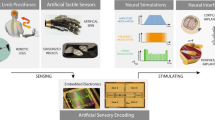
Abstract
By relaying neural signals from the motor cortex to muscles, devices for neurorehabilitation can enhance the movement of limbs in which nerves have been damaged as a consequence of injuries affecting the spinal cord or the lower motor neurons. However, conventional neuroprosthetic devices are rigid and power-hungry. Here we report a stretchable neuromorphic implant that restores coordinated and smooth motions in the legs of mice with neurological motor disorders, enabling the animals to kick a ball, walk or run. The neuromorphic implant acts as an artificial efferent nerve by generating electrophysiological signals from excitatory post-synaptic signals and by providing proprioceptive feedback. The device operates at low power (~1/150 that of a typical microprocessor system), and consists of hydrogel electrodes connected to a stretchable transistor incorporating an organic semiconducting nanowire (acting as an artificial synapse), connected via an ion gel to an artificial proprioceptor incorporating a carbon nanotube strain sensor (acting as an artificial muscle spindle). Stretchable electronics with proprioceptive feedback may inspire the further development of advanced neuromorphic devices for neurorehabilitation.





Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and its Supplementary Information. The raw and analysed datasets generated during the study are available from the corresponding authors on request.
Change history
08 September 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41551-022-00946-7
References
Feigin, V. L. et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18, 459–480 (2019).
Eggers, R. et al. Timed GDNF gene therapy using an immune-evasive gene switch promotes long distance axon regeneration. Brain 142, 295–311 (2019).
Hu, X. et al. Electric conductivity on aligned nanofibers facilitates the transdifferentiation of mesenchymal stem cells into schwann cells and regeneration of injured peripheral nerve. Adv. Healthc. Mater. 9, 1901570 (2020).
Borton, D., Micera, S., Millan, J. D. R. & Courtine, G. Personalized neuroprosthetics. Sci. Transl. Med. 5, 210rv2 (2013).
Capogrosso, M. et al. A brain–spine interface alleviating gait deficits after spinal cord injury in primates. Nature 539, 284–288 (2016).
Ethier, C., Oby, E. R., Bauman, M. J. & Miller, L. E. Restoration of grasp following paralysis through brain-controlled stimulation of muscles. Nature 485, 368–371 (2012).
Song, K.-I. et al. Adaptive self-healing electronic epineurium for chronic bidirectional neural interfaces. Nat. Commun. 11, 4195 (2020).
Wang, J., Wang, H. & Lee, C. Mechanism and applications of electrical stimulation disturbance on motoneuron excitability studied using flexible intramuscular electrode. Adv. Biosyst. 3, 1800281 (2019).
Broderick, B. J., O’Briain, D. E., Breen, P. P., Kearns, S. R. & ÓLaighin, G. A pilot evaluation of a neuromuscular electrical stimulation (NMES) based methodology for the prevention of venous stasis during bed rest. Med. Eng. Phys. 32, 349–355 (2010).
Bijak, M. et al. Stimulation parameter optimization for FES supported standing up and walking in SCI patients. Artif. Organs 29, 220–223 (2005).
Lee, Y. & Lee, T.-W. Organic synapses for neuromorphic electronics: from brain-inspired computing to sensorimotor nervetronics. Acc. Chem. Res. 52, 964–974 (2019).
Park, H.-L. et al. Flexible neuromorphic electronics for computing, soft robotics, and neuroprosthetics. Adv. Mater. 32, 1903558 (2020).
Lee, W. W. et al. A neuro-inspired artificial peripheral nervous system for scalable electronic skins. Sci. Robot. 4, eaax2198 (2019).
Zhang, S. et al. Selective release of different neurotransmitters emulated by a p–i–n junction synaptic transistor for environment‐responsive action control. Adv. Mater. 33, 2007350 (2021).
Park, H. et al. Retina‐inspired carbon nitride‐based photonic synapses for selective detection of UV light. Adv. Mater. 32, 1906899 (2020).
Wan, C. et al. An artificial sensory neuron with tactile perceptual learning. Adv. Mater. 30, 1801291 (2018).
