Abstract
Early life stress (ELS) is associated with the later development of schizophrenia. In the rodent model, the maternal separation (MS) stress may induce neuronal apoptosis and schizophrenia-like behavior. Although the TRPV1 agonist capsaicin (CAP) has been reported to reduce apoptosis in the central nervous system, its effect in MS models is unclear. Twenty-four hours of MS of Wistar rat pups on postnatal day (PND9) was used as an ELS. Male rats in the adult stage were the subjects of the study. CAP (1 mg/kg/day) intraperitoneal injection pretreatment was undertaken before behavioral tests for 1 week and continued during the tests. Behavioral tests included open field, novel object recognition, Barnes maze test, and pre-pulse inhibition (PPI) test. MS rats showed behavioral deficits and cognitive impairments mimicking symptoms of schizophrenia compared with controls. MS decreased the expression of TRPV1 in the frontal association cortex (FrA) and in the hippocampal CA1, CA3, and dentate gyrus (DG) regions compared with the control group resulting in the increase of pro-apoptotic proteins (BAX, Caspase3, Cleaved-Caspase3) and the decrease of anti-apoptotic proteins (Bcl-2). The number of NeuN++TUNEL+ cells increased in the MS group in the FrA, CA1, CA3, and DG compared with the control group. Neuronal and behavioral impairments of MS were reversed by treatment with CAP. Exposure to ELS may lead to increased neuronal apoptosis and impaired cognitive function with decreased TRPV1 expression in the prefrontal cortex and hippocampus in adulthood. Sustained low-dose administration of CAP improved neuronal apoptosis and cognitive function. Our results provide evidence for future clinical trials of chili peppers or CAP as dietary supplements for the reversal treatment of schizophrenia.
Similar content being viewed by others
Introduction
Schizophrenia is a severe mental illness with a lifetime prevalence of ~0.7% in the world1. It is a mental disorder characterized by positive symptoms (delusions, hallucinations, thought disorders), negative symptoms (anhedonia, social withdrawal, poverty of thought), and cognitive dysfunction2. Although many researchers have tried to clarify the pathogenesis of schizophrenia, the detailed mechanism is still not fully understood.
Apoptosis is one of the significant mechanisms to be explained in schizophrenia. Risk alleles related to apoptotic genes have been recently associated with schizophrenia3. Magnetic resonance imaging (MRI) studies have given evidence of reduced cortical gray matter volume in prefrontal temporal and parietal cortex areas, which is consistent with the postmortem evidence of reduced cortical neuropil4. Studies of postmortem temporal cortex in schizophrenic patients have found that Bcl-2 was reduced and the BAX/Bcl-2 ratio was increased5,6. Besides, some animal studies have suggested that apoptosis signaling is involved in the cellular pathology of schizophrenia7,8,9. Therefore, neuronal apoptosis is associated with the occurrence of schizophrenia.
Transient receptor potential vanilloid 1 (TRPV1) is a calcium-permeable cation channel belonging to the transient receptor potential (TRP) family of transmembrane proteins10. TRPV1 can be activated by temperature (>43 °C), pH (<6.0), numerous endogenous and exogenous ligands (vanilloids, cannabinoids, pro-inflammatory lipid mediators, plant and animal toxins)11. TRPV1 upregulation and activation mediated by pro-inflammatory mediators have brought some detrimental effects12. Tumor necrosis factor alpha (TNF-α) mediates inflammation in neurons through rapidly increasing sensitization and upregulation of TRPV113. Interleukin-1β (IL-1β) causes anxiety in mice through TRPV114. IL-1β regulates the function and activity of TRPV1 immediately through Interleukin-1 receptor type 1 (IL-1R1) combined with TRPV115. Besides, interleukin-6 (IL-6) has been shown to up-regulate TRPV1 through the activation of the Janus kinase (JAK)/phosphatidylinositol3-kinase (PI3K) signaling pathway16. Since neuroinflammation is one of the pathogenesis of schizophrenia, this may be a possible mechanism for TRPV1 to participate in schizophrenia17. 8-methyl-N-geranyl-6-nonamide (capsaicin (CAP)), a major ingredient in hot peppers of the plant Capsicum genus, is a commonly used pharmacological agonist for TRPV118. Many recent publications have reported the benefits of CAP in cardiovascular disease19, obesity20, cancer21, and the central nervous system (CNS)22. In the CNS, CAP has functions that reduce neuronal apoptosis. For example, CAP therapy alleviates glutamate-induced cortical neuron death in mice23. CAP protects against hypoxia-reoxygenation-induced apoptosis of hippocampal neurons via the PI3K/Akt-mediated signaling pathway24. CAP can alleviate the apoptosis rate in the cell model of Parkinson’s disease25. Therefore, TRPV1 may be involved in neuronal apoptosis, which could be alleviated by CAP therapy.
Early life stress (ELS) has a profound effect on brain development and is associated with schizophrenia26. Maternal separation (MS) is an animal model of early life stress that disrupts normal mother-child interaction and is thought to be a powerful stressor in rodents27. Previous studies have shown that MS can lead to schizophrenia-like behaviors in adult animals such as increased locomotor activity and pre-pulse inhibition (PPI) impairments28. MS can cause a decrease of neurons in male animals during adolescence29. Our previous study has shown that MS decreased the survival and activity of puberty-born neurons in the hippocampus and cognitive function of rats30. In addition, our study also showed that MS increased apoptosis of hippocampal and prefrontal neurons31. Therefore, the MS rat model can be used as a powerful tool to explore the neurobiological basis of schizophrenia.
At present, the effect of MS on TRPV1 expression in rat is unclear. And how CAP reduces the neuronal apoptosis in the prefrontal cortex and hippocampus and improves cognition in MS rats remains to be explored. The aim of this study is exploring the expression of TRPV1 and neuronal apoptosis in the MS model and observe the effect of CAP on the MS model.
Methods
Animals
Twelve male and twelve female 8-week-old Wistar rats were obtained from Beijing Vital Rival Laboratory Animal Technology Co., Ltd. Animals were mated at 3 months of age and the males were removed one week later. Mated female rats were housed individually under 12 h light and 12 h dark cycle (lights on from 8:00 a.m. to 8:00 p.m.) with free access to food and water in temperature and humidity-controlled plastic cages (22 ± 20 °C, 50 ± 10%).
Maternal separation
MS was performed according to previous literature30,32. Females were checked twice a day for delivery (08:00 and 17:00) and the day of delivery was considered postnatal day (PND)0. Each pregnant rat provided an average of 10 ± 2 offspring. On PND9, about 6 litters of rats were randomly selected to MS for 24 hours. In brief, the mothers were removed at 10:00 while the pups remained at room temperature for 24 hours on PND9. On PND10, the mothers were put back in their cages at 10:00. During the period of separation, cages containing the pups were placed on heating pads maintained at 30 °C and were filled with 3 centimeters of bedding. The control group grew up naturally. All the litters were otherwise left undisturbed except for routine cage cleaning. On PND21, MS, and control pups were weaned and then group-housed by sex (3–4 per cage). Previous studies have shown that estrogen plays a key role in regulating neuronal activity and animal behavior, and alterations in estrogen signaling are linked to a range of neurological and psychiatric conditions33,34. One hypothesis suggests that sex differences in learning and memory may depend on gonadal hormone activity during early brain development35. Therefore, all subsequent experiments were carried out only on male offspring36.
