et al. Polymorphism control of layered MoTe2 through two-dimensional solid-phase crystallization. Sci. Rep. 9, 8810 (2019).
Cui, J. et al. Transport evidence of asymmetric spin–orbit coupling in few-layer superconducting 1Td-MoTe2. Nat. Commun. 10, 2044 (2019).
Qi, Y. et al. Superconductivity in Weyl semimetal candidate MoTe2. Nat. Commun. 7, 11038 (2016).
Chen, F. C. et al. Extremely large magnetoresistance in the type-II Weyl semimetal MoTe2. Phys. Rev. B 94, 235154 (2016).
Benalcazar, W. A., Bernevig, B. A. & Hughes, T. L. Quantized electric multipole insulators. Science 357, 61–66 (2017).
Kowalczyk, H. et al. Gate and temperature driven phase transitions in few-layer MoTe2. ACS Nano 17, 6708–6718 (2023).
Huang, F.-T. et al. Polar and phase domain walls with conducting interfacial states in a Weyl semimetal MoTe2. Nat. Commun. 10, 4211 (2019).
Rhodes, D. A. et al. Enhanced superconductivity in monolayer Td-MoTe2. Nano Lett. 21, 2505–2511 (2021).
Tiwari, A. et al. Giant c-axis nonlinear anomalous Hall effect in Td-MoTe2 and WTe2. Nat. Commun. 12, 2049 (2021).
Zhong, S. et al. Origin of magnetoresistance suppression in thin γ–MoTe2. Phys. Rev. B 97, 241409 (2018).
Ma, T. et al. Growth of bilayer MoTe2 single crystals with strong non-linear Hall effect. Nat. Commun. 13, 5465 (2022).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (grants 22125903, 22209176, 22279137, 12374035, 22075279, E211L5031R and E311L5191R), Basic Science Center Project of the National Natural Science Foundation of China (52188101), the National Key R&D Program of China (grants 2022YFA1504100 and 2023YFB4005204), Innovation Program for Quantum Science and Technology (grant 2021ZD0302600), the China Postdoctoral Science Foundation (2021M703145), International Postdoctoral Exchange Fellowship Program (Talent-Introduction Program) (YJ20210311), the Dalian Innovation Support Plan for High Level Talents (2019RT09), Liaoning Natural Science Foundation (2023-BS-007) and Shanghai Pujiang Talent plan project (23PJ1402000). We thank P. A. Thrower for carefully reviewing and revising the manuscript.
Author information
Authors and Affiliations
Contributions
Z.-S.W., N.K. and H.-M.C. conceived and supervised the project. L.Z. conducted the production and characterization of the TMT nanosheets. Z.Y., Y.F., S.F. and D.-M.S. carried out transport measurements of the TMT nanosheet devices. Q.J. performed the XRD and XPS of the alloyed nanosheets. Y.D. characterized the Li–O2 batteries. P.D. tested the electrochemical performance of the MSCs. Z.B. characterized the lithium batteries. J.Ma., J.Mi, S.W., Z.G. and S.Z. carried out the 3D printing, photolithography and screen printing of the NbTe2 nanosheet ink. H.Z. characterized the EMI shielding performance of the Mo0.5W0.5Te2/CNT film. M.L. obtained the spherical aberration-corrected TEM images. L.Z., Z.Y., P.D., N.K., Z.-S.W. and H.-M.C. wrote the manuscript. All authors contributed to discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Zheng Liu, Kian Ping Loh and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Illustration of the hydrolysis process of lithiated NbTe2 bulk powder into a nanosheet colloid solution.
a–g, Photographs of the exfoliation process of Li1NbTe2 by adding distille water to the glass bottles at different times. Li1NbTe2 is hydrolysed into NbTe2 nanosheets by the generation of H2 gas, resulting in a homogenous colloidal dispersion. h, Tyndall effect of a NbTe2 nanosheet colloid solution.
