Abstract
Osteoarthritis (OA), a prevalent chronic condition with a striking impact on quality of life, represents an enormous societal burden that increases greatly as populations age. Yet no approved pharmacological intervention, biologic therapy or procedure prevents the progressive destruction of the OA joint. Mesenchymal stem cells (MSCs)—multipotent precursors of connective tissue cells that can be isolated from many adult tissues, including those of the diarthrodial joint—have emerged as a potential therapy. Endogenous MSCs contribute to maintenance of healthy tissues by acting as reservoirs of repair cells or as immunomodulatory sentinels to reduce inflammation. The onset of degenerative changes in the joint is associated with aberrant activity or depletion of these cell reservoirs, leading to loss of chondrogenic potential and preponderance of a fibrogenic phenotype. Local delivery of ex vivo cultures of MSCs has produced promising outcomes in preclinical models of joint disease. Mechanistically, paracrine signalling by MSCs might be more important than differentiation in stimulating repair responses; thus, paracrine factors must be assessed as measures of MSC therapeutic potency, to replace traditional assays based on cell-surface markers and differentiation. Several early-stage clinical trials, initiated or underway in 2013, are testing the delivery of MSCs as an intra-articular injection into the knee, but optimal dose and vehicle are yet to be established.
Key Points
-
Osteoarthritis (OA) is associated with progressive and irreversible destruction of joint tissues with no defined aetiology
-
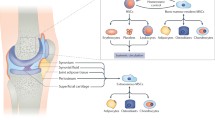
All joint tissues contain resident populations of mesenchymal stem cells (MSCs) capable of differentiating into cartilage, bone and other tissues
-
OA seems to be associated with changes in the quantity, phenotype, and differentiation potential of resident MSCs
-
Transplantation of ex vivo preparations of MSCs to the OA joint can evoke a therapeutically useful repair response in animal models of the disease
-
The repair effect mediated by delivered MSCs seems to arise as a result of paracrine responses
-
Early-stage clinical trials, initiated or underway in 2013, are testing intra-articular injection of MSCs, mostly without scaffold in the knee, but the optimal dose and vehicle have not been established



Similar content being viewed by others
References
Buckwalter, J. A. & Martin, J. A. Osteoarthritis. Adv. Drug Deliv. Rev. 58, 150–167 (2006).
Michaud, C. M. et al. The burden of disease and injury in the United States 1996. Popul. Health Metr. 4, 11 (2006).
Lawrence, R. C. et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 58, 26–35 (2008).
Gore, M., Tai, K. S., Sadosky, A., Leslie, D. & Stacey, B. R. Clinical comorbidities, treatment patterns, and direct medical costs of patients with osteoarthritis in usual care: a retrospective claims database analysis. J. Med. Econ. 14, 497–507 (2011).
McKenna, M. T., Michaud, C. M., Murray, C. J. & Marks, J. S. Assessing the burden of disease in the United States using disability-adjusted life years. Am. J. Prev. Med. 28, 415–423 (2005).
Guilak, F. Biomechanical factors in osteoarthritis. Best Pract. Res. Clin. Rheumatol. 25, 815–823 (2011).
Findlay, D. M. If good things come from above, do bad things come from below? Arthritis Res. Ther. 12, 119 (2010).
Goldring, M. B. & Goldring, S. R. Articular cartilage and subchondral bone in the pathogenesis of osteoarthritis. Ann. NY Acad. Sci. 1192, 230–237 (2010).
de Lange-Brokaar, B. J. et al. Synovial inflammation, immune cells and their cytokines in osteoarthritis: a review. Osteoarthritis Cartilage 20, 1484–1499 (2012).
Ayral, X., Pickering, E. H., Woodworth, T. G., Mackillop, N. & Dougados, M. Synovitis: a potential predictive factor of structural progression of medial tibiofemoral knee osteoarthritis—results of a 1 year longitudinal arthroscopic study in 422 patients. Osteoarthritis Cartilage 13, 361–367 (2005).