All procedures involving animals were approved and performed according to the Institutional Animals Care Committee of Renmin Hospital of Wuhan University guidelines.
Experimental design
Experiment 1: the effects of MS on behavior, neuronal apoptosis, TRPV1 expression in adult rats.
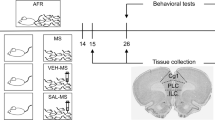
On PND9, mothers and their pups were randomly divided into control group and MS group. Control rats did not receive any treatment (normal controls), while MS rats were exposed to MS as described above. In the adult stage (PND63-70), 12 pups were randomly selected from each group and behavioral tests were conducted. After behavioral tests, animals were killed on PND70. The experimental process is shown in Fig. 1A. The remaining pups were used in experiment 2.
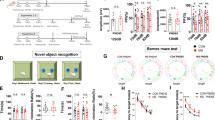
A Experimental protocol. Litters were subjected to MS during lactation on PND9. Behavioral tests were performed during PND63-70. Rats received capsaicin injections intraperitoneally from PND56 to PND70. PND, postnatal day; MS, maternal separation. B Total distance moved in the open field in 10 minutes. C On day 2, time was spent exploring the two identical sample objects within the 10-min period. On day 3, time was spent exploring novel and old objects during the 10-min period. D The latency to the target hole in the Barnes maze test. E The baseline startle response to an auditory-evoked startle stimulus of 120 dB is shown in the left panel. Percentage PPI of the auditory startle reflex across different pre-pulse intensities in the right panel. CON, n = 12; MS, n = 12. n.s. not significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001 as compared with controls. The data are represented as mean ± SEM.
Experiment 2: the effects of administering CAP on behaviors, neuronal apoptosis, TRPV1 expression in the MS rats.
On PND56, each offspring group was randomly divided into two subgroups (10-12 neonates per group). From PND56 to PND70, rats were treated with either saline or CAP, and the resulting four groups were: control and vehicle group (CON + VEH), MS and vehicle group (MS + VEH), control and CAP group (CON + CAP), and MS and CAP group (MS + CAP). For the CON + VEH and MS + VEH groups, rats received vehicle (1 ml/kg/day, intraperitoneal injection) treatment, which is a 1:1:8 mixture of Tween 80: ethanol: saline. Rats in CON + CAP and MS + CAP group were subjected to CAP (1 mg/kg, intraperitoneal injection; a single injection/day) treatment for 1 week before behavioral tests and continued to be administered during the behavioral test. CAP was purchased from Sigma-Aldrich dissolved in a 1:1:8 mixture of Tween 80: ethanol: saline mixture. The experimental flowchart is shown in Fig. 1A.
Behavioral testing of offspring
Experimental rats underwent behavioral experiments during the adult stage (PND63-70). The open field test was completed on the first day (PND63). The novel object recognition test was completed between PND63 to PND65. The Barnes maze test was completed between PND65 to PND69. On the last day (PND70), a PPI test was completed. Each rat was assessed for each behavioral measure. After each trial, the apparatus was cleaned with 75% alcohol.
Open field test
Spontaneous motor activity was studied in a field experiment (OPF). The instrument consists of a 100 × 100 × 40 cm black plastic box with an open top. The experiment was conducted in a room with low sound and dim light (40 W, red light). A camera is fixed above the instruments and connected to monitors and video-tracking motion analysis systems (EthoVision; Noldus Information Technology in the Netherlands). Each rat was placed in the center of the open field for 10 minutes. During the observation period, the system recorded the motor activity of the rats and allowed automatic calculation of the distance the rats moved.
Novel object recognition test
The new object recognition test was conducted using the open-field device described above and consists of three parts: habituation, training, and testing. Each rat was placed in the center of the open field to adapt to the equipment for 10 minutes. In the training experiment, the rats explored two identical objects for 10 minutes. The test experiment began 24 hours later. Rats were allowed to explore a familiar and a novel object (replacing one of the training objects) for 10 minutes. The movement was recorded by video and analyzed by EthoVision. The formula for calculating the preference for exploring novel objects is: [(time spent in the novel subject) / (total time spent in two subjects) × 100%].
Barnes maze test
The Barnes Maze test was used to estimate spatial memory. The maze rises 140 cm above the ground, and consists of 20 round holes, each 10 cm in diameter, evenly distributed around the perimeter. The target hole is connected to a dark chamber that allows the rats to hide from bright light exposure. The day before the formal experiment, the rats were adapted to the target box for four minutes. On day 1, each rat was placed in the maze’s center black cube for 5 seconds and then explored the maze to find the target hole after the cube was removed. Once the rat entered the target hole, it was left there for 30 seconds; if it failed to find the target hole within three minutes, it was taken to the target hole and allowed to stay there for one minute. The time to the target hole was recorded. Each animal was tested twice during the day with an interval of four hours. The experiment was repeated for four days. We averaged the results of two replicates as the result for each rat on that day. The whole process was tracked by cameras.
PPI testing
The PPI test was performed in an anechoic chamber fitted with a smaller plexiglass cage mounted on a gravity sensor platform to digitize the pressure generated by startled rats (AniLab Scientific Instruments Co., Ltd., China. www.anilab.cn.). The white noise was set at 70 dB. After 5 minutes of acclimation, 5 startle stimuli were applied (120 dB startle for 20 ms). In the testing phase, an initial delay of 50 ms was followed by a 20 ms impulse stimulus (75, 78, or 82 dB) and a 40 ms startle stimulus of 120 dB after a 100 ms delay, randomly applied by software control for about 40 trials (Supplementary 1). The interstimulus interval ranged from 10 to 30 s. The results of PPI75, PPI78, and PPI82 are calculated automatically by the system software. The percentage of PPI induced by each pre-pulse intensity was calculated as [1-(startle amplitude on pre-pulse trial)/(startle amplitude on pulse alone)]×100%.
Sample collection
After completing behavioral tests on PND70, sodium thiopental (50 mg/kg, i.p.) was used to kill animals. Immediately after removal from the body, the prefrontal cortex and hippocampus tissues were frozen in dry ice and transferred to -80 °C for storage until further protein expression analysis was required. 3 rats in each group were injected with 4% paraformaldehyde and their brains were taken for immunofluorescence analysis or TUNEL assay. Rats were perfused through the heart initially with PBS and then with 4% paraformaldehyde. The brains were then fixed overnight in 4% paraformaldehyde, embedded in paraffin, and cut at 40 μm thickness.
