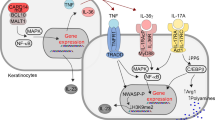
Abstract
Psoriasis is a common inflammatory skin disease of unknown etiology, for which there is no cure. This heterogeneous, cutaneous, inflammatory disorder is clinically characterized by prominent epidermal hyperplasia and a distinct inflammatory infiltrate. Crosstalk between immunocytes and keratinocytes, which results in the production of cytokines, chemokines and growth factors, is thought to mediate the disease. Given that psoriasis is only observed in humans, numerous genetic approaches to model the disease in mice have been undertaken. In this Review, we describe and critically assess the mouse models and transplantation experiments that have contributed to the discovery of novel disease-relevant pathways in psoriasis. Research performed using improved mouse models, combined with studies employing human cells, xenografts and patient material, will be key to our understanding of why such distinctive patterns of inflammation develop in patients with psoriasis. Indeed, a combination of genetic and immunological investigations will be necessary to develop both improved drugs for the treatment of psoriasis and novel curative strategies.
Key Points
-
Psoriasis is a common inflammatory skin disease that is genetically heterogeneous and for which there is currently no cure
-
Mouse models do not reproduce all aspects of psoriasis, but are nevertheless important tools to dissect the molecular and cellular events underlying the pathophysiology of the disease
-
Increased understanding of the complexity of psoriasis could be achieved by the development of improved mouse models and xenotransplantation studies involving cell transfer and tissue grafts
-
Novel strategies to treat psoriasis are likely to derive from the identification of new targets and the use of mouse models for drug testing


Similar content being viewed by others
References
Farber, E. M. & Nall, L. In Psoriasis 3rd edn (eds Roenigk, H. H. Jr & Maibach, H. I.) 107–157 (Marcel Dekker, Inc., New York, 1998).
Gudjonsson, J. E. & Elder, J. T. In Fitzpatrick's Dermatology in General Medicine 7th edn (eds Wolff, K. et al.) 169–193 (McGraw-Hill, New York, 2008).
Griffiths, C. E. & Barker, J. N. Pathogenesis and clinical features of psoriasis. Lancet 370, 263–271 (2007).
Weiss, G., Shemer, A. & Trau, H. The Koebner phenomenon: review of the literature. J. Eur. Acad. Dermatol. Venereol. 16, 241–248 (2002).
Blumberg, B. S., Bunim, J. J., Calkins, E., Pirani, C. L. & Zvaifler, N. J. Ara nomenclature and classification of arthritis and rheumatism (tentative). Arthritis Rheum. 7, 93–97 (1964).
Winchester, R. In Fitzpatrick's Dermatology in General Medicine 7th edn (eds Wolff, K. et al.) 194–207 (McGraw-Hill, New York, 2008).
Ibrahim, G., Waxman, R. & Helliwell, P. S. The prevalence of psoriatic arthritis in people with psoriasis. Arthritis Rheum. 61, 1373–1378 (2009).
McGonagle, D. Enthesitis: an autoinflammatory lesion linking nail and joint involvement in psoriatic disease. J. Eur. Acad. Dermatol. Venereol. 23 (Suppl. 1), 9–13 (2009).
Bovenschen, H. J., Seyger, M. M. & Van de Kerkhof, P. C. Plaque psoriasis vs. atopic dermatitis and lichen planus: a comparison for lesional T-cell subsets, epidermal proliferation and differentiation. Br. J. Dermatol. 153, 72–78 (2005).
Clark, R. A. Skin-resident T cells: the ups and downs of on site immunity. J. Invest. Dermatol. 130, 362–370 (2010).
Capon, F., Trembath, R. C. & Barker, J. N. An update on the genetics of psoriasis. Dermatol. Clin. 22, 339–347, vii (2004).
Duffin, K. C. & Krueger, G. G. Genetic variations in cytokines and cytokine receptors associated with psoriasis found by genome-wide association. J. Invest. Dermatol. 129, 827–833 (2009).
