Abstract
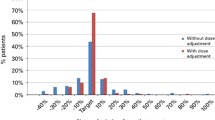
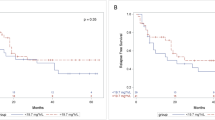
To identify relationships between busulfan (Bu) exposure and outcomes of a cohort pediatric patients receiving hematopoietic stem cell transplantation (HSCT), along with a targeted busulfan-based conditioning regimen. We retrospectively evaluated targeted busulfan concentrations in 53 pediatric patients (age 0.4–16 years) who received busulfan 4 times daily according to recommended weight-based doses in a single-center analysis between 2018 and 2020. In this trial, individual busulfan pharmacokinetics were performed following dose 5 of the conditioning regimen. Twenty four of 53 patients (45.3%) studies did not require dose adjustments. Equal number of patients (24/53) required one dose adjustments while two-dose adjustment applied for 5 of 53 (9.4%). Twenty-one percent of the patients exhibited ll-lV aGVHD. The incidence of veno-occlusive disease (VOD) was in 3.8% of the 53 patients, while incidence of hemorrhagic cystitis (II–III) reached to 9.7%. Engraftment was successful in 98% of the 53 patients with relapse in 2% of cases. The probability of overall survival and disease-free survival at day 100 was 96% and 94%, respectively. In conclusion, therapeutic drug monitoring (TDM) and individualization of Bu dosage are essential to improve the efficacy and safety of busulfan-based regimen in Chinese pediatric HSCT recipients.




Similar content being viewed by others
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
References
Aschan J. Risk assessment in haematopoietic stem cell transplantation: conditioning. Best Pract Res Clin Haematol. 2007;20:295–310. https://doi.org/10.1016/j.beha.2006.09.004.
Philippe M, Neely M, Rushing T, Bertrand Y, Bleyzac N, Goutelle S. Maximal concentration of intravenous busulfan as a determinant of veno-occlusive disease: a pharmacokinetic-pharmacodynamic analysis in 293 hematopoietic stem cell transplanted children. Bone Marrow Transplant. 2019;54:448–57. https://doi.org/10.1038/s41409-018-0281-7.
Bartelink IH, Bredius RG, Belitser SV, Suttorp MM, Bierings M, Knibbe CA, et al. Association between busulfan exposure and outcome in children receiving intravenous busulfan before hematologic stem cell transplantation. Biol Blood Marrow Transplant. 2009;15:231–41. https://doi.org/10.1016/j.bbmt.2008.11.022.
Long-Boyle JR, Savic R, Yan S, Bartelink I, Musick L, French D, et al. Population pharmacokinetics of busulfan in pediatric and young adult patients undergoing hematopoietic cell transplant: a model-based dosing algorithm for personalized therapy and implementation into routine clinical use. Ther Drug Monit. 2015;37:236–45. https://doi.org/10.1097/FTD.0000000000000131.
Ansari M, Theoret Y, Rezgui MA, Peters C, Mezziani S, Desjean C, et al. Association between busulfan exposure and outcome in children receiving intravenous busulfan before hematopoietic stem cell transplantation. Ther Drug Monit. 2014;36:93–9. https://doi.org/10.1097/FTD.0b013e3182a04fc7.
Bartelink IH, Lalmohamed A, van Reij EM, Dvorak CC, Savic RM, Zwaveling J, et al. Association of busulfan exposure with survival and toxicity after haemopoietic cell transplantation in children and young adults: a multicentre, retrospective cohort analysis. Lancet Haematol. 2016;3:e526–36. https://doi.org/10.1016/S2352-3026(16)30114-4.
Bolinger AM, Zangwill AB, Slattery JT, Risler LJ, Sultan DH, Glidden DV, et al. Target dose adjustment of busulfan in pediatric patients undergoing bone marrow transplantation. Bone Marrow Transplant. 2001;28:1013–8. https://doi.org/10.1038/sj.bmt.1703264.
Nguyen L, Fuller D, Lennon S, Leger F, Puozzo C. I.V. busulfan in pediatrics: a novel dosing to improve safety/efficacy for hematopoietic progenitor cell transplantation recipients. Bone Marrow Transplant. 2004;33:979–87. https://doi.org/10.1038/sj.bmt.1704446.
