Abstract
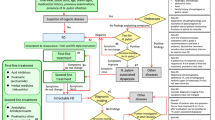
Symptoms related to abnormal gastrointestinal motility and function are common. Oropharyngeal and oesophageal dysphagia, heartburn, bloating, abdominal pain and alterations in bowel habits are among the most frequent reasons for seeking medical attention from internists or general practitioners and are also common reasons for referral to gastroenterologists and colorectal surgeons. However, the nonspecific nature of gastrointestinal symptoms, the absence of a definitive diagnosis on routine investigations (such as endoscopy, radiology or blood tests) and the lack of specific treatments make disease management challenging. Advances in technology have driven progress in the understanding of many of these conditions. This Review serves as an introduction to a series of Consensus Statements on the clinical measurements of gastrointestinal motility, function and sensitivity. A structured, evidence-based approach to the initial assessment and empirical treatment of patients presenting with gastrointestinal symptoms is discussed, followed by an outline of the contribution of modern physiological measurement on the management of patients in whom the cause of symptoms has not been identified with other tests. Discussions include the indications for and utility of high-resolution manometry, ambulatory pH-impedance monitoring, gastric emptying studies, breath tests and investigations of anorectal structure and function in day-to-day practice and clinical management.
Key points
-
Symptoms have poor specificity for gastrointestinal diseases, and there is a marked overlap between ‘organic disease’ (including major motility disorders) and functional gastrointestinal disease, underlining the need for testing to guide treatment.
-
New technology has driven progress, improved our understanding of gastrointestinal physiology and revolutionized the clinical measurement of gastrointestinal motility and function from oropharynx to anorectum.
-
Adherence to validated methodology is essential for the assessment of gastrointestinal motility and function to provide meaningful results.
-
Diagnoses based on valid, objective metrics (for example, Chicago Classification for oesophageal motility disorders, gastric emptying retention time and colonic transit times) are replacing subjective assessments from physiological studies.
-
High-resolution manometry has improved the inter-observer agreement and accuracy of diagnoses in patients with disorders of oesophageal and anorectal motility and function.
-
The importance of abnormal visceral sensitivity has been demonstrated in patients with functional gastrointestinal diseases.




Similar content being viewed by others
References
Farthing, M. et al. Survey of digestive health across Europe: Final report. Part 1: The burden of gastrointestinal diseases and the organisation and delivery of gastroenterology services across Europe. United Eur. Gastroenterol. J. 2, 539–543 (2014).
Anderson, P. et al. Survey of digestive health across Europe: Final report. Part 2: The economic impact and burden of digestive disorders. United Eur. Gastroenterol. J. 2, 544–546 (2014).
Canavan, C., West, J. & Card, T. The epidemiology of irritable bowel syndrome. Clin. Epidemiol. 6, 71–80 (2014).
Whitehead, W. E., Burnett, C. K., Cook, E. W. 3rd & Taub, E. Impact of irritable bowel syndrome on quality of life. Dig. Dis. Sci. 41, 2248–2253 (1996).
Drossman, D. A. Functional gastrointestinal disorders: history, pathophysiology, clinical features, and Rome IV. Gastroenterology 150, 1262–1279.e2 (2016).
Fox, M. R. & Bredenoord, A. J. Oesophageal high-resolution manometry: moving from research into clinical practice. Gut 57, 405–423 (2008).
Boeckxstaens, G. et al. Fundamentals of neurogastroenterology: physiology/motility — sensation. Gastroenterology 150, 1292–1304.e2 (2016).
Kahrilas, P. J. et al. Expert consensus document: Advances in the management of oesophageal motility disorders in the era of high-resolution manometry: a focus on achalasia syndromes. Nat. Rev. Gastroenterol. Hepatol. 14, 677–688 (2017).
Savarino, E. et al. Expert consensus document: Advances in the physiological assessment and diagnosis of GERD. Nat. Rev. Gastroenterol. Hepatol. 14, 665–676 (2017).
Keller, J. et al. Expert consensus document: Advances in the diagnosis and classification of gastric and intestinal motility disorders. Nat. Rev. Gastroenterol. Hepatol. https://doi.org/10.1038/nrgastro.2018.7 (2018).
Carrington, E. V. et al. Expert consensus document: Advances in the evaluation of anorectal function. Nat. Rev. Gastroenterol. Hepatol. https://doi.org/10.1038/nrgastro.2018.27 (2018).
Ford, A. C., Marwaha, A., Lim, A. & Moayyedi, P. What is the prevalence of clinically significant endoscopic findings in subjects with dyspepsia? Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 8, 830–837 (2010).