Benito, M. J., Veale, D. J., FitzGerald, O., van den Berg, W. B. & Bresnihan, B. Synovial tissue inflammation in early and late osteoarthritis. Ann. Rheum. Dis. 64, 1263–1267 (2005).
Buckwalter, J. A., Saltzman, C. & Brown, T. The impact of osteoarthritis: implications for research. Clin. Orthop. Relat. Res. 427 (Suppl.), S6–S15 (2004).
Kurtz, S., Ong, K., Lau, E., Mowat, F. & Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 89, 780–785 (2007).
Friedenstein, A. J., Piatetzky, S. & Petrakova, K. V. Osteogenesis in transplants of bone marrow cells. J. Embryol. Exp. Morphol. 16, 381–390 (1966).
Friedenstein, A. J., Chailakhjan, R. K. & Lalykina, K. S. The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Tissue Kinet. 3, 393–403 (1970).
Friedenstein, A. J., Chailakhyan, R. K., Latsinik, N. V., Panasyuk, A. F. & Keiliss-Borok, I. V. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation 17, 331–340 (1974).
Friedenstein, A. J. Marrow stromal fibroblasts. Calcif. Tissue Int. 56 (Suppl. 1), S17 (1995).
Owen, M. & Friedenstein, A. J. Stromal stem cells: marrow-derived osteogenic precursors. Ciba Found. Symp. 136, 42–60 (1988).
Bunnell, B. A., Estes, B. T., Guilak, F. & Gimble, J. M. Differentiation of adipose stem cells. Methods Mol. Biol. 456, 155–171 (2008).
Gimble, J. M., Katz, A. J. & Bunnell, B. A. Adipose-derived stem cells for regenerative medicine. Circ. Res. 100, 1249–1260 (2007).
Meliga, E., Strem, B. M., Duckers, H. J. & Serruys, P. W. Adipose-derived cells. Cell Transplant. 16, 963–970 (2007).
Crisan, M. et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3, 301–313 (2008).
Troyer, D. L. & Weiss, M. L. Wharton's jelly-derived cells are a primitive stromal cell population. Stem Cells 26, 591–599 (2008).
Weiss, M. L. et al. Human umbilical cord matrix stem cells: preliminary characterization and effect of transplantation in a rodent model of Parkinson's disease. Stem Cells 24, 781–792 (2006).
Weiss, M. L. & Troyer, D. L. Stem cells in the umbilical cord. Stem Cell Rev. 2, 155–162 (2006).
Flynn, A., Barry, F. & O'Brien, T. UC blood-derived mesenchymal stromal cells: an overview. Cytotherapy 9, 717–726 (2007).
Barry, F. P. & Murphy, J. M. Mesenchymal stem cells: clinical applications and biological characterization. Int. J. Biochem. Cell Biol. 36, 568–584 (2004).
Delorme, B. & Charbord, P. Culture and characterization of human bone marrow mesenchymal stem cells. Methods Mol. Med. 140, 67–81 (2007).
Bianco, P., Robey, P. G. & Simmons, P. J. Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell 2, 313–319 (2008).
Sacchetti, B. et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131, 324–336 (2007).
Tavian, M. et al. The vascular wall as a source of stem cells. Ann. NY Acad. Sci. 1044, 41–50 (2005).
Zannettino, A. C. et al. Multipotential human adipose-derived stromal stem cells exhibit a perivascular phenotype in vitro and in vivo. J. Cell. Physiol. 214, 413–421 (2008).
Caplan, A. I. All MSCs are pericytes? Cell Stem Cell 3, 229–230 (2008).
Dulauroy, S., Di Carlo, S. E., Langa, F., Eberl, G. & Peduto, L. Lineage tracing and genetic ablation of ADAM12+ perivascular cells identify a major source of profibrotic cells during acute tissue injury. Nat. Med. 18, 1262–1270 (2012).
Kurth, T. B. et al. Functional mesenchymal stem cell niches in adult mouse knee joint synovium in vivo. Arthritis Rheum. 63, 1289–1300 (2011).