Bearman SI, Appelbaum FR, Buckner CD, Petersen FB, Fisher LD, Clift RA, et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol. 1988;6:1562–8. https://doi.org/10.1200/JCO.1988.6.10.1562.
McDonald GB, Hinds MS, Fisher LD, Schoch HG, Wolford JL, Banaji M, et al. Veno-occlusive disease of the liver and multiorgan failure after bone marrow transplantation: a cohort study of 355 patients. Ann Intern Med. 1993;118:255–67. https://doi.org/10.7326/0003-4819-118-4-199302150-00003.
Martino R, Romero P, Subira M, Bellido M, Altes A, Sureda A, et al. Comparison of the classic Glucksberg criteria and the IBMTR Severity Index for grading acute graft-versus-host disease following HLA-identical sibling stem cell transplantation. International Bone Marrow Transplant Registry. Bone Marrow Transplant. 1999;24:283–7. https://doi.org/10.1038/sj.bmt.1701899.
Lee JW, Kang HJ, Lee SH, Yu KS, Kim NH, Yuk YJ, et al. Highly variable pharmacokinetics of once-daily intravenous busulfan when combined with fludarabine in pediatric patients: phase I clinical study for determination of optimal once-daily busulfan dose using pharmacokinetic modeling. Biol Blood Marrow Transplant. 2012;18:944–50. https://doi.org/10.1016/j.bbmt.2011.11.025.
Bashey A, Zhang X, Sizemore CA, Manion K, Brown S, Holland HK, et al. T-cell-replete HLA-haploidentical hematopoietic transplantation for hematologic malignancies using post-transplantation cyclophosphamide results in outcomes equivalent to those of contemporaneous HLA-matched related and unrelated donor transplantation. J Clin Oncol. 2013;31:1310–6. https://doi.org/10.1200/JCO.2012.44.3523.
Hong KT, Kang HJ, Choi JY, Hong CR, Cheon JE, Park JD, et al. Favorable outcome of post-transplantation cyclophosphamide haploidentical peripheral blood stem cell transplantation with targeted busulfan-based myeloablative conditioning using intensive pharmacokinetic monitoring in pediatric patients. Biol Blood Marrow Transplant. 2018;24:2239–44. https://doi.org/10.1016/j.bbmt.2018.06.034.
Lee JW, Kang HJ, Kim S, Lee SH, Yu KS, Kim NH, et al. Favorable outcome of hematopoietic stem cell transplantation using a targeted once-daily intravenous busulfan-fludarabine-etoposide regimen in pediatric and infant acute lymphoblastic leukemia patients. Biol Blood Marrow Transplant. 2015;21:190–5. https://doi.org/10.1016/j.bbmt.2014.09.013.
Rhee SJ, Lee JW, Yu KS, Hong KT, Choi JY, Hong CR, et al. Pediatric patients undergoing hematopoietic stem cell transplantation can greatly benefit from a novel once-daily intravenous busulfan dosing nomogram. Am J Hematol. 2017;92:607–13. https://doi.org/10.1002/ajh.24734.
Geddes M, Kangarloo SB, Naveed F, Quinlan D, Chaudhry MA, Stewart D, et al. High busulfan exposure is associated with worse outcomes in a daily i.v. busulfan and fludarabine allogeneic transplant regimen. Biol Blood Marrow Transplant. 2008;14:220–8. https://doi.org/10.1016/j.bbmt.2007.10.028.
Malar R, Sjoo F, Rentsch K, Hassan M, Gungor T. Therapeutic drug monitoring is essential for intravenous busulfan therapy in pediatric hematopoietic stem cell recipients. Pediatr Transplant. 2011;15:580–8. https://doi.org/10.1111/j.1399-3046.2011.01529.x.
Schechter T, Finkelstein Y, Doyle J, Verjee Z, Moretti M, Koren G, et al. Pharmacokinetic disposition and clinical outcomes in infants and children receiving intravenous busulfan for allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2007;13:307–14. https://doi.org/10.1016/j.bbmt.2006.10.026.
Takamatsu Y, Sasaki N, Eto T, Nagafuji K, Abe Y, Choi I, et al. Individual dose adjustment of oral busulfan using a test dose in hematopoietic stem cell transplantation. Int J Hematol. 2007;86:261–8. https://doi.org/10.1532/IJH97.07013.