Kapoor, N., Bassi, A., Sturgess, R. & Bodger, K. Predictive value of alarm features in a rapid access upper gastrointestinal cancer service. Gut 54, 40–45 (2005).
NICE. Dyspepsia: managing dyspepsia in adults in primary care. NICE https://www.nice.org.uk/Guidance/cg17 (2004).
Talley, N. & Vakil, N. Guidelines for the management of dyspepsia. Am. J. Gastroenterol. 100, 2324–2337 (2005).
Ford, A. C., Marwaha, A., Lim, A. & Moayyedi, P. Systematic review and meta-analysis of the prevalence of irritable bowel syndrome in individuals with dyspepsia. Clin. Gastroenterol. Hepatol. 8, 401–409 (2010).
Locke, G. R. et al. Overlap of gastrointestinal symptom complexes in a US community. Neurogastroenterol Motil. 17, 29–34 (2005).
Hungin, A. P., Hill, C. & Raghunath, A. Systematic review: frequency and reasons for consultation for gastro-oesophageal reflux disease and dyspepsia. Aliment. Pharmacol. Ther. 30, 331–342 (2009).
Ford, A. C., Forman, D., Bailey, A. G., Axon, A. T. & Moayyedi, P. Initial poor quality of life and new onset of dyspepsia: results from a longitudinal 10-year follow-up study. Gut 56, 321–327 (2007).
Drossman, D. A. et al. What determines severity among patients with painful functional bowel disorders? Am. J. Gastroenterol. 95, 974–980 (2000).
Delaney, B., Ford, A. C., Forman, D., Moayyedi, P. & Qume, M. Initial management strategies for dyspepsia. Cochrane Database Syst. Rev. 4, CD001961 (2005).
Wang, W. H. et al. Effects of proton-pump inhibitors on functional dyspepsia: a meta-analysis of randomized placebo-controlled trials. Clin. Gastroenterol. Hepatol. 5, 178–185 (2007).
Delaney, B. C. et al. Helicobacter pylori test and treat versus proton pump inhibitor in initial management of dyspepsia in primary care: multicentre randomised controlled trial (MRC-CUBE trial). BMJ 336, 651–654 (2008).
Ford, A. C. et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ 337, a2313 (2008).
Moayyedi, P. et al. Systematic review: Antacids, H2-receptor antagonists, prokinetics, bismuth and sucralfate therapy for non-ulcer dyspepsia. Aliment. Pharmacol. Ther. 17, 1215–1227 (2003).
Vanheel, H. & Tack, J. Therapeutic options for functional dyspepsia. Dig. Dis. 32, 230–234 (2014).
Ford, A. C., Talley, N. J., Schoenfeld, P. S., Quigley, E. M. & Moayyedi, P. Efficacy of antidepressants and psychological therapies in irritable bowel syndrome: systematic review and meta-analysis. Gut 58, 367–378 (2009).
Yin, J., Song, J., Lei, Y., Xu, X. & Chen, J. D. Prokinetic effects of mirtazapine on gastrointestinal transit. Am. J. Physiol. Gastrointest. Liver Physiol. 306, G796–G801 (2014).
Talley, N. J. et al. Effect of amitriptyline and escitalopram on functional dyspepsia: a multicenter, randomized controlled study. Gastroenterology 149, 340–349.e2 (2015).
Malamood, M., Roberts, A., Kataria, R., Parkman, H. P. & Schey, R. Mirtazapine for symptom control in refractory gastroparesis. Drug Des. Devel. Ther. 11, 1035–1041 (2017).
Santoro, G. A., Eitan, B. Z., Pryde, A. & Bartolo, D. C. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis. Colon Rectum 43, 1676–1681 (2000).
Lomer, M. C. Review article: the aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Aliment. Pharmacol. Ther. 41, 262–275 (2014).
Olausson, E. A. et al. A small particle size diet reduces upper gastrointestinal symptoms in patients with diabetic gastroparesis: a randomized controlled trial. Am. J. Gastroenterol. 109, 375–385 (2014).
Tucker, E., Knowles, K., Wright, J. & Fox, M. R. Rumination variations: aetiology and classification of abnormal behavioural responses to digestive symptoms based on high-resolution manometry studies. Aliment. Pharmacol. Ther. 37, 263–274 (2013).
Barba, E. et al. Abdominothoracic mechanisms of functional abdominal distension and correction by biofeedback. Gastroenterology 148, 732–739 (2015).
Cook, I. J. Diagnostic evaluation of dysphagia. Nat. Clin. Pract. Gastroenterol. Hepatol. 5, 393–403 (2008).
Hiss, S. G. & Postma, G. N. Fiberoptic endoscopic evaluation of swallowing. Laryngoscope 113, 1386–1393 (2003).