De Bari, C., Dell'Accio, F., Tylzanowski, P. & Luyten, F. P. Multipotent mesenchymal stem cells from adult human synovial membrane. Arthritis Rheum. 44, 1928–1942 (2001).
Karystinou, A. et al. Distinct mesenchymal progenitor cell subsets in the adult human synovium. Rheumatology (Oxford) 48, 1057–1064 (2009).
De Bari, C. et al. Skeletal muscle repair by adult human mesenchymal stem cells from synovial membrane. J. Cell Biol. 160, 909–918 (2003).
Meng, J. et al. The contribution of human synovial stem cells to skeletal muscle regeneration. Neuromuscul. Disord. 20, 6–15 (2010).
Jones, E. A. et al. Synovial fluid mesenchymal stem cells in health and early osteoarthritis: detection and functional evaluation at the single-cell level. Arthritis Rheum. 58, 1731–1740 (2008).
Jones, E. A. et al. Enumeration and phenotypic characterization of synovial fluid multipotential mesenchymal progenitor cells in inflammatory and degenerative arthritis. Arthritis Rheum. 50, 817–827 (2004).
Sekiya, I. et al. Human mesenchymal stem cells in synovial fluid increase in the knee with degenerated cartilage and osteoarthritis. J. Orthop. Res. 30, 943–949 (2012).
Morito, T. et al. Synovial fluid-derived mesenchymal stem cells increase after intra-articular ligament injury in humans. Rheumatology (Oxfo rd) 47, 1137–1143 (2008).
Lee, D. H. et al. Synovial fluid CD34− CD44+ CD90+ mesenchymal stem cell levels are associated with the severity of primary knee osteoarthritis. Osteoarthritis Cartilage 20, 106–109 (2012).
Sakaguchi, Y., Sekiya, I., Yagishita, K. & Muneta, T. Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum. 52, 2521–2529 (2005).
Fan, J., Varshney, R. R., Ren, L., Cai, D. & Wang, D. A. Synovium-derived mesenchymal stem cells: a new cell source for musculoskeletal regeneration. Tissue Eng. Part B Rev. 15, 75–86 (2009).
Khan, W. S., Adesida, A. B., Tew, S. R., Longo, U. G. & Hardingham, T. E. Fat pad-derived mesenchymal stem cells as a potential source for cell-based adipose tissue repair strategies. Cell Prolif. 45, 111–120 (2012).
Dowthwaite, G. P. et al. The surface of articular cartilage contains a progenitor cell population. J. Cell Sci. 117, 889–897 (2004).
Khan, I. M., Bishop, J. C., Gilbert, S. & Archer, C. W. Clonal chondroprogenitors maintain telomerase activity and Sox9 expression during extended monolayer culture and retain chondrogenic potential. Osteoarthritis Cartilage 17, 518–528 (2009).
Williams, R. et al. Identification and clonal characterisation of a progenitor cell sub-population in normal human articular cartilage. PLoS ONE 5, e13246 (2010).
Alsalameh, S., Amin, R., Gemba, T. & Lotz, M. Identification of mesenchymal progenitor cells in normal and osteoarthritic human articular cartilage. Arthritis Rheum. 50, 1522–1532 (2004).
Grogan, S. P., Miyaki, S., Asahara, H., D'Lima, D. D. & Lotz, M. K. Mesenchymal progenitor cell markers in human articular cartilage: normal distribution and changes in osteoarthritis. Arthritis Res. Ther. 11, R85 (2009).
Fickert, S., Fiedler, J. & Brenner, R. E. Identification of subpopulations with characteristics of mesenchymal progenitor cells from human osteoarthritic cartilage using triple staining for cell surface markers. Arthritis Res. Ther. 6, R422–R432 (2004).
Huang, T. F. et al. Isolation and characterization of mesenchymal stromal cells from human anterior cruciate ligament. Cytotherapy 10, 806–814 (2008).