Protein extraction and western blotting analysis
Total protein was extracted from hippocampal and prefrontal tissues with RIPA buffer (Beyotime Biotech, China). Protein concentration was determined with BCA kit (P0010S, Beyotime Biotech). Identical amounts of RIPA-extracted protein were loaded and separated with 8% SDS polyacrylamide gel and transferred onto polyvinylidene fluoride (PVDF) membranes. Western blot analysis used the following primary antibodies: Mouse anti-TRPV1 (dilution 1:800, ab203103, Abcam, Cambridge, UK), rabbit anti- BAX (dilution 1:1000, GB11690, Servicebio, China), rabbit anti-Bcl-2(dilution 1:1000, abs147821, Absin, China), rabbit anti-Caspase3(dilution 1:2000, abs124299, Absin, China), rabbit anti-Cleaved-Caspase3(dilution 1:2000, abs132005, Absin, China), and rabbit anti-β-actin (dilution 1:1000, ab181602, Abcam, UK). The following secondary horseradish peroxidase (HRP)-conjugated antibodies were used at 1:5000 dilution: goat anti-rabbit HRP (12–348, Millipore, USA) and goat anti-mouse HRP (sc-2005, Santa Cruz Biotechnology, USA).
Immunofluorescent staining assay
Rats were anesthetized and perfused through the heart initially with PBS and then with 4% paraformaldehyde. The brains were then fixed overnight in 4% paraformaldehyde, embedded in paraffin, and cut at 40μm thickness. The paraffin-embedded tissue sections were dewaxed in glycol, rehydrated, placed in sodium citrate, microwaved for antigen retrieval, washed with PBS, and then blocked in 1% BSA (Roche, Basel, Switzerland) at room temperature for 2 h. Sections were incubated with primary antibody mouse anti-TRPV1 (dilution 1:500, ab203103, Abcam, Cambridge, UK) and rabbit anti-NeuN (dilution 1:250, GB11138, Servicebio, China) overnight at 4 °C. The secondary antibody (Cy3 conjugated donkey Anti-Mouse IgG (H + L), dilution 1:200, GB21401, Servicebio, China; Alexa Fluor 488-conjugated goat Anti-Mouse IgG (H + L) dilution 1:200, GB21401, Servicebio, China) was then incubated at 37 degrees for 1 hour. DAPI (Thermo Fisher Scientific, Waltham, USA) was used as a nuclear stain. Observe and photograph with a fluorescence microscope (IX53, Olympus, Tokyo, Japan).
TUNEL assay
Terminal-deoxynucleotidyl transferase-mediated nick end labeling (TUNEL) can be used to detect apoptotic cells. In late apoptotic cells, the sticky 3’-OH terminal is generated by the break of DNA. Under the catalysis of deoxyribonucleotide terminal transferase (TdT), dUTP with fluorescein molecule is labeled to the 3’ -terminal of DNA, and then observed by fluorescence microscope.
Paraffin-embedded tissue cross-sections were dewaxed in xylol, rehydrated, placed in sodium citrate, microwaved for antigen retrieval, and then washed with PBS and blocked with 1% BSA (Roche, Basel, Switzerland) in PBS for 2 h at room temperature. Sections were incubated at 4 °C overnight with primary antibodies targeting mouse NeuN (1:100, ab279296, Abcam, Cambridge UK), followed by incubation with Fluorescein’s TUNEL Cell Apoptosis Detection Kit (G1501-50T, Servicebio, China). Completed the experiment according to the instructions. Paraffin sections routinely deparaffinized, dehydrated with gradient ethanol, digested with proteinase K at 37 °C for 20 min, and incubated with equilibration buffer at room temperature for 10 minutes, then incubated with TUNEL mixture (TdT enzyme: FITC-12-dUTP Labeling Mix: Equilibration Buffer = 1 µL: 5 µL: 50 µL for each sample) away from light at 37 °C for 1 h. After DAPI staining the nuclear, the slides were photographed by a fluorescence microscope and analyzed for positive cells.
Data analysis and statistics
The results are presented as the mean ± SEM. Experiment 1 was analyzed using an independent sample t-test. In Experiment 2, the data were analyzed by analysis of variance (ANOVA) to determine group differences. P values less than 0.05 (two-sided) were considered significant. All analyses were performed with GraphPad Prism software, version 7.0, or SPSS software, version 20.0.
Results
Effects of MS on behavior, neuron apoptosis, and trpv1 expression in rats
MS rats exhibit behavioral impairment in adulthood
To investigate the effects of MS on rat behavior, several behavioral tests were performed on PND70. We first evaluated the effects of MS on the locomotor activity of the rats. The open field test demonstrated that MS-treated rats had significantly increased total distances moved when compared with control rats, suggesting that the MS rats were hyperactive (CON: 5889 ± 347.2, n = 12; MS:8398 ± 399.0, n = 12; P < 0.0001; Fig. 1B). During the novel object recognition test, MS and control rats spent similar amounts of time exploring two identical objects during the habituation phase (Fig. 1C left panel). However, when the object2 was replaced 24 hours later, the MS group spent relatively little time exploring the new object (CON: 40.20% ± 5.397%, n = 12; MS: 12.59% ± 6.749%, n = 12; P = 0.0234; Fig. 1C right panel). In the Barnes maze test, from day 1 to day 4, MS rats took longer to find the target hole than controls. The results showed that MS rats developed deficits in spatial learning and memory (DAY 1: 117.9 ± 10.59 for CON n = 12, 158.0 ± 6.260 for MS n = 12, P = 0.0036; DAY 2: 52.28 ± 6.367 for CON n = 12, 112.3 ± 8.917 for MS n = 12, P = 0.0001; DAY 3: 31.34 ± 5.913 for CON n = 12,78.51 ± 14.97 for MS n = 12, P = 0.0173; DAY 4: 14.51 ± 5.509 for CON n = 12, 47.99 ± 9.378 for MS n = 12, P = 0.0116; Fig. 1D). In the PPI test, there was no difference in baseline response between the two groups for the 120 dB startle stimulus (Fig. 1E, left panel). However, MS rats showed impaired PPI at varying pre-pulse intensities, indicating reduced inhibition of starling stimuli compared to control rats (75 dB: 62.28 ± 7.602 for CON n = 12, 39.90 ± 6.258 for MS n = 12, P = 0.0355; 78 dB: 72.57 ± 6.954 for CON n = 12, 42.77 ± 5.279 for MS n = 12, P = 0.0068; 82 dB: 79.97 ± 4.689 for CON n = 12, 52.82 ± 5.185 for MS n = 12, P = 0.0021 Fig. 1E, right panel).
Depletion of TRPV1 in the prefrontal cortex and hippocampus of MS rats
We then measured TRPV1 concentrations in adult rat brains. Western blotting results showed that TRPV1 expression levels decreased significantly in the prefrontal cortex (MS: 0.5175 ± 0.0238 relative to CON, P = 0.0031, n = 6, Fig. 2A) and hippocampus (MS: 0.6250 ± 0.048 relative to CON, P = 0.0014, n = 6, Fig. 2B) in the MS group compared with the control.
A, B The protein expression of TRPV1 was measured by Western blot in the prefrontal cortex and hippocampus (n = 6 per group). C, L Representative immunofluorescence images show the expression of TRPV1 (red pixels), NeuN (green pixels), and DAPI (blue pixels) in the frontal association cortex (FrA) and in the hippocampal CA1, CA3, and dentate gyrus (DG) regions (n = 3 per group). D–K Quantitative analyses of the percentage of NeuN and TRPV1 co-labeling. Quantitative analysis of results and data are represented as mean ± SEM; *P < 0.05; **P < 0.01.