Gudjonsson, J. E. et al. Psoriasis patients who are homozygous for the HLA-Cw*0602 allele have a 2.5-fold increased risk of developing psoriasis compared with Cw6 heterozygotes. Br. J. Dermatol. 148, 233–235 (2003).
Nair, R. P. et al. Sequence and haplotype analysis supports HLA-C as the psoriasis susceptibility 1 gene. Am. J. Hum. Genet. 78, 827–851 (2006).
Nair, R. P. et al. Genome-wide scan reveals association of psoriasis with IL-23 and NF-kappaB pathways. Nat. Genet. 41, 199–204 (2009).
de Guzman Strong, C. et al. A milieu of regulatory elements in the epidermal differentiation complex syntenic block: implications for atopic dermatitis and psoriasis. Hum. Mol. Genet. 19, 1453–1460 (2010).
de Cid, R. et al. Deletion of the late cornified envelope LCE3B and LCE3C genes as a susceptibility factor for psoriasis. Nat. Genet. 41, 211–215 (2009).
Menter, A. The status of biologic therapies in the treatment of moderate to severe psoriasis. Cutis 84 (Suppl. 4), 14–24 (2009).
Menter, A. et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 1. Overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J. Am. Acad. Dermatol. 58, 826–850 (2008).
A study in patients with moderate to severe psoriasis. ClinicalTrials.gov identifier: NCT01107457 [online], (2010).
Assessment of long-term infliximab for psoriasis (study P05319) (REALITY). ClinicalTrials.gov identifier: NCT00779675 [online], (2010).
Lowes, M. A., Bowcock, A. M. & Krueger, J. G. Pathogenesis and therapy of psoriasis. Nature 445, 866–873 (2007).
Berking, C. et al. Photocarcinogenesis in human adult skin grafts. Carcinogenesis 23, 181–187 (2002).
Jameson, J. M., Sharp, L. L., Witherden, D. A. & Havran, W. L. Regulation of skin cell homeostasis by gamma delta T cells. Front. Biosci. 9, 2640–2651 (2004).
Khavari, P. A. Modelling cancer in human skin tissue. Nat. Rev. Cancer 6, 270–280 (2006).
Schön, M. P., Blume-Peytavi, U., Schön, M. & Orfanos, C. E. The human hair follicle: glycoprotein-related antigenic profile of distinct keratinocyte populations in vivo and their alterations in vitro. Arch. Dermatol. Res. 287, 591–598 (1995).
Nestle, F. O., Di Meglio, P., Qin, J. Z. & Nickoloff, B. J. Skin immune sentinels in health and disease. Nat. Rev. Immunol. 9, 679–691 (2009).
Blanpain, C. & Fuchs, E. Epidermal homeostasis: a balancing act of stem cells in the skin. Nat. Rev. Mol. Cell Biol. 10, 207–217 (2009).
Watt, F. M., Lo Celso, C. & Silva-Vargas, V. Epidermal stem cells: an update. Curr. Opin. Genet. Dev. 16, 518–524 (2006).
Gudjonsson, J. E., Johnston, A., Dyson, M., Valdimarsson, H. & Elder, J. T. Mouse models of psoriasis. J. Invest. Dermatol. 127, 1292–1308 (2007).
Richardson, S. K. & Gelfand, J. M. Update on the natural history and systemic treatment of psoriasis. Adv. Dermatol. 24, 171–196 (2008).
Johnson-Huang, L. M., McNutt, N. S., Krueger, J. G. & Lowes, M. A. Cytokine-producing dendritic cells in the pathogenesis of inflammatory skin diseases. J. Clin. Immunol. 29, 247–256 (2009).
Zaba, L. C., Krueger, J. G. & Lowes, M. A. Resident and “inflammatory” dendritic cells in human skin. J. Invest. Dermatol. 129, 302–308 (2009).