Steinert, A. F. et al. Mesenchymal stem cell characteristics of human anterior cruciate ligament outgrowth cells. Tissue Eng. Part A 17, 1375–1388 (2011).
Murray, M. M., Bennett, R., Zhang, X. & Spector, M. Cell outgrowth from the human ACL in vitro: regional variation and response to TGF-β1. J. Orthop. Res. 20, 875–880 (2002).
Murray, M. M. & Spector, M. The migration of cells from the ruptured human anterior cruciate ligament into collagen-glycosaminoglycan regeneration templates in vitro. Biomaterials 22, 2393–2402 (2001).
Cheng, M. T., Yang, H. W., Chen, T. H. & Lee, O. K. Isolation and characterization of multipotent stem cells from human cruciate ligaments. Cell Prolif. 42, 448–460 (2009).
Segawa, Y. et al. Mesenchymal stem cells derived from synovium, meniscus, anterior cruciate ligament, and articular chondrocytes share similar gene expression profiles. J. Orthop. Res. 27, 435–441 (2009).
Park, D. et al. Endogenous bone marrow MSCs are dynamic, fate-restricted participants in bone maintenance and regeneration. Cell Stem Cell 10, 259–272 (2012).
Rinkevich, Y., Lindau, P., Ueno, H., Longaker, M. T. & Weissman, I. L. Germ-layer and lineage-restricted stem/progenitors regenerate the mouse digit tip. Nature 476, 409–413 (2011).
Mendez-Ferrer, S. et al. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 466, 829–834 (2010).
Flannery, C. R. et al. Articular cartilage superficial zone protein (SZP) is homologous to megakaryocyte stimulating factor precursor and Is a multifunctional proteoglycan with potential growth-promoting, cytoprotective, and lubricating properties in cartilage metabolism. Biochem. Biophys. Res. Commun. 254, 535–541 (1999).
Koyama, E. et al. A distinct cohort of progenitor cells participates in synovial joint and articular cartilage formation during mouse limb skeletogenesis. Dev. Biol. 316, 62–73 (2008).
Lee, C. H. et al. Regeneration of the articular surface of the rabbit synovial joint by cell homing: a proof of concept study. Lancet 376, 440–448 (2010).
Murphy, J. M. et al. Reduced chondrogenic and adipogenic activity of mesenchymal stem cells from patients with advanced osteoarthritis. Arthritis Rheum. 46, 704–713 (2002).
Scharstuhl, A. et al. Chondrogenic potential of human adult mesenchymal stem cells is independent of age or osteoarthritis etiology. Stem Cells 25, 3244–3251 (2007).
Im, G. I., Jung, N. H. & Tae, S. K. Chondrogenic differentiation of mesenchymal stem cells isolated from patients in late adulthood: the optimal conditions of growth factors. Tissue Eng. 12, 527–536 (2006).
Coutu, D. L., Francois, M. & Galipeau, J. Inhibition of cellular senescence by developmentally regulated FGF receptors in mesenchymal stem cells. Blood 117, 6801–6812 (2011).
De Bari, C., Dell'Accio, F. & Luyten, F. P. Human periosteum-derived cells maintain phenotypic stability and chondrogenic potential throughout expansion regardless of donor age. Arthritis Rheum. 44, 85–95 (2001).
Jones, E. et al. Large-scale extraction and characterization of CD271+ multipotential stromal cells from trabecular bone in health and osteoarthritis: implications for bone regeneration strategies based on uncultured or minimally cultured multipotential stromal cells. Arthritis Rheum. 62, 1944–1954 (2010).
Hiraoka, K., Grogan, S., Olee, T. & Lotz, M. Mesenchymal progenitor cells in adult human articular cartilage. Biorheology 43, 447–454 (2006).
Lotz, M. K. et al. Cartilage cell clusters. Arthritis Rheum. 62, 2206–2218 (2010).
Koelling, S. et al. Migratory chondrogenic progenitor cells from repair tissue during the later stages of human osteoarthritis. Cell Stem Cell 4, 324–335 (2009).