Next, a double-label immunofluorescence assay was used to detect NeuN and TRPV1 expression. As shown in Fig. 2D, F, H the number of TRPV1 positive cells (MS: 44.00% ± 5.033% relative to CON, p = 0.0079, n = 3, frontal association cortex (FrA); MS: 65.67% ±10.59% relative to CON, P = 0.0407, n = 3, dentate gyrus (DG); MS: 49.00% ±8.505% relative to CON, P = 0.0062, n = 3, CA1; MS: 35.67% ±5.897% relative to CON, P = 0.0022, n = 3, CA3) and the number of NeuN/TRPV1 co-labeled positive cells (MS: 41.00% ± 3.512% relative to CON, p = 0.0054, n = 3, FrA; MS: 67.33% ±9.684% relative to CON, P = 0.0407, n = 3, DG; MS: 51.33% ± 4.333% relative to CON, P = 0.0023, n = 3, CA1; MS: 36.67% ±8.819% relative to CON, P = 0.0061, n = 3, CA3) in the MS group decreased significantly compared with the control group.
Increased apoptosis in the prefrontal cortex and hippocampus in MS rats
We examined the level of apoptosis in rat brains. The expression of important apoptotic indicators (Bcl-2, BAX, Caspase-3, Cleaved Caspase-3) in the prefrontal cortex and hippocampus was detected. Pro-apoptotic protein expressions of BAX, Caspase3 and Cleaved-Caspase3 in prefrontal cortex and hippocampus were significantly increased in MS rats compared with control group (Fig. 3A, B) (prefrontal cortex: MS: 1.560 ± 0.1879 relative to CON, P = 0.0287, n = 6 BAX; MS: 1.859 ± 0.2128 relative to CON, P = 0.0061, n = 6 Caspase-3; MS: 2.088 ± 0.2280 relative to CON, P = 0.0023, n = 6 Cleaved Caspase-3) (hippocampus: MS: 1.510 ± 0.1498 relative to CON, P = 0.0129, n = 6 BAX; MS: 1.334 ± 0.1004 relative to CON, P = 0.0447, n = 6 Caspase-3; MS: 1.586 ± 0.06283 relative to CON, P = 0.0007, n = 6 Cleaved Caspase-3). However, anti-apoptotic protein expression of bcl-2 (prefrontal cortex: MS: 0.5175 ± 0.0238 relative to CON, P = 0.0031, n = 6) (hippocampus: MS: 0.7572 ± 0.0582 relative to CON, P = 0.0191, n = 6) was decreased dramatically in MS rats.
A, B The protein expression of BAX, Bcl2, Caspase-3, and Cleaved Caspase-3 were measured by western blot in the prefrontal cortex and hippocampus (n = 6 per group). C, D Neuronal apoptosis was assessed by TUNEL staining in the frontal association cortex (FrA) and in the hippocampal CA1, CA3, and dentate gyrus (DG) regions. TUNEL positive cells (red), NeuN (green), and DAPI (blue) labeling (n = 3 per group). E–H Quantification of TUNEL‐positive cells in the FrA, CA1, CA3, and DG regions. Original magnification ×400. Data are presented as mean ± SEM for each group. P values were calculated using Student’s t test. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
In addition, TUNEL staining was used to detect neuronal apoptosis. In PND70, compared with control group, the number of NeuN++TUNEL+/NeuN+ cells in the MS group increased significantly in the FrA, CA1, CA3, and DG regions (Fig. 3C–H) (FrA: MS: 3.655 ± 0.6923 relative to CON, P = 0.0212, n = 3; DG: MS: 2.783 ± 0.3322 relative to CON, P = 0.0071, n = 3; CA1: MS: 6.540 ± 1.022 relative to CON, P = 0.0056, n = 3; CA3: MS: 7.569 ± 0.3340 relative to CON, P < 0.0001, n = 3).
The effects of CAP administration on behaviors, neuronal apoptosis, and TRPV1 expression in the prefrontal cortex and hippocampus of MS rats
CAP administration improved the behavioral performances of MS rats
Behavioral tests showed that CAP 1 mg/kg/day administration improved behavior and cognition. In open field experiment, the total moved distance of MS + CAP group was lower than that of MS + VEH group (MS + VEH: 8587 ± 105.9, n = 10; MS + CAP: 4236 ± 91.12 n = 12; P = 0.0022, Fig. 4A). In the novel object recognition test, the MS + CAP group spent more time sniffing for the new object than MS + VEH group (MS + VEH: 0.1032 ± 0.03256, n = 10; MS + CAP: 0.2939 ± 0.02091 n = 12; P = 0.0023 Fig. 4B right panel). In addition, in the Barnes maze test, the MS + CAP group had decreased latency entering the target hole than the MS + VEH group (DAY 1: 158.6 ± 4.9 for MS + VEH n = 10, 129.6 ± 14.2 for MS + CAP n = 12, P = 0.2681; DAY 2: 116.7 ± 6.67 for MS + VEH n = 10, 87.93 ± 5.15 for MS + CAP n = 12, P = 0.0464; DAY 3: 75.3 ± 6.17 for MS + VEH n = 10, 42.8 ± 4.312 for MS + CAP n = 12, P = 0.0330; DAY 4: 30.5 ± 6.69 for MS + VEH n = 10, 17.1 ± 2.689 for MS + CAP n = 12, P = 0.1030; Fig. 4C). In the PPI test, there was no difference in baseline response among the four groups for the 120 dB startle stimulus (Fig. 4D, left panel). However, CAP treatment partially alleviated MS induced impaired PPI response of 75 dB, 78 dB, and 82 dB (75 dB: 39.58 ± 4.262 for MS + VEH n = 10, 60.71 ± 5.825 for MS + CAP n = 12, P = 0.0417; 78 dB: 47.24 ± 3.439 for MS + VEH n = 10, 64.77 ± 2.593 for MS + CAP n = 12, P = 0.0292; 82 dB: 55.84 ± 2.095 for MS + VEH n = 10, 72.56 ± 5.335 for MS + CAP n = 12, P = 0.0455) (Fig. 4D, right panel).
A Total distance moved in the open field in 10 minutes. B Effects of capsaicin administration on the novel object recognition assay in different groups. C Effects of capsaicin administration on the latency to the target hole in the Barnes maze test. D The baseline startle response to an auditory-evoked startle stimulus of 120 dB is shown in the left panel. Effects of capsaicin administration (1 mg/kg/day) on PPI at different pre-pulse intensities in the right panel. CON + VEH, n = 10; MS + VEH n = 10, CON + CAP n = 11, MS + CAP n = 12 n.s. not significant; *p < 0.05; **p < 0.0; ***p < 0.001; ****p < 0.0001 as compared with controls. The data are represented as mean ± SEM.
Side effects of CAP as pain, spasticity, and crouching were described in Supplementary 2.