Ghoreschi, K., Weigert, C. & Röcken, M. Immunopathogenesis and role of T cells in psoriasis. Clin. Dermatol. 25, 574–580 (2007).
Di Cesare, A., Di Meglio, P. & Nestle, F. O. The IL-23/Th17 axis in the immunopathogenesis of psoriasis. J. Invest. Dermatol. 129, 1339–1350 (2009).
Conrad, C. et al. Alpha1beta1 integrin is crucial for accumulation of epidermal T cells and the development of psoriasis. Nat. Med. 13, 836–842 (2007).
Plant, D., Young, H. S., Watson, R. E., Worthington, J. & Griffiths, C. E. The CX3CL1-CX3CR1 system and psoriasis. Exp. Dermatol. 15, 900–903 (2006).
Sabat, R. et al. Immunopathogenesis of psoriasis. Exp. Dermatol. 16, 779–798 (2007).
Nestle, F. O., Kaplan, D. H. & Barker, J. Psoriasis. N. Engl. J. Med. 361, 496–509 (2009).
Conrad, C. & Nestle, F. O. Animal models of psoriasis and psoriatic arthritis: an update. Curr. Rheumatol. Rep. 8, 342–347 (2006).
Schön, M. P. Animal models of psoriasis—what can we learn from them? J. Invest. Dermatol. 112, 405–410 (1999).
Brown, W. R. & Hardy, M. H. A hypothesis on the cause of chronic epidermal hyperproliferation in asebia mice. Clin. Exp. Dermatol. 13, 74–77 (1988).
HogenEsch, H. et al. A spontaneous mutation characterized by chronic proliferative dermatitis in C57BL mice. Am. J. Pathol. 143, 972–982 (1993).
Sundberg, J. P. et al. Development and progression of psoriasiform dermatitis and systemic lesions in the flaky skin (fsn) mouse mutant. Pathobiology 65, 271–286 (1997).
Gijbels, M. J. et al. Therapeutic interventions in mice with chronic proliferative dermatitis (cpdm/cpdm). Exp. Dermatol. 9, 351–358 (2000).
Peters, T. et al. CD18 in monogenic and polygenic inflammatory processes of the skin. J. Investig. Dermatol. Symp. Proc. 11, 7–15 (2006).
Bullard, D. C. et al. A polygenic mouse model of psoriasiform skin disease in CD18-deficient mice. Proc. Natl Acad. Sci. USA 93, 2116–2121 (1996).
Sano, S. et al. Stat3 links activated keratinocytes and immunocytes required for development of psoriasis in a novel transgenic mouse model. Nat. Med. 11, 43–49 (2005).
Song, J. I. & Grandis, J. R. STAT signaling in head and neck cancer. Oncogene 19, 2489–2495 (2000).
Bartoli, M. et al. VEGF differentially activates STAT3 in microvascular endothelial cells. FASEB J. 17, 1562–1564 (2003).
Detmar, M. et al. Overexpression of vascular permeability factor/vascular endothelial growth factor and its receptors in psoriasis. J. Exp. Med. 180, 1141–1146 (1994).
Schonthaler, H. B., Huggenberger, R., Wculek, S. K., Detmar, M. & Wagner, E. F. Systemic anti-VEGF treatment strongly reduces skin inflammation in a mouse model of psoriasis. Proc. Natl Acad. Sci. USA 106, 21264–21269 (2009).
Doi, H., Shibata, M. A., Kiyokane, K. & Otsuki, Y. Downregulation of TGFbeta isoforms and their receptors contributes to keratinocyte hyperproliferation in psoriasis vulgaris. J. Dermatol. Sci. 33, 7–16 (2003).
Elder, J. T. et al. Overexpression of transforming growth factor alpha in psoriatic epidermis. Science 243, 811–814 (1989).
