Abstract
Study design
Preclinical pharmacology.
Objectives
Our study aims to evaluate the combined effect of Methylprednisolone (MP) and growth factor-rich serum (GFRS) on structural and functional recovery in rats following spinal cord injury (SCI).
Setting
Shiraz University of Medical Sciences, Shiraz, Iran
Methods
Male Sprague-Dawley rats were randomly assigned to five groups: sham group (laminectomy); SCI group (the spinal cord clip compression model); SCI-MP group (30 mg/kg MP was administrated intraperitoneally (IP) immediately after SCI); SCI-GFRS group (GFRS (200 µl, IP) was administrated for six consecutive days); and SCI-MP + GFRS group (the rats received MP (30 mg/kg, IP) immediately after SCI, and GFRS (200 µl, IP) for six consecutive days). Motor function was assessed weekly using the Basso, Beattie, and Bresnahan (BBB) scale. After 4 weeks, we conducted the rotarod test, then removed and prepared the spinal cords (including the epicenter of injury) for stereological and histological estimation, and biochemical assays.
Results
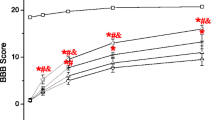
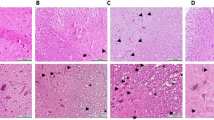
The results showed that MP and GFRS combining treatment enhanced functional recovery, which was associated with a decrement in lesion volume, increased spared white and gray matter volume, reduced neuronal loss, as well as decreased necrosis and hemorrhage after SCI. Moreover, administration of MP and GFRS inhibited lipid peroxidation (malondialdehyde (MDA) content), and increased antioxidant enzymes including glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) after rat SCI.
Conclusions
We suggests that the combination treatment of MP and GFRS may ameliorate the structure and functional changes following SCI by reducing oxidative stress, and increasing the level of antioxidants enzymes.





Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anjum A, Yazid MDI, Fauzi Daud M, Idris J, Ng AMH, Selvi Naicker A, et al. Spinal cord injury: pathophysiology, multimolecular interactions, and underlying recovery mechanisms. Int J Mol Sci. 2020;21:7533.
Schuld C, Franz S, Brüggemann K, Heutehaus L, Weidner N, Kirshblum SC, et al. International standards for neurological classification of spinal cord injury: impact of the revised worksheet (revision 02/13) on classification performance. J Spinal Cord Med. 2016;39:504–12.
Hausmann OB. Post-traumatic inflammation following spinal cord injury. Spinal Cord. 2003;41:369–78.
Okada S. The pathophysiological role of acute inflammation after spinal cord injury. Inflamm Regeneration. 2016;36:1–7.
Sayer FT, Kronvall E, Nilsson OG. Methylprednisolone treatment in acute spinal cord injury: the myth challenged through a structured analysis of published literature. Spine J. 2006;6:335–43.
Bartholdi D, Schwab ME. Methylprednisolone inhibits early inflammatory processes but not ischemic cell death after experimental spinal cord lesion in the rat. Brain Res. 1995;672:177–86.
Fu ES, Saporta S. Methylprednisolone inhibits production of interleukin-1β and interleukin-6 in the spinal cord following compression injury in rats. J Neurosurgical Anesthesiol. 2005;17:82–5.
Xu J, Fan G, Chen S, Wu Y, Xu XM, Hsu CY. Methylprednisolone inhibition of TNF-α expression and NF-kB activation after spinal cord injury in rats. Mol Brain Res. 1998;59:135–42.
Chikuda H, Yasunaga H, Takeshita K, Horiguchi H, Kawaguchi H, Ohe K, et al. Mortality and morbidity after high-dose methylprednisolone treatment in patients with acute cervical spinal cord injury: a propensity-matched analysis using a nationwide administrative database. Emerg Med J. 2014;31:201–6.
Pavlovic V, Ciric M, Jovanovic V, Stojanovic P. Platelet rich plasma: a short overview of certain bioactive components. Open Med. 2016;11:242–7.
Anitua E, Prado R, Sánchez M, Orive G. Platelet-rich plasma: preparation and formulation. Operative Tech Orthop. 2012;22:25–32.
Kojima A, Tator CH. Intrathecal administration of epidermal growth factor and fibroblast growth factor 2 promotes ependymal proliferation and functional recovery after spinal cord injury in adult rats. J Neurotrauma. 2002;19:223–38.
Chen N-F, Sung C-S, Wen Z-H, Chen C-H, Feng C-W, Hung H-C, et al. Therapeutic effect of platelet-rich plasma in rat spinal cord injuries. Front Neurosci. 2018;12:252.