CAP administration reversed the effects of MS on apoptosis indices but not TRPV1 expression
The pro-apoptotic protein expression of BAX, Caspase3 and Cleaved-Caspase3 in the prefrontal cortex and hippocampus were increased dramatically in MS + VEH compared with those in the CON + VEH group, whereas was inhibited in the MS + CAP group (prefrontal cortex: 2.433 ± 0.2963 for MS + VEH n = 3, 1.233 ± 0.1453 for MS + CAP n = 3, P = 0.0086, BAX; 1.933 ± 0.1202 for MS + VEH n = 3, 1.067 ± 0.08819 for MS + CAP n = 3, P = 0.0044, Caspase-3; 2.433 ± 0.3480 for MS + VEH n = 3, 1.367 ± 0.2028 for MS + CAP n = 3, P = 0.0425, Cleaved Caspase-3) (hippocampus: 2.300 ± 0.2082 for MS + VEH n = 3, 1.200 ± 0.05774 for MS + CAP n = 3, P = 0.0070, BAX; 2.133 ± 0.2028 for MS + VEH n = 3, 0.9333 ± 0.08819 for MS + CAP n = 3, P = 0.0025, Caspase-3; 2.433 ± 0.2963 for MS + VEH n = 3, 1.333 ± 0.08819 for MS + CAP n = 3, P = 0.0047, Cleaved Caspase-3). In addition, anti-apoptotic protein Bcl-2 expression was significantly decreased in the prefrontal cortex and hippocampus in MS + VEH treated rats, while CAP pretreatment significantly reduced these increases (prefrontal cortex: 0.3167 ± 0.1452 for MS + VEH n = 3, 1.500 ± 0.1732 for MS + CAP n = 3, P = 0.0020) (hippocampus: 0.2733 ± 0.1157 for MS + VEH n = 3, 1.417 ± 0.3420 for MS + CAP n = 3, P = 0.0273) (Fig. 5A–J). Besides, CAP administration in the MS + CAP group decreased neuronal apoptosis in the FrA, CA1, CA3, and DG regions as assessed by TUNEL staining, compared with the MS + VEH group (FrA: 3.185 ± 0.2639 for MS + VEH n = 3, 0.6601 ± 0.1614 for MS + CAP n = 3, P = 0.0006; DG: 8.677 ± 1.216 for MS + VEH n = 3, 1.539 ± 0.4332 for MS + CAP n = 3, P = 0.0003; CA1: 11.01 ± 1.542 for MS + VEH n = 3, 2.206 ± 0.3541 for MS + CAP n = 3, P = 0.0002; CA3: 13.26 ± 1.256 for MS + VEH n = 3, 3.154 ± 0.3346 for MS + CAP n = 3, P < 0.0001) (Fig. 5K–P).
A–J The protein expression of BAX, Bcl2, Caspase-3, and Cleaved Caspase-3 were measured by Western blot in the prefrontal cortex and hippocampus (n = 3 per group). K–P Neuronal apoptosis was assessed by TUNEL staining in the frontal association cortex (FrA) and in the hippocampal CA1, CA3, and dentate gyrus (DG) regions. TUNEL positive cells (red), NeuN (green), and DAPI (blue) labeling (n = 3 per group). Data are presented as mean ± SEM for each group. n.s. not significant; *p < 0.05; **p < 0.01;***p < 0.001; ****p < 0.0001 as compared with controls.
In addition, CAP administration did not increase TRPV1 expression in the MS + CAP group compared to the MS + VEH group, according to the western blotting and double-label immunofluorescence assay (Fig. 6).
A, B The protein expression of TRPV1 was measured by Western blot in the prefrontal cortex and hippocampus (n = 3 per group). C, L Representative immunofluorescence images show the expression of TRPV1 (red pixels), NeuN (green pixels), and DAPI (blue pixels) in the frontal association cortex (FrA) and in the hippocampal CA1, CA3, and dentate gyrus (DG) regions (n = 3 per group). D–K Quantitative analyses of the percentage of NeuN and TRPV1 co-labeling. Quantitative analysis of results and data are represented as mean ± SEM, n = 3 for each group; *P < 0.05; **P < 0.01.
Discussion
In this study, we demonstrated that a single 24-hour MS in early life resulted in increased neuronal apoptosis and impaired cognitive function with decreased TRPV1 expression in the prefrontal cortex and hippocampus. And administration of the TRPV1 agonist CAP, an active component of hot peppers, significantly improved neuronal apoptosis, behavioral and cognitive function impaired by MS (Fig. 7).
The duration of separation from the mother during the first 2 years of life predicted elevated schizotypal personality disorder symptoms, which provided support for the role of early childhood psychosocial risk factors in the subsequent development of schizophrenia spectrum symptoms in emotionally vulnerable children37. In the study by Ellenbroek et al., the disturbance of PPI was strongest when MS was performed on PND9 compared to MS on PND3 and PND638. Moreover, impairment in long-term potentiation (LTP) was evident only in adolescent animals exposed to MS on PND9, but not on PND4 and PND1839. Therefore, we studied in PND9 MS male rats.
Both the hippocampus and the interconnected prefrontal regions are targets that seem sensitive to early stress-induced changes40,41, since they mediate neuroendocrine stress responses through neuronal connections to the hypothalamic-pituitary-adrenal (HPA) axis. One hypothesis suggested that sex differences in learning and memory may depend on gonadal hormone activity during early brain development35. Therefore, our study focused on the prefrontal cortex and hippocampus.
There is increasing evidence that apoptosis is involved in the pathological process of schizophrenia. Many clinical studies have investigated the level of apoptosis in patients with schizophrenia3,5,6,42. In addition, some second-generation antipsychotics, such as risperidone paliperidone, olanzapine, and clozapine have anti-apoptotic effects3. It has been reported that early MS can lead to neuronal apoptosis43,44. Apoptotic cell death is characterized by cell shrinkage, chromatin condensation, membrane blebbing, and DNA fragmentation. Consisted with previous research, our study showed increased apoptosis in the prefrontal cortex and hippocampus in MS rats through TUNEL staining. The relative expression of Bcl-2 and BAX genes regulates apoptosis. The expression of Caspase3 and Caspase 9 genes may activate the cell death mechanism through endogenous apoptotic genes, and the expression of them in schizophrenia patients is higher than that in healthy controls45. In general, increased expression of BAX and decreased expression of Bcl-2 suggested apoptosis. Our study showed that MS increased BAX, Caspase3, Cleaved-Caspase3 and decreased Bcl-2. However, the specific mechanism of inducing neuronal apoptosis in schizophrenia is not fully understood.
In recent years, much literature has provided evidence that TRPV1 may play an important role in the development of schizophrenia. From a clinical perspective, pain perception deficits in schizophrenia may be related to TRPV1 changes in primary sensory neurons46. One of the mechanisms of cannabidiol is the activation of TRPV1. Cannabidiol can aggravate positive symptoms in patients with schizophrenia47. In addition, some animal studies have explored the association between TRPV1 and schizophrenia-like behavior. However, the results are not entirely consistent. Some studies have shown that TRPV1 induces or exacerbates schizophrenia-like behaviors. For example, three different indirect endocannabinoid agonists, AM404, VDM11, and AA5HT, respectively attenuated hypermotility in dopamine transporter (DAT) knockout mice, and this effect was reversed by the TRPV1 antagonist capsaepine48. Administration of TRPV1 agonist CAP (50 mg/kg s.c.) on day 2 of life resulted in hyperactive schizophrenia-like behavior at weeks 5–7, but increased PPI at weeks 8-1249. However, other studies suggested that TRPV1 activation reduces schizophrenia-like behaviors. The study conducted by Leonora E Long et al. has shown that MK-801 (0.3–1 mg/kg i.p.) dose-dependent reduced PPI inhibition and cannabidiol (5 mg/kg i.p.) successfully reversed the impairment in PPI inhibition induced by MK-801 (1 mg/kg i.p.)50. Results on the effects of cannabinoids on PPI in rodents indicate the involvement of TRPV1 in sensorimotor gated function and pathophysiology in schizophrenia51. The spontaneously hypertensive rats (SHR) showed schizophrenia-like behavior with hyperactivity and decreased sociability. TRPV1 agonist CAP (2.5 mg/kg) treatment reduced hyperactivity of SHR51,52.