Kane, C. J., Knapp, A. M., Mansbridge, J. N. & Hanawalt, P. C. Transforming growth factor-beta 1 localization in normal and psoriatic epidermal keratinocytes in situ. J. Cell. Physiol. 144, 144–150 (1990).
Cui, W. et al. Concerted action of TGF-beta 1 and its type II receptor in control of epidermal homeostasis in transgenic mice. Genes Dev. 9, 945–955 (1995).
Fowlis, D. J., Cui, W., Johnson, S. A., Balmain, A. & Akhurst, R. J. Altered epidermal cell growth control in vivo by inducible expression of transforming growth factor beta 1 in the skin of transgenic mice. Cell Growth Differ. 7, 679–687 (1996).
Li, A. G., Wang, D., Feng, X. H. & Wang, X. J. Latent TGFbeta1 overexpression in keratinocytes results in a severe psoriasis-like skin disorder. EMBO J. 23, 1770–1781 (2004).
Liu, X. et al. Conditional epidermal expression of TGFbeta 1 blocks neonatal lethality but causes a reversible hyperplasia and alopecia. Proc. Natl Acad. Sci. USA 98, 9139–9144 (2001).
Sellheyer, K. et al. Inhibition of skin development by overexpression of transforming growth factor beta 1 in the epidermis of transgenic mice. Proc. Natl Acad. Sci. USA 90, 5237–5241 (1993).
Wang, X. J., Liefer, K. M., Tsai, S., O'Malley, B. W. & Roop, D. R. Development of gene-switch transgenic mice that inducibly express transforming growth factor beta1 in the epidermis. Proc. Natl Acad. Sci. USA 96, 8483–8488 (1999).
Wippel-Slupetzky, K. & Stingl, G. Future perspectives in the treatment of psoriasis. Curr. Probl. Dermatol. 38, 172–189 (2009).
Cheng, J. et al. Cachexia and graft-vs.-host-disease-type skin changes in keratin promoter-driven TNF alpha transgenic mice. Genes Dev. 6, 1444–1456 (1992).
Westergaard, M. et al. Expression and localization of peroxisome proliferator-activated receptors and nuclear factor kappaB in normal and lesional psoriatic skin. J. Invest. Dermatol. 121, 1104–1117 (2003).
Romanowska, M., Reilly, L., Palmer, C. N., Gustafsson, M. C. & Foerster, J. Activation of PPARbeta/delta causes a psoriasis-like skin disease in vivo. PLoS ONE 5, e9701 (2010).
A study of the safety and efficacy of CNTO 1275 in patients with severe plaque-type psoriasis. ClinicalTrials.gov identifier: NCT00267969 [online], (2010).
A study of the safety and efficacy of CNTO 1275 in subjects with moderate to severe psoriasis. ClinicalTrials.gov identifier: NCT00307437 [online], (2010).
A verification study of CNTO 1275 in patients with plaque psoriasis. ClinicalTrials.gov identifier: NCT00723528 [online], (2010).
Kopp, T. et al. Inflammatory skin disease in K14/p40 transgenic mice: evidence for interleukin-12-like activities of p40. J. Invest. Dermatol. 117, 618–626 (2001).
Tarutani, M. et al. Neutrophil-dominant psoriasis-like skin inflammation induced by epidermal-specific expression of Raf in mice. J. Dermatol. Sci. 58, 28–35 (2010).
Mee, J. B., Antonopoulos, C., Poole, S., Kupper, T. S. & Groves, R. W. Counter-regulation of interleukin-1alpha (IL-1alpha) and IL-1 receptor antagonist in murine keratinocytes. J. Invest. Dermatol. 124, 1267–1274 (2005).
Mee, J. B., Cork, M. J., di Giovine, F. S., Duff, G. W. & Groves, R. W. Interleukin-1: a key inflammatory mediator in psoriasis? Cytokine 33, 72–78 (2006).