Pal, A., Williams, R. B., Cook, I. J. & Brasseur, J. G. Intrabolus pressure gradient identifies pathological constriction in the upper esophageal sphincter during flow. Am. J. Physiol. Gastrointest. Liver Physiol. 285, G1037–G1048 (2003).
Omari, T. I. et al. A method to objectively assess swallow function in adults with suspected aspiration. Gastroenterology 140, 1454–1463 (2011).
Vardar, R., Sweis, R., Anggiansah, A., Wong, T. & Fox, M. R. Upper esophageal sphincter and esophageal motility in patients with chronic cough and reflux: assessment by high-resolution manometry. Dis. Esophagus 26, 219–225 (2013).
Rommel, N. & Hamdy, S. Oropharyngeal dysphagia: manifestations and diagnosis. Nat. Rev. Gastroenterol. Hepatol. 13, 49–59 (2016).
Fox, M. & Sweis, R. Future directions in esophageal motility and function - new technology and methodology. Neurogastroenterol. Motil. 24 (Suppl. 1), 48–56 (2012).
Kahrilas, P. J. et al. The Chicago Classification of esophageal motility disorders, v3.0. Neurogastroenterol Motil. 27, 160–174 (2015).
Kwiatek, M. A. et al. Esophagogastric junction distensibility after fundoplication assessed with a novel functional luminal imaging probe. J. Gastrointest. Surg. 14, 268–276 (2010).
Xiao, Y. et al. Lack of correlation between HRM metrics and symptoms during the manometric protocol. Am. J. Gastroenterol. 109, 521–526 (2014).
Cisternas, D. et al. Anxiety can significantly explain bolus perception in the context of hypotensive esophageal motility: results of a large multicenter study in asymptomatic individuals. Neurogastroenterol. Motil. 29, e13088 (2017).
Carlson, D. A. et al. Diagnosis of esophageal motility disorders: esophageal pressure topography versus conventional line tracing. Am. J. Gastroenterol. 110, 967–977 (2015).
Fox, M. R. et al. Inter-observer agreement for diagnostic classification of esophageal motility disorders defined in high-resolution manometry. Dis. Esophagus 28, 711–719 (2014).
Roman, S. et al. High-resolution manometry improves the diagnosis of esophageal motility disorders in patients with dysphagia: a randomized multicenter study. Am. J. Gastroenterol. 111, 372–380 (2016).
Pandolfino, J. E. et al. Achalasia: a new clinically relevant classification by high-resolution manometry. Gastroenterology 135, 1526–1533 (2008).
Rohof, W. O. et al. Outcomes of treatment for achalasia depend on manometric subtype. Gastroenterology 144, 718–725 (2013).
Sweis, R., Anggiansah, A., Wong, T., Brady, G. & Fox, M. Assessment of esophageal dysfunction and symptoms during and after a standardized test meal: development and clinical validation of a new methodology utilizing high-resolution manometry. Neurogastroenterol. Motil. 26, 215–228 (2014).
Hollenstein, M. et al. Pharyngeal swallowing and oesophageal motility during a solid meal test: a prospective study in healthy volunteers and patients with major motility disorders. Lancet Gastroenterol. Hepatol. 2, 644–653 (2017).
Ang, D. et al. Diagnostic yield of high-resolution manometry with a solid test meal for clinically relevant, symptomatic oesophageal motility disorders: serial diagnostic study. Lancet Gastroenterol. Hepatol. 2, 654–661 (2017).
Wang, Y. T. et al. Investigation of dysphagia after antireflux surgery by high-resolution manometry: impact of multiple water swallows and a solid test meal on diagnosis, management, and clinical outcome. Clin. Gastroenterol. Hepatol. 13, 1575–1583 (2015).
Ang, D. et al. High resolution manometry with a solid test meal increases diagnostic sensitivity for clinically relevant, symptomatic esophageal motility disorders: results from a large case series. Lancet Gastroenterol. Hepatol. (in press).
Fox, M. & Forgacs, I. Gastro-oesophageal reflux disease. BMJ 332, 88–93 (2006).
Katz, P. O., Gerson, L. B. & Vela, M. F. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am. J. Gastroenterol. 108, 308–328 (2013).
Jobe, B. A. et al. Preoperative diagnostic workup before antireflux surgery: an evidence and experience-based consensus of the Esophageal Diagnostic Advisory Panel. J. Am. Coll. Surg. 217, 586–597 (2013).
Poh, C. H. et al. Upper GI tract findings in patients with heartburn in whom proton pump inhibitor treatment failed versus those not receiving antireflux treatment. Gastrointest. Endosc. 71, 28–34 (2010).