Extended Data Fig. 2 Morphology and elemental characterization of various TMT nanosheets using a long lithium intercalation time when the molar ratio of LiBH4 to MTe2 is equal to 1.
a–a4, MoTe2 nanosheets after lithium intercalation for 24 h. a, HAADF-STEM image. EDS elemental maps of Mo (a1), Te (a2) and mixed Mo and Te (a3). a4, Table of the elemental content of the marked area in a3. b–b4, WTe2 nanosheets after lithium intercalation for 6 h. b, HAADF-STEM image. EDS elemental maps of W (b1), Te (b2) and mixed W and Te (b3). b4, Table of the elemental content of the marked area in b3. c–c4, NbTe2 nanosheets after lithium intercalation for 24 h. c, HAADF-STEM image. EDS elemental maps of Nb (c1), Te (c2) and mixed Nb and Te (c3). c4, Table of the elemental content of the marked area in c3. d–d4, TaTe2 nanosheets after lithium intercalation for 6 h. d, HAADF-STEM image. EDS elemental maps of Ta (d1), Te (d2) and mixed Ta and Te (d3). d4, Table of the elemental content of the marked area in d3. e–e4, TiTe2 nanosheets after lithium intercalation for 6 h. e, HAADF-STEM image, EDS elemental maps of Ti (e1), Te (e2) and mixed Ti and Te (e3). e4, Table of the elemental content of the marked area in e3. The HAADF-STEM EDS elemental maps show that the morphology of the decomposition products is a nanobelt and that Te is present in all samples after a long lithium intercalation time.
Extended Data Fig. 3 The influence of LiBH4 content on exfoliation yield and solution concentration.
a, MoTe2 nanosheets. b, WTe2 nanosheets. c, NbTe2 nanosheets. d, TaTe2 nanosheets. e, TiTe2 nanosheets. We found that molar ratios of LiBH4 to MTe2 ranging from 0.75 to 1 are suitable for achieving a high exfoliation yield of more than 60% for MoTe2, 50% for WTe2, 80% for NbTe2, 80% for TaTe2 and 40% for TiTe2.
Extended Data Fig. 4 Summary of synthesizable materials using solid lithiation and exfoliation.
a, Periodic table showing the metals (highlighted in brown) and chalcogens (highlighted in blue) that have the possibility of forming layered sulfides, selenides and tellurides. b–m, AFM images and height profiles (insets) of 12 types of telluride, selenide and sulfide nanosheet. b, Mo0.5W0.5Te2. c, Nb0.5Ta0.5Te2. d, NbSeTe. e, Mo(SSeTe)2. f, MoSSe. g, TaSSe. h, Mo0.5Nb0.5Se2. i, (MoNbTa)Se2. j, Mo0.5Ta0.5S2. k, Ta0.5Re0.5S2. l, (MoWTa)S2. m, (TiZrHf)S2.
Extended Data Fig. 5 Structural characterizations of a (MoNbTa)Se2 nanosheet.
a, Low-magnification STEM image. b, Atomic-resolution HAADF-STEM image. c, FFT pattern. d, Magnified HAADF-STEM image of b. e–e4, HAADF-STEM image and EDS elemental maps of Mo, Nb, Ta and Se.
Extended Data Fig. 6 Transport properties of a Mo0.5W0.5Te2 nanosheet.
a, Low-temperature transport measurement of a Mo0.5W0.5Te2 thin nanosheet. The temperature dependence of resistance shows metallic behaviour. Upper inset, SEM image of a fabricated device with multiterminal Hall-bar geometry. Lower inset, the Hall measurement shows a carrier density of 9.6 × 1013 cm−2. b, Magnetoresistance, defined as (R(B) − R(B = 0))/R(B = 0), plotted as a function of magnetic field for Mo0.5W0.5Te2 thin films with thicknesses ranging from 10 to 70 nm. It is seen that the positive magnetoresistance is substantially decreased with decreasing thickness.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, L., Yang, Z., Feng, S. et al. Metal telluride nanosheets by scalable solid lithiation and exfoliation. Nature 628, 313–319 (2024). https://doi.org/10.1038/s41586-024-07209-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-024-07209-2
- Springer Nature Limited