TRPV1 is widely expressed in the nervous systems. Several studies have reported the expression of TRPV1 in neurons, astrocytes, and microglia53. However, its physiological roles remain controversial. TRPV1 activation in neurons increases ROS expression54, mitochondrial disruption, cell death55, mediates long-term depression56. However, TRPV1 activation in neurons has also been reported to protect neurons via down-regulating N-methyl-D-aspartic acid (NMDA) receptors23. TRPV1 activation in astrocytes secrets glutamate57, prolongs post synaptic excitatory58, mediates ER stress59. TRPV1 activation in microglia increases ROS60, promotes phagocytosis61, mitochondrial disruption, cytochrome C release62. Besides, TRPV1 activation in microglia also inhibits oxidative stress63, regulates microglial/macrophage M1/M2 polarization64, and alleviates neuroinflammation. It appears that TRPV1 activation could play a therapeutic role by stabilizing glial cells in some disease models. However, it can also cause damage under physiological conditions. This may be related to the disease model and the dose and type of agonist.
Previous studies have reported that CAP, as an activator of TRPV1, improved cognition and behavior. CAP treatment daily significantly can improve spatial learning, memory and synaptic function in Aβ42-treated Alzheimer’s disease mice65. CAP reduced the brain’s Aβ content and reversed cognitive decline in Alzheimer’s model mice66. Furthermore, a diet rich in CAP may have beneficial effects on AD cognitive function in middle-aged and elderly adults67. TRPV1 activation by CAP enhances NMDAR-dependent CA1 LTP and ameliorates stress-induced memory decline, which can be blocked by TRPV1 antagonists capsazepine and SB36679168. Consistent with previous results, our study also showed that the use of TRPV1 agonists improved cognition and other schizophrenia-like behavior. However, other studies have come to different conclusions. Repeated oral administration of CAP increases anxiety-like behaviors with prolonged stress response in rats69. Epileptic activity was increased in hippocampal slices of rats by CAP administration70. Therefore, we consider that the different effects caused by CAP may be related to the use of different disease models. The dose was given in reference to Jeong Yeob Baek’s study71.
Some studies have reported that CAP plays a protective role in the central nervous system. For example, in a rat model of the middle cerebral artery occlusion/reperfusion, peri-infarct injection of CAP (1 or 3 nmol) reduced infarct volume and improved neurological function23. Intraperitoneal CAP can reduce neurodegeneration in the substantia nigra in the lipopolysaccharide (LPS)-lesioned inflammatory rat model of Parkinson’s disease72. Our study found that CAP (1 mg/kg, intraperitoneal injection for 1 week), as an activator of TRPV1, could decrease the apoptosis of neuron, inhibit the expression of BAX, Caspase3, and Cleaved-Caspase3 and increase the expression of anti-apoptotic protein Bcl-2 in the hippocampus and prefrontal cortex of MS rats, which might demonstrate the anti-apoptotic effect of CAP in our model of MS. Furthermore, CAP 1 mg/kg/day intraperitoneal injection mediated paroxysm, spasticity and couching behavior last for 1–2 min only at 1st day, and did not cause death in rats, which is considered as a small and safe dose71.
Our study suggested that CAP administration might improve cognitive function and behavior by inhibiting neuronal apoptosis at least in MS models. However, this study still has some limitations. Because only total cell extract levels were measured, it was not possible to infer how much TRPV1 was indeed expressed and fully functional in the membrane. In addition, we only studied the expression of TRPV1 in neurons, the expression of TRPV1 in microglia and astrocytes still needs to be studied in the future. Because TRPV1 expression was decreased in MS group compared to control, we used TRPV1 agonists CAP, but not detect the activation of TRPV1, although many research have reported TRPV1 was activated by CAP73,74. Furthermore, we only studied male rats in order to eliminate the additional effects of estrogen, future work needs to explore the effects in females and possible sex differences.
Conclusions
This study suggested that exposure to MS may lead to increased neuronal apoptosis and impaired cognitive function with decreased TRPV1 expression in the prefrontal cortex and hippocampus in adulthood. Sustained low-dose administration of CAP improved neuronal apoptosis and cognitive function. CAP has potential advantages as an intervention strategy for schizophrenia as a natural component of spicy foods. Our study provided new information on the etiology of schizophrenia. Our results provided evidence for future clinical trials of chili peppers or CAP as dietary supplements for the therapy of schizophrenia.
Availability of data and material
Data will be made available on request.
References
Mcgrath, J., Saha, S., Chant, D. & Welham, J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol. Rev. 30, 67–76 (2008).
Powell, S. B. & Geyer, M. A. Overview of animal models of schizophrenia. Curr. Protoc. Neurosci. Chapter 9, 9–24 (2007).
Morén, C. et al. Systematic review of the therapeutic role of apoptotic inhibitors in neurodegeneration and their potential use in schizophrenia. Antioxidants 11, 2275 (2022).
Glantz, L. A., Gilmore, J. H., Lieberman, J. A. & Jarskog, L. F. Apoptotic mechanisms and the synaptic pathology of schizophrenia. Schizophr. Res. 81, 47–63 (2006).
Jarskog, L. F., Gilmore, J. H., Selinger, E. S. & Lieberman, J. A. Cortical bcl-2 protein expression and apoptotic regulation in schizophrenia. Biol. Psychiatry 48, 641–650 (2000).
Jarskog, L. F., Selinger, E. S., Lieberman, J. A. & Gilmore, J. H. Apoptotic proteins in the temporal cortex in schizophrenia: high bax/bcl-2 ratio without caspase-3 activation. Am. J. Psychiatry 161, 109–115 (2004).
Faizi, M., Salimi, A., Rasoulzadeh, M., Naserzadeh, P. & Pourahmad, J. Schizophrenia induces oxidative stress and cytochrome c release in isolated rat brain mitochondria: a possible pathway for induction of apoptosis and neurodegeneration. Iran. J. Pharm. Res. 13, 93–100 (2014).
Bubeníková-Valesová, V., Horácek, J., Vrajová, M. & Höschl, C. Models of schizophrenia in humans and animals based on inhibition of nmda receptors. Neurosci. Biobehav. Res. 32, 1014–1023 (2008).
T, A., K, I., S, N., Y, N. & T, K. Effects of aripiprazole and haloperidol on progression to schizophrenia-like behavioural abnormalities and apoptosis in rodents. Schizophr. Res. 125, 77–87 (2011).