Vakil, N., van Zanten, S. V., Kahrilas, P., Dent, J. & Jones, R. The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am. J. Gastroenterol. 101, 1900–1920 (2006).
Roman, S. et al. Majority of symptoms in esophageal reflux PPI non-responders are not related to reflux. Neurogastroenterol. Motil. 27, 1667–1674 (2015).
Dent, J. et al. Accuracy of the diagnosis of GORD by questionnaire, physicians and a trial of proton pump inhibitor treatment: the Diamond Study. Gut 59, 714–721 (2010).
Mainie, I. et al. Acid and non-acid reflux in patients with persistent symptoms despite acid suppressive therapy: a multicentre study using combined ambulatory impedance-pH monitoring. Gut 55, 1398–1402 (2006).
Jasper, D. et al. Prolonged measurement improves the assessment of the barrier function of the esophago-gastric junction by high-resolution manometry. Neurogastroenterol. Motil. 29, e12925 (2016).
Tolone, S. et al. Esophagogastric junction morphology is associated with a positive impedance-pH monitoring in patients with GERD. Neurogastroenterol. Motil. 27, 1175–1182 (2015).
Gor, P. et al. Interrogation of esophagogastric junction barrier function using the esophagogastric junction contractile integral: an observational cohort study. Dis. Esophagus 29, 820–828 (2016).
Roman, S. et al. Validation of criteria for the definition of transient lower esophageal sphincter relaxations using high-resolution manometry. Neurogastroenterol. Motil. 29, e12920 (2016).
Lin, S., Ke, M., Xu, J. & Kahrilas, P. J. Impaired esophageal emptying in reflux disease. Am. J. Gastroenterol. 89, 1003–1006 (1994).
Bredenoord, A. J., Hemmink, G. J. & Smout, A. J. Relationship between gastro-oesophageal reflux pattern and severity of mucosal damage. Neurogastroenterol. Motil. 21, 807–812 (2009).
Ang, D. et al. Rapid Drink Challenge in high-resolution manometry: an adjunctive test for detection of esophageal motility disorders. Neurogastroenterol. Motil. 29, e12902 (2016).
Shaker, A. et al. Multiple rapid swallow responses during esophageal high-resolution manometry reflect esophageal body peristaltic reserve. Am. J. Gastroenterol. 108, 1706–1712 (2013).
Daum, C. et al. Failure to respond to physiologic challenge characterizes esophageal motility in erosive gastro-esophageal reflux disease. Neurogastroenterol. Motil. 23, 517–e200 (2011).
Martinucci, I. et al. Vigor of peristalsis during multiple rapid swallows is inversely correlated with acid exposure time in patients with NERD. Neurogastroenterol. Motil. 28, 243–250 (2016).
Sifrim, D. et al. Weakly acidic reflux in patients with chronic unexplained cough during 24 hour pressure, pH, and impedance monitoring. Gut 54, 449–454 (2005).
Bredenoord, A. J., Weusten, B. L., Sifrim, D., Timmer, R. & Smout, A. J. Aerophagia, gastric, and supragastric belching: a study using intraluminal electrical impedance monitoring. Gut 53, 1561–1565 (2004).
Sweis, R. et al. Patient acceptance and clinical impact of Bravo monitoring in patients with previous failed catheter-based studies. Aliment. Pharmacol. Ther. 29, 669–676 (2009).
Sweis, R., Fox, M., Anggiansah, A. & Wong, T. Prolonged, wireless pH-studies have a high diagnostic yield in patients with reflux symptoms and negative 24-h catheter-based pH-studies. Neurogastroenterol. Motil. 23, 419–426 (2011).
Scarpulla, G., Camilleri, S., Galante, P., Manganaro, M. & Fox, M. The impact of prolonged pH measurements on the diagnosis of gastro-esophageal reflux disease: four day wireless pH studies. Am. J. Gastroenterol. 102, 2642–2647 (2007).
Gyawali, C. P. et al. Modern diagnosis of GERD: the Lyon Consensus. Gut. https://doi.org/10.1136/gutjnl-2017-314722 (2018).
Weijenborg, P. W., Cremonini, F., Smout, A. J. & Bredenoord, A. J. PPI therapy is equally effective in well-defined non-erosive reflux disease and in reflux esophagitis: a meta-analysis. Neurogastroenterol Motil. 24, 747–57, e350 (2012).
Mainie, I. et al. Fundoplication eliminates chronic cough due to non-acid reflux identified by impedance pH monitoring. Thorax 60, 521–523 (2005).
