Abstract
Purpose
This study was designed to investigate the association between fibulin-1(FBLN1) and thyroid-associated ophthalmopathy (TAO).
Method
The plasma FBLN1 levels were measured in 80 participants, including 30 active TAO patients, 25 inactive TAO patients, and 25 Graves disease (GD) patients without TAO using enzyme-linked immunosorbent assay (ELISA).
Results
TAO patients had significantly higher TRAb level than GD patients (p < 0.05). The active TAO patients consumed more tobacco and had higher CAS than inactive TAO patients (all p < 0.05). No significant differences were found in age, sex, and the level of FT3, FT4, and TSH between TAO and GD patients, and between the active and inactive TAO patients (all p > 0.05).
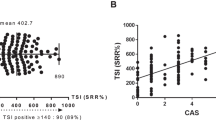
The plasma FBLN1 level in TAO patients was higher than that in GD patients, and that in active patients was higher than that in inactive patients (all p < 0.05). Furthermore, the plasma FBLN1 level showed strong association with clinical activity score (CAS) of TAO (r = 0.67, p < 0.01). By receiver operator characteristic (ROC) curve analysis, FBLN1 demonstrated good efficiency for predicting disease activity at the cut-off value > 625.33 pg/ml with a sensitivity of 93.3% and a specificity of 88.0% (AUC:0.92, p < 0.01).
Conclusion
The plasma FBLN1 levels correlated with TAO activity and a value >625.33 pg/ml was associated with active disease. Our results suggest that the plasma FBLN1 level could be a novel biomarker for predicting disease activity of TAO.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
Bartalena L, Tanda ML. Clinical practice. Graves’ ophthalmopathy. N Engl J Med. 2009;360:994–1001.
Bartalena L, Piantanida E, Gallo D, Lai A, Tanda ML. Epidemiology, Natural History, Risk Factors, and Prevention of Graves’ Orbitopathy. Front Endocrinol. 2020;11:615993.
Pappa A, Lawson JM, Calder V, Fells P, Lightman S. T cells and fibroblasts in affected extraocular muscles in early and late thyroid associated ophthalmopathy. Br J Ophthalmol. 2000;84:517–22.
Wang Y, Smith TJ. Current concepts in the molecular pathogenesis of thyroid-associated ophthalmopathy. Investig Ophthalmol Vis Sci. 2014;55:1735–48.
Bahn RS. Graves’ ophthalmopathy. N Engl J Med. 2010;362:726–38.
Bartalena L, Pinchera A, Marcocci C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev. 2000;21:168–99.
Hai YP, Lee ACH, Frommer L, Diana T, Kahaly GJ. Immunohistochemical analysis of human orbital tissue in Graves’ orbitopathy. J Endocrinol Investig. 2020;43:123–37.
Weetman AP, Cohen S, Gatter KC, Fells P, Shine B. Immunohistochemical analysis of the retrobulbar tissues in Graves’ ophthalmopathy. Clin Exp Immunol. 1989;75:222–7.
Khong JJ, McNab AA, Ebeling PR, Craig JE, Selva D. Pathogenesis of thyroid eye disease: review and update on molecular mechanisms. Br J Ophthalmol. 2016;100:142–50.
Timpl R, Sasaki T, Kostka G, Chu ML. Fibulins: a versatile family of extracellular matrix proteins. Nat Rev Mol Cell Biol. 2003;4:479–89.
Cangemi C, Hansen ML, Argraves WS, Rasmussen LM. Fibulins and their role in cardiovascular biology and disease. Adv Clin Chem. 2014;67:245–65.
Cooley MA, Kern CB, Fresco VM, Wessels A, Thompson RP, McQuinn TC, et al. Fibulin-1 is required for morphogenesis of neural crest-derived structures. Dev Biol. 2008;319:336–45.
Watany MM, Elmashad NM, Badawi R, Hawash N. Serum FBLN1 and STK31 as biomarkers of colorectal cancer and their ability to noninvasively differentiate colorectal cancer from benign polyps. Clin Chim Acta. 2018;483:151–5.
Corvilain B, Hamy A, Brunaud L, Borson-Chazot F, Orgiazzi J, Bensalem Hachmi L, et al. Treatment of adult Graves’ disease. Ann Endocrinol. 2018;79:618–35.
Bartalena L, Kahaly GJ, Baldeschi L, Dayan CM, Eckstein A, Marcocci C, et al. The 2021 European Group on Graves’ orbitopathy (EUGOGO) clinical practice guidelines for the medical management of Graves’ orbitopathy. Eur J Endocrinol. 2021;185:G43–67.
Ge Q, Chen L, Jaffar J, Argraves WS, Twal WO, Hansbro P, et al. Fibulin1C peptide induces cell attachment and extracellular matrix deposition in lung fibroblasts. Sci Rep. 2015;5:9496.
Liu G, Cooley MA, Jarnicki AG, Borghuis T, Nair PM, Tjin G, et al. Fibulin-1c regulates transforming growth factor-β activation in pulmonary tissue fibrosis. JCI Insight, 2019;5:e124529.
Liu G, Cooley MA, Nair PM, Donovan C, Hsu AC, Jarnicki AG, et al. Airway remodelling and inflammation in asthma are dependent on the extracellular matrix protein fibulin-1c. J Pathol. 2017;243:510–23.
Li B, Smith TJ. Regulation of IL-1 receptor antagonist by TSH in fibrocytes and orbital fibroblasts. J Clin Endocrinol Metab. 2014;99:E625–33.
Gong J, Jie Y, Xiao C, Zhou W, Li X, Chen Y, et al. Increased Expression of Fibulin-1 Is Associated With Hepatocellular Carcinoma Progression by Regulating the Notch Signaling Pathway. Front Cell Dev Biol. 2020;8:478.
Lau JY, Oliver BG, Baraket M, Beckett EL, Hansbro NG, Moir LM, et al. Fibulin-1 is increased in asthma-a novel mediator of airway remodeling? PLoS ONE. 2010;5:e13360.
Funding
This study is supported by Key projects of Chongqing Natural Science Foundation (NO:cstc2020jcyj-zdxmX0016).
Author information
Authors and Affiliations
Contributions
JL and CL contributed to the conception of the study. HH, XJ, and ZF performed the experiment. HH and JL performed the data analyses and wrote the paper. LL and XZ contributed to the diagnosis and assessment of TAO.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The research was conducted ethically following the World Medical Association Declaration of Helsinki. The study was approved by the First Affiliated Hospital of Chongqing Medical University Ethical Committee.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41433_2022_2318_MOESM1_ESM.doc
Heatmap of the log2 Fold Change (logFC) of differential expression proteins in the plasma of TAO and GD group, through Tandem mass tag proteomics(TMT).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, H., Liang, L., Zheng, X. et al. Fibulin-1: a novel biomarker for predicting disease activity of the thyroid-associated ophthalmopathy. Eye 37, 2216–2219 (2023). https://doi.org/10.1038/s41433-022-02318-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02318-6
- Springer Nature Limited