Blaney Davidson, E. N. et al. Elevated extracellular matrix production and degradation upon bone morphogenetic protein-2 (BMP-2) stimulation point toward a role for BMP-2 in cartilage repair and remodeling. Arthritis Res. Ther. 9, R102 (2007).
Guo, X., Thomas, A. & Pirkko, L. A study on abnormal chondrocyte differentiation and abnormal expression of collagen types in articular cartilage from patients with Kaschin-Beck disease [Chinese]. Zhonghua Bing Li Xue Za Zhi 27, 19–22 (1998).
Guo, X. et al. Abnormal expression of Col X, PTHrP, TGF-β, bFGF, and VEGF in cartilage with Kashin-Beck disease. J. Bone Miner. Metab. 24, 319–328 (2006).
Pufe, T., Bartscher, M., Petersen, W., Tillmann, B. & Mentlein, R. Pleiotrophin, an embryonic differentiation and growth factor, is expressed in osteoarthritis. Osteoarthritis Cartilage 11, 260–264 (2003).
Plaas, A. et al. The relationship between fibrogenic TGFβ1 signaling in the joint and cartilage degradation in post-injury osteoarthritis. Osteoarthritis Cartilage 19, 1081–1090 (2011).
Sandy, J. D., Neame, P. J., Boynton, R. E. & Flannery, C. R. Catabolism of aggrecan in cartilage explants. Identification of a major cleavage site within the interglobular domain. J. Biol. Chem. 266, 8683–8685 (1991).
Glasson, S. S. et al. Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434, 644–648 (2005).
Stanton, H. et al. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434, 648–652 (2005).
Li, J. et al. Knockout of ADAMTS5 does not eliminate cartilage aggrecanase activity but abrogates joint fibrosis and promotes cartilage aggrecan deposition in murine osteoarthritis models. J. Orthop. Res. 29, 516–522 (2011).
Velasco, J. et al. Adamts5 deletion blocks murine dermal repair through CD44-mediated aggrecan accumulation and modulation of transforming growth factor β 1 (TGF β 1) signaling. J. Biol. Chem. 286, 26016–26027 (2011).
Zhang, Y. W. et al. Targeted disruption of Mig-6 in the mouse genome leads to early onset degenerative joint disease. Proc. Natl Acad. Sci. USA 102, 11740–11745 (2005).
Blaney Davidson, E. N. et al. Resemblance of osteophytes in experimental osteoarthritis to transforming growth factor beta-induced osteophytes: limited role of bone morphogenetic protein in early osteoarthritic osteophyte formation. Arthritis Rheum. 56, 4065–4073 (2007).
van der Kraan, P. M., Goumans, M. J., Blaney Davidson, E. & ten Dijke, P. Age-dependent alteration of TGF- β signalling in osteoarthritis. Cell Tissue Res. 347, 257–265 (2012).
Anitua, E. et al. Relationship between investigative biomarkers and radiographic grading in patients with knee osteoarthritis. Int. J. Rheumatol. 747432 (2009).
Matsumoto, T. et al. Articular cartilage repair with autologous bone marrow mesenchymal cells. J. Cell. Physiol. 225, 291–295 (2010).
Wakitani, S. et al. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J. Bone Joint Surg. Am. 76, 579–592 (1994).
Wakitani, S. et al. Repair of large full-thickness articular cartilage defects with allograft articular chondrocytes embedded in a collagen gel. Tissue Eng. 4, 429–444 (1998).
Murphy, J. M., Fink, D. J., Hunziker, E. B. & Barry, F. P. Stem cell therapy in a caprine model of osteoarthritis. Arthritis Rheum. 48, 3464–3474 (2003).
Diekman, B. O. et al. Intra-articular delivery of purified mesenchymal stem cells from C57BL/6 or MRL/MpJ superhealer mice prevents post-traumatic arthritis. Cell Transplant. http://dx.doi.org/10.3727/096368912X653264
Toghraie, F. et al. Scaffold-free adipose-derived stem cells (ASCs) improve experimentally induced osteoarthritis in rabbits. Arch. Iran. Med. 15, 495–499 (2012).