Gladkikh, I. N., Sintsova, O. V., Leychenko, E. V. & Kozlov, S. A. Trpv1 ion channel: structural features, activity modulators, and therapeutic potential. Biochemistry 86, S50–S70 (2021).
Ma, A. Modulation of trpv1 channel function by natural products in the treatment of pain. Chem. Biol. Interact. 330, 109178 (2020).
Escelsior, A. et al. Transient receptor potential vanilloid 1 antagonism in neuroinflammation, neuroprotection and epigenetic regulation: potential therapeutic implications for severe psychiatric disorders treatment. Psychiatr. Genet. 30, 39–48 (2020).
Khan, A. A. et al. Tumor necrosis factor alpha enhances the sensitivity of rat trigeminal neurons to capsaicin. Neuroscience 155, 503–509 (2008).
Rossi, S. et al. Interleukin-1β causes anxiety by interacting with the endocannabinoid system. J. Neurosci. 32, 13896–13905 (2012).
Huang, W. X. et al. Trpv1 promotes repetitive febrile seizures by pro-inflammatory cytokines in immature brain. Brain Behav. Immun. 48, 68–77 (2015).
Mcguire, T., Jackson, C., Dommers, E. & Nyst, P. Interleukin-6-mediated functional upregulation of trpv1 receptors in dorsal root ganglion neurons through the activation of jak/pi3k signaling pathway: roles in the development of bone cancer pain in a rat model. Pain 156, 1124–1144 (2015).
Rantala, M. J., Luoto, S., Borráz-León, J. I. & Krams, I. Schizophrenia: the new etiological synthesis. Neurosci. Biobehav. Res. 142, 104894 (2022).
Srinivasan, K. Biological activities of red pepper (capsicum annuum) and its pungent principle capsaicin: a review. Crit. Rev. Food Sci. Nutr. 56, 1488 (2016).
Moseley, A., Laipply, K. & Rubinstein, J. Capsaicin mediates remote ischemic pre-conditioning to explain improved cardiovascular mortality with chili pepper intake. J. Am. Coll. Cardiol. 75, 1865 (2020).
R, L. et al. Anti-obesity effects of capsaicin and the underlying mechanisms: a review. Food Funct 11, 7356–7370 (2020).
Jc, M. et al. Anti-cancer activity of sustained release capsaicin formulations. Pharmacol. Ther. 238, 108177 (2022).
Pasierski, M. & Szulczyk, B. Beneficial effects of capsaicin in disorders of the central nervous system. Molecules 27, 2484 (2022).
Huang, M. et al. Capsaicin protects cortical neurons against ischemia/reperfusion injury via down-regulating nmda receptors. Exp. Neurol. 295, 66–76 (2017).
Guo, S. Y. et al. Protection of capsaicin against hypoxia-reoxygenation-induced apoptosis of rat hippocampal neurons. Can. J. Physiol. Pharm. 86, 785–792 (2008).
Liu, J. et al. Regulation of actg1 and gsta2 is possible mechanism by which capsaicin alleviates apoptosis in cell model of 6-ohda-induced parkinson’s disease. Bioscience. Rep. 40, BSR20191796 (2020).
Perrin, M., Kleinhaus, K., Messinger, J. & Malaspina, D. Critical periods and the developmental origins of disease: an epigenetic perspective of schizophrenia. Ann. NY Acad. Sci. 1204, E8–E13 (2010).
Kalinichev, M., Easterling, K. W. & Holtzman, S. G. Early neonatal experience of long-evans rats results in long-lasting changes in morphine tolerance and dependence. Psychopharmacology 157, 305–312 (2001).
Lopez-Gallardo et al. The maternal deprivation animal model revisited. Neurosci. Biobehav. Res. 51, 151–163 (2015).
Marco, E. M. et al. Maternal deprivation effects on brain plasticity and recognition memory in adolescent male and female rats. Neuropharmacology 68, 223–231 (2013).
Hao, K. et al. Nicotinamide reverses deficits in puberty-born neurons and cognitive function after maternal separation. J. Neuroinflamm. 19, 232 (2022).
K, H. et al. Nicotinamide ameliorates mitochondria-related neuronal apoptosis and cognitive impairment via the nad/sirt3 pathway. Schizophrenia 9, 32 (2023).
Roceri, M., Hendriks, W., Racagni, G., Ellenbroek, B. A. & Riva, M. A. Early maternal deprivation reduces the expression of bdnf and nmda receptor subunits in rat hippocampus. Mol. Psychiatry 7, 609–616 (2002).
Hughes, Z. A. et al. Estrogen receptor neurobiology and its potential for translation into broad spectrum therapeutics for cns disorders. Curr. Mol. Pharmacol. 2, 215–236 (2009).
Galea, L., Frick, K. M., Hampson, E., Sohrabji, F. & Choleris, E. Why estrogens matter for behavior and brain health. Neurosci. Biobehav. Res. 76, 363–379 (2017).
Dalla, C. & Shors, T. J. Sex differences in learning processes of classical and operant conditioning. Physiol. Behav. 97, 229–238 (2009).
Gu, X. et al. Gsg1l suppresses ampa receptor-mediated synaptic transmission and uniquely modulates ampa receptor kinetics in hippocampal neurons. Nat. Commun. 7, 10873 (2016).
Anglin, D. M., Cohen, P. R. & Chen, H. Duration of early maternal separation and prediction of schizotypal symptoms from early adolescence to midlife. Schizophr. Res. 103, 143–150 (2008).
Ba, E., van den Kroonenberg, P. T. & Ar, C. The effects of an early stressful life event on sensorimotor gating in adult rats. Schizophr. Res. 30, 251–260 (1998).
Gruss, M., Braun, K., Frey, J. U. & Korz, V. Maternal separation during a specific postnatal time window prevents reinforcement of hippocampal long-term potentiation in adolescent rats. Neuroscience 152, 1–7 (2008).
Mcewen, B. S. Stress and hippocampal plasticity. Annu. Rev. Neurosci. 22, 105–122 (1999).
Mcewen, B. S., Nasca, C. & Gray, J. D. Stress effects on neuronal structure: hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology 41, 3–23 (2016).
Szymona, K. et al. Analysis of the expression of bax, bcl2, birc6, casp3, casp9 apoptosis genes during the first episode of schizophrenia. Psychiatr. Pol. 53, 1293–1303 (2019).
Yang, S., Li, J., Han, L. & Zhu, G. Early maternal separation promotes apoptosis in dentate gyrus and alters neurological behaviors in adolescent rats. Int. J. Clin. Exp. Pathol. 10, 10812–10820 (2017).
Fabricius, K., Wörtwein, G. & Pakkenberg, B. The impact of maternal separation on adult mouse behaviour and on the total neuron number in the mouse hippocampus. Brain Struct. Funct. 212, 403–416 (2008).
Dirican, E., özcan, H., Karabulut Uzunçakmak, S. & Takım, U. Evaluation expression of the caspase-3 and caspase-9 apoptotic genes in schizophrenia patients. Clin. Psychopharmacol. Neurosci 21, 171–178 (2023).
V, A. et al. Role of the endocannabinoid and endovanilloid systems in an animal model of schizophrenia-related emotional processing/cognitive deficit. Neuropharmacology 155, 44–53 (2019).