Broeders, J. A. et al. Oesophageal acid hypersensitivity is not a contraindication to Nissen fundoplication. Br. J. Surg. 96, 1023–1030 (2009).
Broeders, J. A. et al. Long-term outcome of Nissen fundoplication in non-erosive and erosive gastro-oesophageal reflux disease. Br. J. Surg. 97, 845–852 (2010).
Weijenborg, P. W., de Schepper, H. S., Smout, A. J. & Bredenoord, A. J. Effects of antidepressants in patients with functional esophageal disorders or gastroesophageal reflux disease: a systematic review. Clin. Gastroenterol. Hepatol. 13, 251–259.e1 (2015).
Rao, S. S. et al. Evaluation of gastrointestinal transit in clinical practice: position paper of the American and European Neurogastroenterology and Motility Societies. Neurogastroenterol. Motil. 23, 8–23 (2011).
Camilleri, M. et al. American Neurogastroenterology and Motility Society consensus statement on intraluminal measurement of gastrointestinal and colonic motility in clinical practice. Neurogastroenterol. Motil. 20, 1269–1282 (2008).
Stanghellini, V. & Tack, J. Gastroparesis: separate entity or just a part of dyspepsia? Gut 63, 1972–1978 (2014).
Tougas, G. et al. Assessment of gastric emptying using a low fat meal: establishment of international control values. Am. J. Gastroenterol. 95, 1456–1462 (2000).
Tougas, G. et al. Standardization of a simplified scintigraphic methodology for the assessment of gastric emptying in a multicenter setting. Am. J. Gastroenterol. 95, 78–86 (2000).
Sarnelli, G., Caenepeel, P., Geypens, B., Janssens, J. & Tack, J. Symptoms associated with impaired gastric emptying of solids and liquids in functional dyspepsia. Am. J. Gastroenterol. 98, 783–788 (2003).
Karamanolis, G., Caenepeel, P., Arts, J. & Tack, J. Determinants of symptom pattern in idiopathic severely delayed gastric emptying: gastric emptying rate or proximal stomach dysfunction? Gut 56, 29–36 (2007).
Pasricha, P. J. et al. Characteristics of patients with chronic unexplained nausea and vomiting and normal gastric emptying. Clin. Gastroenterol. Hepatol. 9, 567–576.e4 (2011).
Khayyam, U. et al. Assessment of symptoms during gastric emptying scintigraphy to correlate symptoms to delayed gastric emptying. Neurogastroenterol. Motil. 22, 539–545 (2010).
Ardila-Hani, A. et al. Severity of dyspeptic symptoms correlates with delayed and early variables of gastric emptying. Dig. Dis. Sci. 58, 478–487 (2013).
Olausson, E. A. et al. Measurement of gastric emptying by radiopaque markers in patients with diabetes: correlation with scintigraphy and upper gastrointestinal symptoms. Neurogastroenterol. Motil. 25, e224–e232 (2013).
Tseng, P. H. et al. Normal values and symptom correlation of a simplified oatmeal-based gastric emptying study in the Chinese population. J. Gastroenterol. Hepatol. 29, 1873–1882 (2014).
Parkman, H. P. et al. Early satiety and postprandial fullness in gastroparesis correlate with gastroparesis severity, gastric emptying, and water load testing. Neurogastroenterol. Motil. 29, e12981 (2016).
Talley, N. J. et al. Functional dyspepsia, delayed gastric emptying, and impaired quality of life. Gut 55, 933–939 (2006).
Karamanolis, G., Caenepeel, P., Arts, J. & Tack, J. Association of the predominant symptom with clinical characteristics and pathophysiological mechanisms in functional dyspepsia. Gastroenterology 130, 296–303 (2006).
Tack, J., Bisschops, R. & Sarnelli, G. Pathophysiology and treatment of functional dyspepsia. Gastroenterology 127, 1239–1255 (2004).
Janssen, P. et al. The relation between symptom improvement and gastric emptying in the treatment of diabetic and idiopathic gastroparesis. Am. J. Gastroenterol. 108, 1382–1391 (2013).
Kim, D. Y., Myung, S. J. & Camilleri, M. Novel testing of human gastric motor and sensory functions: rationale, methods, and potential applications in clinical practice. Am. J. Gastroenterol. 95, 3365–3373 (2000).
Schwizer, W., Steingoetter, A. & Fox, M. Magnetic resonance imaging for the assessment of gastrointestinal function. Scand. J. Gastroenterol. 41, 1245–1260 (2006).
Tucker, E. et al. Gastric volume responses and emptying after a large liquid nutrient meal in functional dyspepsia and health assessed by non-invasive gastric scintigraphy (GS) and magnetic resonance imaging (MRI): a pilot study to identify candidate biomarkers [abstract]. Gastroenterology 142 (Suppl. 1), 1083 (2012).