Qian, C. et al. Oxygen-detecting synaptic device for realization of artificial autonomic nervous system for maintaining oxygen homeostasis. Adv. Mater. 32, 2002653 (2020).
Choi, Y., Oh, S., Qian, C., Park, J. H. & Cho, J. H. Vertical organic synapse expandable to 3D crossbar array. Nat. Commun. 11, 4595 (2020).
Zhang, C. et al. Bioinspired artificial sensory nerve based on nafion memristor. Adv. Funct. Mater. 29, 1808783 (2019).
Seo, D.-G. et al. Versatile neuromorphic electronics by modulating synaptic decay of single organic synaptic transistor: from artificial neural networks to neuro-prosthetics. Nano Energy 65, 104035 (2019).
Lee, Y. et al. Stretchable organic optoelectronic sensorimotor synapse. Sci. Adv. 4, eaat7387 (2018).
He, K. et al. An artificial somatic reflex arc. Adv. Mater. 32, 1905399 (2020).
Wei, H. et al. Mimicking efferent nerves using a graphdiyne-based artificial synapse with multiple ion diffusion dynamics. Nat. Commun. 12, 1068 (2021).
Shim, H. et al. Stretchable elastic synaptic transistors for neurologically integrated soft engineering systems. Sci. Adv. 5, eaax4961 (2019).
Kim, S. et al. Artificial stimulus-response system capable of conscious response. Sci. Adv. 7, eabe3996 (2021).
You, I. et al. Artificial multimodal receptors based on ion relaxation dynamics. Science 370, 961–965 (2020).
Park, S. et al. Self-powered ultra-flexible electronics via nano-grating-patterned organic photovoltaics. Nature 561, 516–521 (2018).
Tan, Y. J. et al. A transparent, self-healing and high-κ dielectric for low-field-emission stretchable optoelectronics. Nat. Mater. 19, 182–188 (2020).
Li, P., Anwar Ali, H. P., Cheng, W., Yang, J. & Tee, B. C. K. Bioinspired prosthetic interfaces. Adv. Mater. Technol. 5, 1900856 (2020).
Cai, P. et al. Biomechano-interactive materials and interfaces. Adv. Mater. 30, 1800572 (2018).
Kim, Y. et al. A bioinspired flexible organic artificial afferent nerve. Science 360, 998–1003 (2018).
Xu, W., Min, S.-Y., Hwang, H. & Lee, T.-W. Organic core–sheath nanowire artificial synapses with femtojoule energy consumption. Sci. Adv. 2, e1501326 (2016).
Lee, Y., Park, H.-L., Kim, Y. & Lee, T.-W. Organic electronic synapses with low energy consumption. Joule 5, 1–17 (2021).
You, I., Kong, M. & Jeong, U. Block copolymer elastomers for stretchable electronics. Acc. Chem. Res. 52, 63–72 (2019).
Sekitani, T. et al. Ultraflexible organic amplifier with biocompatible gel electrodes. Nat. Commun. 7, 11425 (2016).
Sim, K. et al. An epicardial bioelectronic patch made from soft rubbery materials and capable of spatiotemporal mapping of electrophysiological activity. Nat. Electron. 3, 775–784 (2020).
Lee, Y. et al. Deformable organic nanowire field-effect transistors. Adv. Mater. 30, 1704401 (2018).
Head, S. I. & Arber, M. B. An active learning mammalian skeletal muscle lab demonstrating contractile and kinetic properties of fast- and slow-twitch muscle. Adv. Physiol. Educ. 37, 405–414 (2013).
Liu, Y. et al. Soft and elastic hydrogel-based microelectronics for localized low-voltage neuromodulation. Nat. Biomed. Eng. 3, 58–68 (2019).
Liu, Y. et al. Morphing electronics enable neuromodulation in growing tissue. Nat. Biotechnol. 38, 1031–1036 (2020).
Formento, E. et al. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat. Neurosci. 21, 1728–1741 (2018).
Liu, Z. et al. Thickness-gradient films for high gauge factor stretchable strain sensors. Adv. Mater. 27, 6230–6237 (2015).
