Abstract
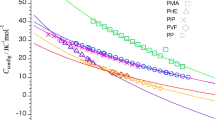
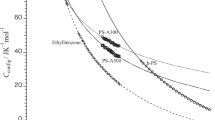
In this study, the absolute values for the configurational heat capacity of polyisobutylene (PIB), isobutane (which corresponds to the monomer of PIB), and 2,2,4-isomethylpentane (which corresponds to the dimer of PIB) were estimated based on the assumption that skeletal and group vibration modes do not change before and after the glass transition. The configurational heat capacities evaluated for these three substances decreased with increasing temperature. This temperature dependence of the configurational heat capacity is the same as that previously observed for molecular glass, which has a simple molecular structure. Additionally, the configurational heat capacities for the three substances were found to be different at the same temperature. This suggests that interactions with the surroundings that determine the configuration and orientation of the molecules differ as to the molecular weight changes.



Similar content being viewed by others
References
Wagman DD, Evans WH, Parker VB, Schumm RH, Halow I, Bailey SM. The NBS tables of chemical thermodynamic properties: selected values for inorganic and C1 and C2 organic substances in SI units. J Phys Chem Ref Dat. 1982;11:1–392.
Gopal ESR. Specific heats at low temperatures. London: Springer; 2012.
Wunderlich B. Thermal analysis of polymeric materials. Heidelberg: Springer; 2005.
Glassy, amorphous and nano-crystalline materials: thermal physics, analysis, structure and properties. In: Šesták J, Mareš JJ, Hubík P, editors. Hot topics in thermal analysis and calorimetry. vol. 8. Springer Science & Business Media; 2010.
Gibson GE, Giauque WF. The third law of thermodynamics. Evidence from the specific heats of glycerol that the entropy of a glass exceeds that of a crystal at the absolute zero. J Am Chem Soc. 1923;45:93–104.
Haida O, Matsuo T, Suga H, Seki S. Calorimetric study of the glassy state X. Enthalpy relaxation at the glass-transition temperature of hexagonal ice. J Chem Thermodyn. 1974;6:815–25.
Tajima Y, Matsuo T, Suga H. Calorimetric study of phase transition in hexagonal ice doped with alkali hydroxides. J Phys Chem Solids. 1984;45:1135–44.
Kume Y, Muraoka H, Yamamuro O, Matsuo T. Deuteration-induced phase transition in ammonium hexachloroplumbate. J Chem Phys 1988;108:4090–7.
Miyazaki Y, Wang Q, Sato A, Saito K, Yamamoto M, Kitagawa H, et al. Heat capacity of the halogen-bridged mixed-valence complex Pt2(dta)4I(dta = CH3CS2−). J Phys Chem B. 2002;106:197–202.
Yamamura Y, Nakajima N, Tsuji T, Koyano M, Iwasa Y, Katayama S, et al. Low temperature heat capacities and Raman spectra of negative thermal expansion compounds ZrW2O8 and HfW2O8. Phys Rev B. 2002;66:014301.
Matsuo T, Maekawa T, Inaba A, Yamamuro O, Ohama M, Ichikawa M, et al. Isotope-dependent crystalline phases at ambient temperature: spectroscopic and calorimetric evidence for a deuteration-induced phase transition at 320 K in α-DCrO2. J Mol Struct. 2006;790:129–34.
Saito K, Sato A, Kikuchi K, Nishikawa H, Ikemoto I, Sorai M. Calorimetric study of metal-insulator transition in (DIMET)2I3. J Phys Soc Jpn. 2000;69:3602–6.
Yamamuro O, Tsukushi I, Lindqvist A, Takahara S, Ishikawa M, Matsuo T. Calorimetric study of glassy and liquid toluene and ethylbenzene: thermodynamic approach to spatial heterogeneity in glass-forming molecular liquids. J Phys Chem B. 1998;102:1605–9.
Pyda M, Bartkowiak M, Wunderlich B. Computation of heat capacities of solids using a general Tarasov equation. J Therm Anal 1998;52:631–56.