Parker, H. L. et al. Clinical assessment of gastric emptying and sensory function utilizing gamma scintigraphy: Establishment of reference intervals for the liquid and solid components of the Nottingham test meal in healthy subjects. Neurogastroenterol. Motil. 29, e13122 (2017).
Frank, J. W., Sarr, M. G. & Camilleri, M. Use of gastroduodenal manometry to differentiate mechanical and functional intestinal obstruction: an analysis of clinical outcome. Am. J. Gastroenterol. 89, 339–344 (1994).
Dinning, P. G., Carrington, E. V. & Scott, S. M. Colonic and anorectal motility testing in the high-resolution era. Curr. Opin. Gastroenterol. 32, 44–48 (2016).
Rao, S. S. Constipation: evaluation and treatment of colonic and anorectal motility disorders. Gastrointest. Endosc. Clin. N. Am. 19, 117–139 (2009).
Kuo, B. et al. Comparison of gastric emptying of a nondigestible capsule to a radio-labelled meal in healthy and gastroparetic subjects. Aliment. Pharmacol. Ther. 27, 186–196 (2008).
Rao, S. S. et al. Investigation of colonic and whole-gut transit with wireless motility capsule and radiopaque markers in constipation. Clin. Gastroenterol. Hepatol. 7, 537–544 (2009).
Simren, M. & Stotzer, P. O. Use and abuse of hydrogen breath tests. Gut 55, 297–303 (2006).
Rezaie, A. et al. Hydrogen and methane-based breath testing in gastrointestinal disorders: the North American Consensus. Am. J. Gastroenterol. 112, 775–784 (2017).
Read, N. W. et al. Interpretation of the breath hydrogen profile obtained after ingesting a solid meal containing unabsorbable carbohydrate. Gut 26, 834–842 (1985).
Yang, J. et al. Prevalence and presentation of lactose intolerance and effects on dairy product intake in healthy subjects and patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 11, 262–268.e1 (2013).
Zhu, Y. et al. Bloating and distention in irritable bowel syndrome: the role of gas production and visceral sensation after lactose ingestion in a population with lactase deficiency. Am. J. Gastroenterol. 108, 1516–1525 (2013).
Yu, D., Cheeseman F. & Vanner, S. Combined oro-caecal scintigraphy & lactulose hydrogen breath testing demonstrate that breath testing detects oro-caecal transit, not small intestinal bacterial overgrowth in patients with IBS. Gut 60, 334–340 (2011).
Lin, E. C. & Massey, B. T. Scintigraphy demonstrates high rate of false-positive results from glucose breath tests for small bowel bacterial overgrowth. Clin. Gastroenterol. Hepatol. 14, 203–208 (2016).
Zhao, J. et al. A study of the methodological and clinical validity of the combined lactulose hydrogen breath test with scintigraphic oro-cecal transit test for diagnosing small intestinal bacterial overgrowth in IBS patients. Neurogastroenterol. Motil. 26, 794–802 (2014).
Fox, M., Thumshirn, M., Fruhauf, H., Fried, M. & Schwizer, W. Determinants of fecal continence in healthy, continent subjects: a comprehensive analysis by anal manometry, rectal barostat and a stool substitute retention test. Digestion 83, 46–53 (2010).
Lunniss, P. J., Gladman, M. A., Hetzer, F. H., Williams, N. S. & Scott, S. M. Risk factors in acquired faecal incontinence. J. R. Soc. Med. 97, 111–116 (2004).
Bharucha, A. E., Zinsmeister, A. R., Schleck, C. D. & Melton, L. J. 3rd. Bowel disturbances are the most important risk factors for late onset fecal incontinence: a population-based case-control study in women. Gastroenterology 139, 1559–1566 (2010).
Enck, P. et al. Epidemiology of faecal incontinence in selected patient groups. Int. J. Colorectal Dis. 6, 143–146 (1991).
Ribas, Y. et al. Initiative to improve detection of faecal incontinence in primary care: The GIFT Project. Fam. Pract. 34, 175–179 (2017).
Nurko, S. & Scott, S. M. Coexistence of constipation and incontinence in children and adults. Best Pract. Res. Clin. Gastroenterol. 25, 29–41 (2011).
Bharucha, A. E. & Rao, S. S. An update on anorectal disorders for gastroenterologists. Gastroenterology 146, 37–45.e2 (2014).
Sauter, M. et al. Toward more accurate measurements of anorectal motor and sensory function in routine clinical practice: validation of high-resolution anorectal manometry and Rapid Barostat Bag measurements of rectal function. Neurogastroenterol. Motil. 26, 685–695 (2014).