Minnikanti, S. et al. Lifetime assessment of atomic-layer-deposited Al2O3–parylene C bilayer coating for neural interfaces using accelerated age testing and electrochemical characterization. Acta Biomater. 10, 960–967 (2014).
Hukins, D. W. L., Mahomed, A. & Kukureka, S. N. Accelerated aging for testing polymeric biomaterials and medical devices. Med. Eng. Phys. 30, 1270–1274 (2008).
Perich, M. G. et al. Extracellular Neural Recordings from Macaque Primary and Dorsal Premotor Motor Cortex during a Sequential Reaching Task. (CRCNS, 2018); https://doi.org/10.6080/K0FT8J72
Acknowledgements
This work was supported by the Creative-Pioneering Researchers Program through Seoul National University (SNU), the National Research Foundation of Korea (NRF) grant funded by the Korean government (Ministry of Science and ICT) (grant no. NRF-2016R1A3B1908431), and the Pioneer Research Center Program through the National Research Foundation of Korea funded by the Ministry of Science, ICT & Future Planning (grant no. NRF-2022M3C1A3081211). Y. Liu acknowledges National Science Scholarship (NSS) funding support from Agency for Science Technology and Research Singapore (A*STAR).
Author information
Authors and Affiliations
Contributions
Y. Lee, Y. Liu, D.-G.S., Z.B. and T.-W.L. conceived of and designed the overall experiments. Y. Lee, Y. Liu and D.-G.S. conducted experiments and collected related data. J.Y.O. helped to fabricate synaptic transistors. Y.K. contributed to analysis of electrical circuit. J.L. helped with experiments on mice. J. Kang, J. Kim and J.M. contributed to strain sensor fabrication and measurements. A.M.F. aided in image visualization. Y. Lee, Y. Liu, D.-G.S., Z.B. and T.-W.L. analysed all data and co-wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Silvestro Micera, Cunjiang Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Demonstration of practical locomotion of ‘kicking a ball’.
a, Design of the mouse for kicking a ball. An extensor was connected to SNEN system and the swing motion was controlled by synaptic signals. b,c, Photographs of the mouse kicking a ball with a weak and short muscle contraction (a small swing) (b) and a strong and long muscle contraction (a full swing) (c).
Extended Data Fig. 2 Signals of SNEN for bipedal walking locomotion.
a–d, Presynaptic input spike patterns (upper) and resultant EPSCs (lower) with different moving speeds of 0.8 cm/s (slow walking) (a), 1 cm/s (fast walking) (b), 1.6 cm/s (jogging) (c), and 2.5 cm/s (running) (d).
Extended Data Fig. 3 SNEN with electrophysiological signals with different firing rates.
a, b, Presynaptic input spike patterns referred from neural data (a) and resultant EPSCs (b) with high firing rate (34.8 Hz) (neuron 1, red) and low firing rate (2.8 Hz) (neuron 2, black).
Supplementary information
Supplementary Information
Supplementary figures, tables, notes, video captions and references.
Supplementary Video 1
Leg locomotion depending on AP firing frequency from 1 Hz to 11 Hz.
Supplementary Video 2
Synchronized movement of flexion and extension.
Supplementary Video 3
Kicking motions.
Supplementary Video 4
Bipedal walking locomotion.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, Y., Liu, Y., Seo, DG. et al. A low-power stretchable neuromorphic nerve with proprioceptive feedback. Nat. Biomed. Eng 7, 511–519 (2023). https://doi.org/10.1038/s41551-022-00918-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00918-x
- Springer Nature Limited
This article is cited by
-
Strain-insensitive viscoelastic perovskite film for intrinsically stretchable neuromorphic vision-adaptive transistors
Nature Communications (2024)
-
Visualized in-sensor computing
Nature Communications (2024)
-
Neuromorphic electro-stimulation based on atomically thin semiconductor for damage-free inflammation inhibition
Nature Communications (2024)
-
Neuromorphic hardware for somatosensory neuroprostheses
Nature Communications (2024)
-
Wireless facial biosensing system for monitoring facial palsy with flexible microneedle electrode arrays
npj Digital Medicine (2024)