Abstract
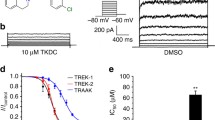
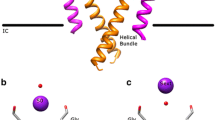
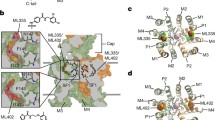
Potassium channels catalyze the selective transfer of potassium across the cell membrane and are essential for setting the resting potential in cells, controlling heart rate and modulating the firing pattern in neurons. Tetraethylammonium (TEA) blocks ion conduction through potassium channels in a voltage-dependent manner from both sides of the membrane. Here we show the structural basis of TEA blockade by cocrystallizing the prokaryotic potassium channel KcsA with two selective TEA analogs. TEA binding at both sites alters ion occupancy in the selectivity filter; these findings underlie the mutual destabilization and voltage-dependence of TEA blockade. We propose that TEA blocks potassium channels by acting as a potassium analog at the dehydration transition step during permeation.




Similar content being viewed by others
References
Hille, B. Ion Channels of Excitable Membranes (Sinauer, Sunderland, Massachusetts, USA, 2001).
Armstrong, C.M. Interaction of tetraethylammonium ion derivatives with the potassium channels of giant axons. J. Gen. Physiol. 58, 413–437 (1971).
Choi, K.L., Mossman, C., Aube, J. & Yellen, G. The internal quaternary ammonium receptor site of Shaker potassium channels. Neuron 10, 533–541 (1993).
MacKinnon, R. & Yellen, G. Mutations affecting TEA blockade and ion permeation in voltage-activated K+ channels. Science 250, 276–279 (1990).
Kavanaugh, M.P. et al. Interaction between tetraethylammonium and amino acid residues in the pore of cloned voltage-dependent potassium channels. J. Biol. Chem. 266, 7583–7587 (1991).
Heginbotham, L., LeMasurier, M., Kolmakova-Partensky, L. & Miller, C. Single streptomyces lividans K+ channels: functional asymmetries and sidedness of proton activation. J. Gen. Physiol. 114, 551–560 (1999).
Heginbotham, L. & MacKinnon, R. The aromatic binding site for tetraethylammonium ion on potassium channels. Neuron 8, 483–491 (1992).
Crouzy, S., Berneche, S. & Roux, B. Extracellular blockade of K+ channels by TEA: results from molecular dynamics simulations of the KcsA channel. J. Gen. Physiol. 118, 207–217 (2001).
Luzhkov, V.B. & Aqvist, J. Mechanisms of tetraethylammonium ion block in the KcsA potassium channel. FEBS Lett. 495, 191–196 (2001).
Guidoni, L. & Carloni, P. Tetraethylammonium binding to the outer mouth of the KcsA potassium channel: implications for ion permeation. J. Recept. Signal Transduct. Res. 22, 315–331 (2002).
Woodhull, A.M. Ionic blockage of sodium channels in nerve. J. Gen. Physiol. 61, 687–708 (1973).
Spassova, M. & Lu, Z. Coupled ion movement underlies rectification in an inward-rectifier K+ channel. J. Gen. Physiol. 112, 211–221 (1998).
Thompson, J. & Begenisich, T. External TEA block of Shaker K+ channels is coupled to the movement of K+ ions within the selectivity filter. J. Gen. Physiol. 122, 239–246 (2003).
Kutluay, E., Roux, B. & Heginbotham, L. Rapid intracellular TEA block of the KcsA potassium channel. Biophys. J. 88, 1018–1029 (2005).
Zhou, M., Morais-Cabral, J.H., Mann, S. & MacKinnon, R. Potassium channel receptor site for the inactivation gate and quaternary amine inhibitors. Nature 411, 657–661 (2001).
Newland, C.F., Adelman, J.P., Tempel, B.L. & Almers, W. Repulsion between tetraethyl-ammonium ions in cloned voltage-gated potassium channels. Neuron 8, 975–982 (1992).
Thompson, J. & Begenisich, T. Interaction between quaternary ammonium ions in the pore of potassium channels. Evidence against an electrostatic repulsion mechanism. J. Gen. Physiol. 115, 769–782 (2000).
Grissmer, S. & Cahalan, M. TEA prevents inactivation while blocking open K+ channels in human T lymphocytes. Biophys. J. 55, 203–206 (1989).
Choi, K.L., Aldrich, R.W. & Yellen, G. Tetraethylammonium blockade distinguishes two inactivation mechanisms in voltage-activated K+ channels. Proc. Natl. Acad. Sci. USA 88, 5092–5095 (1991).
Hoshi, T., Zagotta, W.N. & Aldrich, R.W. Two types of inactivation in Shaker K+ channels: effects of alterations in the carboxy-terminal region. Neuron 7, 547–556 (1991).