Mion, F. et al. 3D high-definition anorectal manometry: values obtained in asymptomatic volunteers, fecal incontinence and chronic constipation. Results of a prospective multicenter study (NOMAD). Neurogastroenterol. Motil. 29, e13049 (2017).
Gladman, M. A., Scott, S. M., Chan, C. L., Williams, N. S. & Lunniss, P. J. Rectal hyposensitivity: prevalence and clinical impact in patients with intractable constipation and fecal incontinence. Dis. Colon Rectum 46, 238–246 (2003).
Chan, C. L., Lunniss, P. J., Wang, D., Williams, N. S. & Scott, S. M. Rectal sensorimotor dysfunction in patients with urge faecal incontinence: evidence from prolonged manometric studies. Gut 54, 1263–1272 (2005).
Chiarioni, G., Bassotti, G., Stanganini, S., Vantini, I. & Whitehead, W. E. Sensory retraining is key to biofeedback therapy for formed stool fecal incontinence. Am. J. Gastroenterol. 97, 109–117 (2002).
Wald, A. Biofeedback therapy for fecal incontinence. Ann. Intern. Med. 95, 146–149 (1981).
Thaha, M. A., Abukar, A. A., Thin, N. N., Ramsanahie, A. & Knowles, C. H. Sacral nerve stimulation for faecal incontinence and constipation in adults. Cochrane Database Syst. Rev. 8, CD004464 (2015).
Altomare, D. F. et al. Long-term outcomes of sacral nerve stimulation for faecal incontinence. Br. J. Surg. 102, 407–415 (2015).
Bharucha, A. E. Difficult defecation: difficult problem assessment and management; what really helps? Gastroenterol. Clin. North Am. 40, 837–844 (2011).
Heinrich, H. et al. Assessment of obstructive defecation by high-resolution anorectal manometry compared with magnetic resonance defecography. Clin. Gastroenterol. Hepatol. 13, 1310–1317.e1 (2015).
Grossi, U. et al. Diagnostic accuracy study of anorectal manometry for diagnosis of dyssynergic defecation. Gut 65, 447–455 (2016).
Chiarioni, G., Whitehead, W. E., Pezza, V., Morelli, A. & Bassotti, G. Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 130, 657–664 (2006).
Ward, B. W., Wu, W. C., Richter, J. E., Hackshaw, B. T. & Castell, D. O. Long-term follow-up of symptomatic status of patients with noncardiac chest pain: is diagnosis of esophageal etiology helpful? Am. J. Gastroenterol. 82, 215–218 (1987).
Horowitz, N. et al. Clinical trial: evaluation of a clinical decision-support model for upper abdominal complaints in primary-care practice. Aliment. Pharmacol. Ther. 26, 1277–1283 (2007).
Heikkinen, M., Pikkarainen, P., Takala, J., Rasanen, H. & Julkunen, R. Etiology of dyspepsia: four hundred unselected consecutive patients in general practice. Scand. J. Gastroenterol. 30, 519–523 (1995).
Acknowledgements
The International Working Group for Disorders of Gastrointestinal Motility and Function initiated the consensus meetings and provided material support for the consensus process. Five separate groups reviewed the current state of the art in clinical measurement of gastrointestinal motility and function: oropharynx, oesophagus, reflux disease, stomach and/or intestine and anorectum. This process was endorsed by the European Society of Neurogastroenterology and Motility (ESNM) and the European Society of Colo-Proctology (ESCP) with representation and support from the American Neurogastroenterology and Motility Society (ANMS), South and Latin American Neurogastroenterology and Motility Society (SLNG), Asian Neurogastroenterology and Motility Association (ANMA) and members of the Australasian Neurogastroenterology and Motility Association (ANGMA). Financial support was provided by the United European Gastroenterology (UEG) Education Committee, registration fees for meetings and sponsorship from all major manufacturers of physiological measurement equipment.
Author information
Authors and Affiliations
Consortia
Contributions
All authors researched data for the article, made substantial contributions to discussion of content and reviewed or edited the manuscript before submission. M.R.F. wrote the article.