Tarasov VV, Yunitskii GA. Theory of heat capacity of chain and layer structures. Russ J Phys Chem. 1965;39:1109–11.
Debye P. Zur Theorie der spezifischen Wärmen. Ann Phys. 1912;344:789–839.
Einstein A. Die Plancksche Theorie der Strahlung und die Theorie der spezifischen Wärme. Ann Phys. 1907;327:180–90.
Yokota M, Sugane K, Tsukushi I, Shibata M. Evaluation of the heat capacity of amorphous polymers composed of a carbon backbone below the glass-transition temperature. Polym J. 2020;52:765–74.
Yokota M, Nishiyama E, Fujimura J, Tsukushi I. Excess heat capacity for low-molecular-weight amorphous polystyrene below the glass-transition temperature: influence of end groups. Polym J. 2020;52:575–80.
Yokota M, Tsukushi I. Heat capacities of polymer solids composed of polyesters and poly(oxide)s, evaluated below the glass-transition temperature. Polym J. 2020;52:1103–11.
Yokota M, Tsukushi I. Prediction of the heat capacity of main-chain-type polymers below the glass transition temperature. Polym J 2020;52:1113–20.
Yamamuro O, Tsukushi I, Lindqvist A, Takahara S, Ishikawa M, Matsuo T. Calorimetric study of glassy and liquid toluene and ethylbenzene: thermodynamic approach to spatial heterogeneity in glass-forming molecular liquids. J Phys Chem B. 1998;102:1605–9.
Tatsumi S, Aso S, Yamamuro O. Thermodynamic study of simple molecular glasses: universal features in their heat capacity and the size of the cooperatively rearranging regions. Phys Rev Lett. 2012;109:045701.
Aston JG, Kennedy RM, Schumann SC. The heat capacity and entropy, heats of fusion and vaporization and the vapor pressure of isobutane. J Am Chem Soc 1940;62:2059–63.
Pitzer KS. The thermodynamics of n-heptane and 2, 2, 4-trimethylpentane, including heat capacities, heats of fusion and vaporization and entropies. J Am Chem Soc. 1940;62:1224–7.
Furukawa GT, Reilly ML. Heat capacity of polyisobutylene from 0 to 380 K. J Res Natl Bur Stand. 1956;56:285–88.
Nernst W, Lindemann FA. Spezifische Wärme und Quantentheorie. Z Elektrochem. 1911;17:817.
NIST Chemistry WebBook SRD69. Gaithersburg, MD, 301-975-2000. 1901. https://webbook.nist.gov/cgi/cbook.cgi?ID=C106989&Units=SI&Mask=80#IR-Spec. Accessed 7 Nov 2019.
Kauzmann K. The nature of the glassy state and the behavior of liquids at low temperatures. Chem Rev. 1948;43:219–56.
Angell CA, Choi Y. Crystallization and vitrification in aqueous systems. J Microsc. 1986;141:251–61.
Boyer RF. Relationship of first‐ to second‐order transition temperatures for crystalline high polymers. J Appl Phys. 1954;25:825–9.
Miwa Y, Urakawa O, Nobukawa S, Kutsumizu S. Selective determination of glass transition temperature and vibrational properties at the chain end of polystyrene by Fourier transform infrared measurement in combination with deuterium-labeling. Polymer 2015;59:194–9.
Tashiro K, Yoshioka A. Molecular mechanism of solvent-induced crystallization of syndiotactic polystyrene glass. 2. Detection of enhanced motion of the amorphous chains in the induction period of crystallization. Macromolecules 2002;35:410–4.
Acknowledgements
The authors would like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nishiyama, E., Yokota, M. & Tsukushi, I. Estimation of the configurational heat capacity of polyisobutylene, isobutane and 2,2,4-isomethylpentane above the glass transition temperature. Polym J 53, 1031–1036 (2021). https://doi.org/10.1038/s41428-021-00503-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-021-00503-0
- Springer Nature Limited