Yellen, G., Sodickson, D., Chen, T.Y. & Jurman, M.E. An engineered cysteine in the external mouth of a K+ channel allows inactivation to be modulated by metal binding. Biophys. J. 66, 1068–1075 (1994).
Liu, Y., Jurman, M.E. & Yellen, G. Dynamic rearrangement of the outer mouth of a K+ channel during gating. Neuron 16, 859–867 (1996).
Ogielska, E.M. & Aldrich, R.W. Functional consequences of a decreased potassium affinity in a potassium channel pore. Ion interactions and C-type inactivation. J. Gen. Physiol. 113, 347–358 (1999).
Lopez-Barneo, J., Hoshi, T., Heinemann, S.H. & Aldrich, R.W. Effects of external cations and mutations in the pore region on C-type inactivation of Shaker potassium channels. Receptors Channels 1, 61–71 (1993).
Ogielska, E.M. et al. Cooperative subunit interactions in C-type inactivation of K channels. Biophys. J. 69, 2449–2457 (1995).
Baukrowitz, T. & Yellen, G. Modulation of K+ current by frequency and external [K+]: a tale of two inactivation mechanisms. Neuron 15, 951–960 (1995).
Baukrowitz, T. & Yellen, G. Use-dependent blockers and exit rate of the last ion from the multi-ion pore of a K+ channel. Science 271, 653–656 (1996).
Zhou, Y., Morais-Cabral, J.H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel–Fab complex at 2.0 Å resolution. Nature 414, 43–48 (2001).
Morais-Cabral, J.H., Zhou, Y. & MacKinnon, R. Energetic optimization of ion conduction rate by the K+ selectivity filter. Nature 414, 37–42 (2001).
Zhou, Y. & MacKinnon, R. The occupancy of ions in the K+ selectivity filter: charge balance and coupling of ion binding to a protein conformational change underlie high conduction rates. J. Mol. Biol. 333, 965–975 (2003).
Roux, B. & MacKinnon, R. The cavity and pore helices in the KcsA K+ channel: electro-static stabilization of monovalent cations. Science 285, 100–102 (1999).
Zhou, Y. & MacKinnon, R. Ion binding affinity in the cavity of the KcsA potassium channel. Biochemistry 43, 4978–4982 (2004).
Berneche, S. & Roux, B. Energetics of ion conduction through the K+ channel. Nature 414, 73–77 (2001).
Thoden, J.B. et al. Carbamoyl phosphate synthetase: caught in the act of glutamine hydrolysis. Biochemistry 37, 8825–8831 (1998).
Ikeda, S.R. & Korn, S.J. Influence of permeating ions on potassium channel block by external tetraethylammonium. J. Physiol. 486, 267–272 (1995).
Lu, Z., Klem, A.M. & Ramu, Y. Ion conduction pore is conserved among potassium channels. Nature 413, 809–813 (2001).
Doyle, D.A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Friedrich, M.E.P. & Marvel, C.S. Reaction between alkali metal alkyls and quaternary arsonium compounds. J. Am. Chem. Soc. 52, 376–384 (1930).
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Crystallog. 26, 795–800 (1993).
Brunger, A.T. et al. Crystallography & NMR system: a new software suite for macro-molecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallog. 24, 946–950 (1991).
Merritt, E.A. & Bacon, D.J. Raster3D: photorealistic molecular graphics. Methods Enzymol. 277, 505–524 (1997).
Acknowledgements
We thank J. Cieslak, D. Freymann, C. LaBonne, K. Swartz and J. Yeh for critical reading of the manuscript, and Z. Wawrzak for assistance with data collection. This work was supported by US National Institutes of Health grant GM58568 to A.G. Support from the R.H. Lurie Comprehensive Cancer Center of Northwestern University to the Structural Biology Facility is acknowledged. DND-CAT is supported by DuPont, Dow, the US National Science Foundation and the State of Illinois. Portions of this work were carried out at BioCARS (sector 14) and the Structural Biology Center (sector 19) at the Advanced Photon Source (APS). Use of the APS is supported by the US Department of Energy (contract W-31-109-Eng-38).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lenaeus, M., Vamvouka, M., Focia, P. et al. Structural basis of TEA blockade in a model potassium channel. Nat Struct Mol Biol 12, 454–459 (2005). https://doi.org/10.1038/nsmb929
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb929
- Springer Nature America, Inc.
This article is cited by
-
Calcium-gated potassium channel blockade via membrane-facing fenestrations
Nature Chemical Biology (2023)
-
Potassium channel selectivity filter dynamics revealed by single-molecule FRET
Nature Chemical Biology (2019)
-
Distinct patterns of exocytosis elicited by Ca2+, Sr2+ and Ba2+ in bovine chromaffin cells
Pflügers Archiv - European Journal of Physiology (2018)
-
Unambiguous observation of blocked states reveals altered, blocker-induced, cardiac ryanodine receptor gating
Scientific Reports (2016)
-
Structural and functional characterization of a calcium-activated cation channel from Tsukamurella paurometabola
Nature Communications (2016)