White RE, Yin FQ, Jakeman LB. TGF-α increases astrocyte invasion and promotes axonal growth into the lesion following spinal cord injury in mice. Exp Neurol. 2008;214:10–24.
Baffour R, Achanta K, Kaufman J, Berman J, Garb JL, Rhee S, et al. Synergistic effect of basic fibroblast growth factor and methylprednisolone on neurological function after experimental spinal cord injury. J Neurosurg. 1995;83:105–10.
Bi J, Feng E, Sun P, Shen J, Chen C, Tan H, et al. Melatonin synergizes with methylprednisolone to ameliorate acute spinal cord injury. Front Pharmacol. 2022;12:3764.
Al Shoyaib A, Archie SR, Karamyan VT. Intraperitoneal route of drug administration: should it be used in experimental animal studies? Pharm Res. 2020;37:1–17.
Poon PC, Gupta D, Shoichet MS, Tator CH. Clip compression model is useful for thoracic spinal cord injuries: histologic and functional correlates. Spine. 2007;32:2853–9.
Kang B-J, Yu S-H, Cho D-C, Sung J-K, Park J-Y, Cho H-J, et al. Neurologic and histological study of clip compression model for rat thoracic spinal cord injuries. Korean J Spine. 2011;8:24–30.
Rivlin A, Tator C. Effect of duration of acute spinal cord compression in a new acute cord injury model in the rat. Surgical Neurol. 1978;10:38–43.
Khorsand Ghaffari M. A modified method for Preparation of growth factor rich serum (GFRS) from human blood. 2021. https://doi.org/10.13140/RG.2.2.16164.81289.
Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12:1–21.
Avila-Martin G, Mata-Roig M, Galan-Arriero I, Taylor JS, Busquets X, Escriba PV. Treatment with albumin-hydroxyoleic acid complex restores sensorimotor function in rats with spinal cord injury: efficacy and gene expression regulation. Plos one. 2017;12:e0189151.
Erfanizadeh M, Noorafshan A, Naseh M, Karbalay-Doust S. The effects of copper sulfate on the structure and function of the rat cerebellum: a stereological and behavioral study. IBRO Neurosci Rep. 2021;11:119–27.
Asadi-Golshan R, Razban V, Mirzaei E, Rahmanian A, Khajeh S, Mostafavi-Pour Z, et al. Efficacy of dental pulp-derived stem cells conditioned medium loaded in collagen hydrogel in spinal cord injury in rats: stereological evidence. J Chem Neuroanat. 2021;116:101978.
Gundersen H, BENDTSEN TF, KORBO L, MARCUSSEN N, Møller A, Nielsen K, et al. Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. Apmis. 1988;96:379–94.
Kristiansen SLB, Nyengaard JR. Digital stereology in neuropathology. Apmis. 2012;120:327–40.
Aziz I, Che Ramli MD, Mohd Zain NS, Sanusi J. Behavioral and histopathological study of changes in spinal cord injured rats supplemented with Spirulina platensis. Evid Based Complement Alternat Med. 2014;2014:871657.
de Mesquita Coutinho P, Cristante A, de Barros Filho T, Ferreira R, Dos Santos G. Effects of tacrolimus and erythropoietin in experimental spinal cord lesion in rats: functional and histological evaluation. Spinal Cord. 2016;54:439–44.
Naseh M, Dehghanian A, Ketabchi F. Vagotomy improves hypoxic pulmonary vasoconstriction in rats subjected to brain ischemia-reperfusion injury. Iran J Med Sci. 2020;45:250.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54.
Jia Z, Zhu H, Li J, Wang X, Misra H, Li Y. Oxidative stress in spinal cord injury and antioxidant-based intervention. Spinal cord. 2012;50:264–74.
Lucas JH, Wheeler DG, Guan Z, Suntres Z, Stokes BT. Effect of glutathione augmentation on lipid peroxidation after spinal cord injury. J neurotrauma. 2002;19:763–75.
Wang W, Ballatori N. Endogenous glutathione conjugates: occurrence and biological functions. Pharmacol Rev. 1998;50:335–56.
Jiang ZS, Pu ZC, Hao ZH. Carvacrol protects against spinal cord injury in rats via suppressing oxidative stress and the endothelial nitric oxide synthase pathway. Mol Med Rep. 2015;12:5349–54.