Manseau, M. W. & Goff, D. C. Cannabinoids and schizophrenia: risks and therapeutic potential. Neurotherapeutics 12, 816–824 (2015).
Tzavara, E. T. et al. Endocannabinoids activate transient receptor potential vanilloid 1 receptors to reduce hyperdopaminergia-related hyperactivity: therapeutic implications. Biol. Psychiatry 59, 508–515 (2006).
Pn, N., van den Buuse, M., S, M., A, L. & La, C. Effects of neonatal treatment with the trpv1 agonist, capsaicin, on adult rat brain and behaviour. Behav. Brain Res. 272, 55–65 (2014).
Le, L., Dt, M. & Da, T. Cannabidiol reverses mk-801-induced disruption of prepulse inhibition in mice. Neuropsychopharmacology 31, 795–803 (2006).
Peres, F. F. et al. Cannabidiol, among other cannabinoid drugs, modulates prepulse inhibition of startle in the shr animal model: implications for schizophrenia pharmacotherapy. Front. Pharmacol. 7, 303 (2016).
V, A. et al. Effects of cannabinoid and vanilloid drugs on positive and negative-like symptoms on an animal model of schizophrenia: the shr strain. Schizophr. Res. 153, 150–159 (2014).
Kong, W. L., Peng, Y. Y. & Peng, B. W. Modulation of neuroinflammation: role and therapeutic potential of trpv1 in the neuro-immune axis. Brain Behav. Immun. 64, 354–366 (2017).
F, M., L, Z. & Kn, W. Reactive oxygen species mediate tnfr1 increase after trpv1 activation in mouse drg neurons. Mol. Pain 5, 31 (2009).
Olah, Z. et al. Ligand-induced dynamic membrane changes and cell deletion conferred by vanilloid receptor 1. J. Biol. Chem. 276, 11021–11030 (2001).
Gibson, H. E., Edwards, J. G., Page, R. S., Van Hook, M. J. & Kauer, J. A. Trpv1 channels mediate long-term depression at synapses on hippocampal interneurons. Neuron 57, 746–759 (2008).
Peters, J. H., Mcdougall, S. J., Fawley, J. A., Smith, S. M. & Andresen, M. C. Primary afferent activation of thermosensitive trpv1 triggers asynchronous glutamate release at central neurons. Neuron 65, 657–669 (2010).
Sun, F. J. et al. Increased expression of trpv1 in the cortex and hippocampus from patients with mesial temporal lobe epilepsy. J. Mol. Neurosci. 49, 182–193 (2013).
Stock, K. et al. Neural precursor cells induce cell death of high-grade astrocytomas through stimulation of trpv1. Nat. Med. 18, 1232–1238 (2012).
Sappington, R. M. & Calkins, D. J. Contribution of trpv1 to microglia-derived il-6 and nfkappab translocation with elevated hydrostatic pressure. Invest. Ophthalmol. Vis. Sci. 49, 3004–3017 (2008).
Hassan, S. et al. Cannabidiol enhances microglial phagocytosis via transient receptor potential (trp) channel activation. Br. J. Pharmacol. 171, 2426–2439 (2014).
Kim, S. R., Kim, S. U., Oh, U. & Jin, B. K. Transient receptor potential vanilloid subtype 1 mediates microglial cell death in vivo and in vitro via ca2+-mediated mitochondrial damage and cytochrome c release. J. Immunol. 177, 4322–4329 (2006).
Park, E. S., Kim, S. R. & Jin, B. K. Transient receptor potential vanilloid subtype 1 contributes to mesencephalic dopaminergic neuronal survival by inhibiting microglia-originated oxidative stress. Brain Res. Bull. 89, 92–96 (2012).
H, W. et al. Mer regulates microglial/macrophage m1/m2 polarization and alleviates neuroinflammation following traumatic brain injury. J. Neuroinflamm. 18, 2 (2021).
Du et al. Capsaicin attenuates amyloid-beta-induced synapse loss and cognitive impairments in mice. J. Alzheimers Dis 59, 683–694 (2017).
Wang, J. et al. Capsaicin consumption reduces brain amyloid-beta generation and attenuates alzheimer’s disease-type pathology and cognitive deficits in app/ps1 mice. Transl. Psychiatry 10, 230 (2020).
Liu, C. H. et al. The associations between a capsaicin-rich diet and blood amyloid-β levels and cognitive function. J. Alzheimers Dis. 52, 1081–1088 (2016).
Li, H. B. et al. Antistress effect of trpv1 channel on synaptic plasticity and spatial memory. Biol. Psychiatry 64, 286–292 (2008).
Choi, Y. J., Kim, J. Y., Yoo, S. B., Lee, J. H. & Jahng, J. W. Repeated oral administration of capsaicin increases anxiety-like behaviours with prolonged stress-response in rats. J. Biosci. 38, 561–571 (2013).
Iannotti, F. A. et al. Nonpsychotropic plant cannabinoids, cannabidivarin (cbdv) and cannabidiol (cbd), activate and desensitize transient receptor potential vanilloid 1 (trpv1) channels in vitro: potential for the treatment of neuronal hyperexcitability. ACS Chem. Neurosci. 5, 1131–1141 (2014).
Baek, J. Y. et al. Inhibition of microglia-derived oxidative stress by ciliary neurotrophic factor protects dopamine neurons in vivo from mpp+ neurotoxicity. Int. J. Mol. Sci. 19, 3543 (2018).
Bok, E. et al. Modulation of m1/m2 polarization by capsaicin contributes to the survival of dopaminergic neurons in the lipopolysaccharide-lesioned substantia nigra in vivo. Exp. Mol. Med. 50, 1–14 (2018).
Zhao, R. & Tsang, S. Y. Versatile roles of intracellularly located trpv1 channel. J. Cell. Physiol. 232, 1957–1965 (2017).
Gao, Y., Cao, E., Julius, D. & Cheng, Y. Trpv1 structures in nanodiscs reveal mechanisms of ligand and lipid action. Nature 534, 347–351 (2016).
Acknowledgements
The study was supported by the Medical Science Advancement Program of Wuhan University (NO. TFLC2018001), The Interdisciplinary Innovative Talents Foundation from Renmin Hospital of Wuhan University (JCRCFZ-2022-003) and the Key Research and Development Program of Hubei Province (2020BCA064).
Author information
Authors and Affiliations
Contributions
S.X. and H.W. designed the study and wrote the protocol. S.X., K.H., and Y.X. performed the experiments and analyzed the data. H.H. and R.X. performed literature searches and drew the figures. S.X. wrote the manuscript. K.H. and H.W. revised the manuscript. K.H. has supplemented the experiment as staining TUNEL with NeuN.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval
This work was carried out based on the Regulations of Experimental Animal Administration issued by the State Committee of Science and Technology of the People’s Republic of China, with the approval of the Ethics Committee in Renmin Hospital of Wuhan University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xu, S., Hao, K., Xiong, Y. et al. Capsaicin alleviates neuronal apoptosis and schizophrenia-like behavioral abnormalities induced by early life stress. Schizophr 9, 77 (2023). https://doi.org/10.1038/s41537-023-00406-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-023-00406-4
- Springer Nature Limited