Wall DA, Chan KW, Nieder ML, Hayashi RJ, Yeager AM, Kadota R, et al. Safety, efficacy, and pharmacokinetics of intravenous busulfan in children undergoing allogeneic hematopoietic stem cell transplantation. Pediatr Blood Cancer. 2010;54:291–8. https://doi.org/10.1002/pbc.22227.
Philippe M, Goutelle S, Guitton J, Fonrose X, Bergeron C, Girard P, et al. Should busulfan therapeutic range be narrowed in pediatrics? Experience from a large cohort of hematopoietic stem cell transplant children. Bone Marrow Transplant. 2016;51:72–8. https://doi.org/10.1038/bmt.2015.218.
Vassal G, Michel G, Esperou H, Gentet JC, Valteau-Couanet D, Doz F, et al. Prospective validation of a novel IV busulfan fixed dosing for paediatric patients to improve therapeutic AUC targeting without drug monitoring. Cancer Chemother Pharmacol. 2008;61:113–23. https://doi.org/10.1007/s00280-007-0455-2.
Trame MN, Bergstrand M, Karlsson MO, Boos J, Hempel G. Population pharmacokinetics of busulfan in children: increased evidence for body surface area and allometric body weight dosing of busulfan in children. Clin Cancer Res. 2011;17:6867–77. https://doi.org/10.1158/1078-0432.CCR-11-0074.
Tesfaye H, Branova R, Klapkova E, Prusa R, Janeckova D, Riha P, et al. The importance of therapeutic drug monitoring (TDM) for parenteral busulfan dosing in conditioning regimen for hematopoietic stem cell transplantation (HSCT) in children. Ann Transplant. 2014;19:214–24. https://doi.org/10.12659/AOT.889933.
Buffery PJ, Allen KM, Chin PK, Moore GA, Barclay ML, Begg EJ. Thirteen years’ experience of pharmacokinetic monitoring and dosing of busulfan: can the strategy be improved? Ther Drug Monit. 2014;36:86–92. https://doi.org/10.1097/FTD.0b013e31829dc940.
Gaziev J, Nguyen L, Puozzo C, Mozzi AF, Casella M, Perrone Donnorso M, et al. Novel pharmacokinetic behavior of intravenous busulfan in children with thalassemia undergoing hematopoietic stem cell transplantation: a prospective evaluation of pharmacokinetic and pharmacodynamic profile with therapeutic drug monitoring. Blood. 2010;115:4597–604. https://doi.org/10.1182/blood-2010-01-265405.
Acknowledgements
The authors would like to thank the children and their parents who have participated in this research. Extreme gratitude also goes out to the staff nurses of Pediatric Hematology and Oncology, for their very precise work in sample collection and incredible care of our beloved patients. The authors acknowledge the staff of experimental center and Clinical Pharmacy of Hospital of Capital Institute of Pediatrics and the Medical Laboratory of Beijing Boren Hospital.
Funding
This work was partly supported by grants from the youth development program of Capital Institute of Pediatrics (PY-2017-03), young backbone individual project of Beijing Excellent Talents Training Fund (2017000021469G247).
Author information
Authors and Affiliations
Contributions
RL and X-DS designed the research; DFS performed the research, analyzed the data, and wrote the manuscript; JL, TH, Z-XZ, lZ, Jl, JC, SF, RT, DZ, ZS, MY, MH, lX, and MZ contributed to the patient recruitment and treatment; HZ and XW detected some Bu concentrations and collected some samples, respectively, and all authors gave final approval for the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki. Guardians and/or patients provided written informed consent before study procedures began. The protocol was approved by institutional review boards at Children’s Hospital of Capital Institute of Pediatrics.
Consent for publication
This manuscript has been read and approved by all the authors to publish and is not submitted or under consideration for publication elsewhere.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shao, Df., Li, Jh., Hu, T. et al. Clinical outcomes of individualized busulfan-dosing in hematopoietic stem cell transplantation in Chinese children undergoing with therapeutic drug monitoring. Bone Marrow Transplant 57, 473–478 (2022). https://doi.org/10.1038/s41409-021-01545-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01545-x
- Springer Nature Limited