Abstract
Background
To provide insight in the treatment variation of very-low-risk prostate cancer patients and to assess the role of hospital-related factors.
Methods
All patients diagnosed with very-low-risk prostate cancer (cT1c-cT2a, PSA < 10 ng/ml, Gleason score <7 and <3 positive cores) in 2015 and 2016 were identified through the population-based Netherlands Cancer Registry. Multilevel logistic regression analyses were performed to examine the crude and case-mix adjusted probability of immediate treatment vs. active-surveillance (AS) according to hospital of diagnosis and to evaluate the effect of patient-, tumour-, and hospital-related factors.
Results
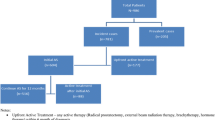
In all, 2047 (85.4%) of the 2396 patients with very-low-risk prostate cancer were managed with AS. The crude proportion of patients with AS varied from 33.3 to 100% between hospitals. Case-mix adjusted probability varied from 71 to 97%. Tumour stage cT2a vs. cT1c (OR 2.0, 95%CI 1.1−3.6), two vs. one positive core (OR 2.8, 95%CI 1.6−4.7), diagnostic MRI (OR 2.8, 95%CI 1.5−5.2), discussion of a patient in a multi-disciplinary team (OR 2.2, 95%CI 1.1−4.5), discussion of treatment options with the patient (OR 3.3, 95%CI 1.5−7.4) and type of hospital (non-university referral hospital vs. community hospital: OR 0.5, 95%CI 0.2−0.9) were associated with immediate treatment.
Conclusion
The majority of Dutch very-low-risk prostate cancer patients is managed with AS but variation between hospitals exists. Part of the variation is explained by patient- and tumour characteristics but also hospital-related factors play a role. This implies that clinical practice could be improved.


Similar content being viewed by others
References
Ferlay JSI, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M. et al. GLOBOCAN 2012v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer. 2013. http://globocan.iarc.fr. Accessed 2012.
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71:618–29.
Loeb S, Folkvaljon Y, Curnyn C, Robinson D, Bratt O, Stattin P. Uptake of active surveillance for very-low-risk prostate cancer in Sweden. JAMA Oncol. 2016;3:1393–8.
Cooperberg MR, Carroll PR. Trends in management for patients with localized prostate cancer, 1990−2013. JAMA. 2015;314:80–82.
Liu J, Womble PR, Merdan S, Miller DC, Montie JE, Denton BT. Factors influencing selection of active surveillance for localized prostate cancer. Urology. 2015;86:901–5.
Womble PR, Montie JE, Ye Z, Linsell SM, Lane BR, Miller DC. Contemporary use of initial active surveillance among men in Michigan with low-risk prostate cancer. Eur Urol. 2015;67:44–50.
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–74.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Sobin LG,M, Wittekind C. TNM classification of malignant tumours, 7th ed. New York: Wiley-Liss; 2009.
van den Bergh RC, Roemeling S, Roobol MJ, Roobol W, Schroder FH, Bangma CH. Prospective validation of active surveillance in prostate cancer: the PRIAS study. Eur Urol. 2007;52:1560–3.
Bul M, van den Bergh RC, Rannikko A, Valdagni R, Pickles T, Bangma CH, et al. Predictors of unfavourable repeat biopsy results in men participating in a prospective active surveillance program. Eur Urol. 2012;61:370–7.
Hoeks CM, Somford DM, van Oort IM, Vergunst H, Oddens JR, Smits GA, et al. Value of 3-T multiparametric magnetic resonance imaging and magnetic resonance-guided biopsy for early risk restratification in active surveillance of low-risk prostate cancer: a prospective multicenter cohort study. Invest Radiol. 2014;49:165–72.
Futterer JJ, Briganti A, De Visschere P, Emberton M, Giannarini G, Kirkham A, et al. Can clinically significant prostate cancer be detected with multiparametric magnetic resonance imaging? A systematic review of the literature. Eur Urol. 2015;68:1045–53.
Watts S, Leydon G, Eyles C, Moore CM, Richardson A, Birch B, et al. A quantitative analysis of the prevalence of clinical depression and anxiety in patients with prostate cancer undergoing active surveillance. BMJ Open. 2015;5:e006674.
Bayliss D, Duff J, Stricker P, Walker K. Decision-making in prostate cancer: active surveillance over other treatment options. Urol Nurs. 2016;36:141–9.
Acknowledgements
This work is part of the ProZIB initiative (acronym for: ProstaatkankerZorg In Beeld or Insight into Prostate Cancer Care), which was supported by the Dutch Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. van Leenders is a consultant for Roche, and has received financial research support of Astra Zeneca and Roche on bladder cancer-related studies. The other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jansen, H., van Oort, I.M., van Andel, G. et al. Immediate treatment vs. active-surveillance in very-low-risk prostate cancer: the role of patient-, tumour-, and hospital-related factors. Prostate Cancer Prostatic Dis 22, 337–343 (2019). https://doi.org/10.1038/s41391-018-0109-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0109-y
- Springer Nature Limited
This article is cited by
-
Kwaliteitsregistratie prostaatcarcinoom: ziekenhuisregistratie versus Nederlandse Kankerregistratie
Tijdschrift voor Urologie (2021)