Yan P, Xu J, Li Q, Chen S, Kim G-M, Hsu CY, et al. Glucocorticoid receptor expression in the spinal cord after traumatic injury in adult rats. J Neurosci. 1999;19:9355–63.
Cabrera-Aldana EE, Ruelas F, Aranda C, Rincon-Heredia R, Martínez-Cruz A, Reyes-Sánchez A, et al. Methylprednisolone administration following spinal cord injury reduces aquaporin 4 expression and exacerbates edema. Mediators of inflammation. 2017;1–7. https://doi.org/10.1155/2017/4792932.
Zou H-j, Guo S-W, Zhu L, Xu X, Liu J-b. Methylprednisolone induces neuro-protective effects via the inhibition of A1 astrocyte activation in traumatic spinal cord injury mouse models. Front Neurosci. 2021;15. https://doi.org/10.3389/fnins.2021.628917.
Liu J-t, Zhang S, Gu B, Li H-n, Wang S-y, Zhang S-y. Methotrexate combined with methylprednisolone for the recovery of motor function and differential gene expression in rats with spinal cord injury. Neural Regeneration Res. 2017;12:1507.
Pereira JE, Costa LM, Cabrita AM, Couto PA, Filipe VM, Magalhães LG, et al. Methylprednisolone fails to improve functional and histological outcome following spinal cord injury in rats. Exp Neurol. 2009;220:71–81.
Li X-G, Lin X-J, Du J-H, Xu S-Z, Lou X-F, Chen Z. Combination of methylprednisolone and rosiglitazone promotes recovery of neurological function after spinal cord injury. Neural Regeneration Res. 2016;11:1678.
Mu X, Azbill RD, Springer JE. Riluzole and methylprednisolone combined treatment improves functional recovery in traumatic spinal cord injury. J Neurotrauma. 2000;17:773–80.
Ji B, Li M, Budel S, Pepinsky RB, Walus L, Engber TM, et al. Effect of combined treatment with methylprednisolone and soluble Nogo‐66 receptor after rat spinal cord injury. Eur J Neurosci. 2005;22:587–94.
Jones T, McDaniel E, Popovich P. Inflammatory-mediated injury and repair in the traumatically injured spinal cord. Curr Pharm Des. 2005;11:1223–36.
Jones LL, Oudega M, Bunge MB, Tuszynski MH. Neurotrophic factors, cellular bridges and gene therapy for spinal cord injury. J Physiol. 2001;533:83–9.
Lu P, Wang Y, Graham L, McHale K, Gao M, Wu D, et al. Long-distance growth and connectivity of neural stem cells after severe spinal cord injury. Cell. 2012;150:1264–73.
Shen Y-X, Fan Z-H, Zhao J-G, Zhang P. The application of platelet-rich plasma may be a novel treatment for central nervous system diseases. Med Hypotheses. 2009;73:1038–40.
Ozturk AM, Sozbilen MC, Sevgili E, Dagci T, Özyalcin H, Armagan G. Epidermal growth factor regulates apoptosis and oxidative stress in a rat model of spinal cord injury. Injury. 2018;49:1038–45.
Acknowledgements
This work was performed at the Histomorphometry and Stereology Research Center, Shiraz University of Medical Sciences, Shiraz, Iran. The authors would like to thank Dr. Seyed Ali Hosseini (Native-Speaking Language Editor) for improving the use of English in the manuscript.
Funding
This work was performed at the Histomorphometry and Stereology Research Center and was financially supported by grant No. 1401.009 from Shiraz University of Medical Sciences, Shiraz, Iran.
Author information
Authors and Affiliations
Contributions
SRM: Designing the study, supervising laboratory works. MRF: Conceptualization, Methodology. MKH: Performing laboratory works, collecting the data and Analysis of the data. FK: Conceptualization, Methodology. SK: Conceptualization, Methodology. ARD: Conceptualization, Methodology. MN: Analysis of the data, writing, editing and revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All experimental procedures in the current study were done in accordance with the National Institutes of Health guidelines for the care and use of laboratory animals (NIH Publications No. 8023, revised 1978) and were approved by the Medical and Research Ethics Committee of Shiraz University of Medical Sciences, Shiraz, Iran (Approval No. IR.SUMS.AEC.1401.009).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mousavi, S.R., Farrokhi, M.R., Ghaffari, M.K. et al. The combination treatment of methylprednisolone and growth factor-rich serum ameliorates the structural and functional changes after spinal cord injury in rat. Spinal Cord 62, 17–25 (2024). https://doi.org/10.1038/s41393-023-00942-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-023-00942-x
- Springer Nature Limited