Corresponding author
Ethics declarations
Competing interests
M.R.F. has received funding of research and/or support of educational projects from Astra Zeneca, Given Imaging/Medtronic, Mui Scientific, Nestlé, Reckitt Benckiser, and Sandhill Scientific Instruments and Medical Measurement Systems. S.R. has served as a consultant for Medtronic. She received research support from Crospon and Sandhill Scientific. C.P.G. has served as a consultant and speaker for Medtronic and has also received research funding from Medtronic. He has served as a consultant for Ironwood, Quintiles and Torax and as a speaker for Allergan. S.M.S. has received honoraria from Medical Measurement Systems/Laborie for providing training webinars and organizing teaching courses. He has also received research funding from Mui Scientific and Nestec. S.S.R. has served on advisory boards for Forest Laboratories, Intone, Synergy and Vibrant and has received research grants from Forest Laboratories, Intone, Medtronic and Synergy. J.K. has received funds for research from Given Imaging/Medtronic and Standard Instruments. She has acted as a paid consultant and/or has been paid for speaking by Abbott, Allergan, AstraZeneca, Falk Foundation, Mundipharma, Nordmark, Shire and Sucampo. M.C. has received honoraria from Biokier and Kallyope, research support from Astra Zeneca, NGM Pharma and Rhythm Pharmaceuticals, and medication supplies for clinical trials from NovoNordisk. P.J.K. declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
International Working Group for Disorders of Gastrointestinal Motility and Function: https://www.idigest.ch/
Glossary
- Dysphagia
-
Difficulty or discomfort in swallowing as a symptom of disease.
- Achalasia
-
A condition in which the lower oesophageal sphincter muscle fails to relax, preventing food from passing from the oesophagus into the stomach.
- High-resolution manometry
-
(HRM). A diagnostic system that measures intraluminal pressure activity from the throat to the stomach using a series of closely spaced pressure sensors.
- Visceral sensitivity
-
A term used to describe the intensity of sensation (for example, fullness or pain) induced by stimulation applied to the abdominal organs (viscera). Hyposensitivity indicates that stimulation induces less intense sensation than normal; hypersensitivity, indicates that stimulation induces more intense sensation than normal.
- Intraluminal impedance
-
A catheter-based method to detect the presence of food, fluid or gas within the lumen of the oesophagus. Measuring impedance at multiple sites (multichannel) allows for determination of the direction of movement of oesophageal contents. Combined with pH measurement, this technique is considered the gold standard for detection of acid and non-acid reflux events.
- Oesophagogastric junction
-
(EGJ). A complex valve composed of an intrinsic, smooth muscle element (lower oesophageal sphincter and gastric cardia) and an extrinsic, striated muscle element (diaphragm).
- Supragastric belching
-
A condition, thought to be a learned habit, in which air is repetitively sucked into the oesophagus and then immediately expelled (belched).
- Contraction reserve
-
A term used to describe the increase in oesophageal contractility seen in response to physiological challenge (for example, multiple rapid swallows or reflux).
- Aerophagia
-
A condition, thought to be a learned habit, in which excessive air swallowing leads to gastric distension and abdominal bloating.
- Gastrointestinal scintigraphy
-
A test in which a radiolabelled substance (for example, an ‘eggbeater’ meal) is ingested to provide a non-invasive and quantitative measure of gastric emptying and/or orocaecal transit.
- Gastric accommodation
-
A term used to describe the relaxation of the stomach (reduction in gastric tone and increase in compliance) that follows ingestion of a meal.
- Defecography
-
A test in which a series of images are taken as a patient goes through the process of having a bowel movement to assess for the presence of any structural or functional pathology.
- Biofeedback therapy
-
A specialist form of physiotherapy used to treat constipation and faecal incontinence in which sensors record muscle activity (for example, of the abdominal wall and anal sphincter) and give feedback to the patient to improve technique.
- Rectocele
-
A herniation (bulge) of the front (anterior) wall of the rectum into the back (posterior) wall of the vagina. It occurs when the fibrous tissue between the rectum and the vagina (rectovaginal septum) becomes thin and weak over time.
- Intussusception
-
A process in which a segment of intestine, in this case rectum, telescopes (invaginates) into the lower rectum or anal canal, causing structural outlet obstruction and difficulty with defecation.
Rights and permissions
About this article
Cite this article
Fox, M.R., Kahrilas, P.J., Roman, S. et al. Clinical measurement of gastrointestinal motility and function: who, when and which test?. Nat Rev Gastroenterol Hepatol 15, 568–579 (2018). https://doi.org/10.1038/s41575-018-0030-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-018-0030-9
- Springer Nature Limited
This article is cited by
-
Intestinale Transitstörungen
Die Gastroenterologie (2024)
-
Diagnose und Behandlung der funktionellen Dyspepsie in der klinischen Praxis
Schweizer Gastroenterologie (2023)
-
Low-cost gastrointestinal manometry via silicone–liquid-metal pressure transducers resembling a quipu
Nature Biomedical Engineering (2022)
-
The London Classification: Improving Characterization and Classification of Anorectal Function with Anorectal Manometry
Current Gastroenterology Reports (2020)