Horie, M. et al. Intra-articular injection of human mesenchymal stem cells (MSCs) promote rat meniscal regeneration by being activated to express Indian hedgehog that enhances expression of type II collagen. Osteoarthritis Cartilage 20, 1197–1207 (2012).
Sato, M. et al. Direct transplantation of mesenchymal stem cells into the knee joints of Hartley strain guinea pigs with spontaneous osteoarthritis. Arthritis Res. Ther. 14, R31 (2012).
Al Faqeh, H., Nor Hamdan, B. M., Chen, H. C., Aminuddin, B. S. & Ruszymah, B. H. The potential of intra-articular injection of chondrogenic-induced bone marrow stem cells to retard the progression of osteoarthritis in a sheep model. Exp. Gerontol. 47, 458–464 (2012).
Guercio, A. et al. Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol. Int. 36, 189–194 (2012).
Frisbie, D. D., Kisiday, J. D., Kawcak, C. E., Werpy, N. M. & McIlwraith, C. W. Evaluation of adipose-derived stromal vascular fraction or bone marrow-derived mesenchymal stem cells for treatment of osteoarthritis. J. Orthop. Res. 27, 1675–1680 (2009).
ter Huurne, M. et al. Antiinflammatory and chondroprotective effects of intraarticular injection of adipose-derived stem cells in experimental osteoarthritis. Arthritis Rheum. 64, 3604–3613 (2012).
Johnson, K. et al. A stem cell-based approach to cartilage repair. Science 336, 717–721 (2012).
Wang, Y. et al. Runx1/AML1/Cbfa2 mediates onset of mesenchymal cell differentiation toward chondrogenesis. J. Bone Miner. Res. 20, 1624–1636 (2005).
Wotton, S. et al. Gene array analysis reveals a common Runx transcriptional programme controlling cell adhesion and survival. Oncogene 27, 5856–5866 (2008).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2013).
Dominici M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Acknowledgements
The authors acknowledge funding support from Science Foundation Ireland (grant number 09/SRC/B1794), the European Union's 7th Framework Programme (grant numbers HEALTH-2007-B-223298 [PurStem], HEALTH-2009-1.4-3-241719 [ADIPOA] and NMP3-SL-2010-245993 [GAMBA]), and the Health Research Board of Ireland.
Author information
Authors and Affiliations
Contributions
Both authors made substantial contributions to researching data for the article, discussions of content, writing the article, and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
F. Barry declares that he owns stocks in Osiris Therapeutics and Orbsen Therapeutics, and is a Director of Orbsen Therapeutics. M. Murphy declares that she owns stocks in Osiris Therapeutics.
Rights and permissions
About this article
Cite this article
Barry, F., Murphy, M. Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol 9, 584–594 (2013). https://doi.org/10.1038/nrrheum.2013.109
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.109
- Springer Nature Limited
This article is cited by
-
Mesenchymal Stem Cell-Enriched Hydrogels for the Treatment of Knee Joint Disorders: A Systematic Review
Regenerative Engineering and Translational Medicine (2024)
-
The influence of inflammation on the characteristics of adipose-derived mesenchymal stem cells (ADMSCs) and tissue repair capability in a hepatic injury mouse model
Stem Cell Research & Therapy (2023)
-
Mesenchymal progenitor cells from non-inflamed versus inflamed synovium post-ACL injury present with distinct phenotypes and cartilage regeneration capacity
Stem Cell Research & Therapy (2023)
-
Construction of ultrasonically treated collagen/silk fibroin composite scaffolds to induce cartilage regeneration
Scientific Reports (2023)
-
Mesenchymal stem cell implantation provides short‐term clinical improvement and satisfactory cartilage restoration in patients with knee osteoarthritis but the evidence is limited: a systematic review performed by the early‐osteoarthritis group of ESSKA‐European knee associates section
Knee Surgery, Sports Traumatology, Arthroscopy (2023)