Cooper, K. D. et al. IL-1 activity is reduced in psoriatic skin. Decreased IL-1 alpha and increased nonfunctional IL-1 beta. J. Immunol. 144, 4593–4603 (1990).
Shepherd, J., Little, M. C. & Nicklin, M. J. Psoriasis-like cutaneous inflammation in mice lacking interleukin-1 receptor antagonist. J. Invest. Dermatol. 122, 665–669 (2004).
Pasparakis, M. et al. TNF-mediated inflammatory skin disease in mice with epidermis-specific deletion of IKK2. Nature 417, 861–866 (2002).
Stratis, A. et al. Pathogenic role for skin macrophages in a mouse model of keratinocyte-induced psoriasis-like skin inflammation. J. Clin. Invest. 116, 2094–2104 (2006).
Koegel, H. et al. Loss of serum response factor in keratinocytes results in hyperproliferative skin disease in mice. J. Clin. Invest. 119, 899–910 (2009).
Mehic, D., Bakiri, L., Ghannadan, M., Wagner, E. F. & Tschachler, E. Fos and jun proteins are specifically expressed during differentiation of human keratinocytes. J. Invest. Dermatol. 124, 212–220 (2005).
Meixner, A. et al. Epidermal JunB represses G-CSF transcription and affects haematopoiesis and bone formation. Nat. Cell Biol. 10, 1003–1011 (2008).
Guinea-Viniegra, J. et al. TNFalpha shedding and epidermal inflammation are controlled by Jun proteins. Genes Dev. 23, 2663–2674 (2009).
Zenz, R. et al. Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature 437, 369–375 (2005).
Zenz, R. et al. Activator protein 1 (Fos/Jun) functions in inflammatory bone and skin disease. Arthritis Res. Ther. 10, 201 (2008).
Zheng, Y. et al. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature 445, 648–651 (2007).
Chan, J. R. et al. IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2-dependent mechanisms with implications for psoriasis pathogenesis. J. Exp. Med. 203, 2577–2587 (2006).
Caruso, R. et al. Involvement of interleukin-21 in the epidermal hyperplasia of psoriasis. Nat. Med. 15, 1013–1015 (2009).
van der Fits, L. et al. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J. Immunol. 182, 5836–5845 (2009).
Schön, M. P., Detmar, M. & Parker, C. M. Murine psoriasis-like disorder induced by naive CD4+ T cells. Nat. Med. 3, 183–188 (1997).
Leon, F. et al. Antibodies to complement receptor 3 treat established inflammation in murine models of colitis and a novel model of psoriasiform dermatitis. J. Immunol. 177, 6974–6982 (2006).
Boyman, O. et al. Spontaneous development of psoriasis in a new animal model shows an essential role for resident T cells and tumor necrosis factor-alpha. J. Exp. Med. 199, 731–736 (2004).
Chang, B. Y. et al. JAK3 inhibition significantly attenuates psoriasiform skin inflammation in CD18 mutant PL/J mice. J. Immunol. 183, 2183–2192 (2009).
Skvara, H. et al. The PKC inhibitor AEB071 may be a therapeutic option for psoriasis. J. Clin. Invest. 118, 3151–3159 (2008).
Sonkoly, E. et al. MicroRNAs: novel regulators involved in the pathogenesis of psoriasis? PLoS ONE 2, e610 (2007).
Zibert, J. R. et al. MicroRNAs and potential target interactions in psoriasis. J. Dermatol. Sci. 58, 177–185 (2010).
Zhang, X. J. et al. Psoriasis genome-wide association study identifies susceptibility variants within LCE gene cluster at 1q21. Nat. Genet. 41, 205–210 (2009).
Liu, Y. et al. A genome-wide association study of psoriasis and psoriatic arthritis identifies new disease loci. PLoS Genet. 4, e1000041 (2008).
Capon, F. et al. Identification of ZNF313/RNF114 as a novel psoriasis susceptibility gene. Hum. Mol. Genet. 17, 1938–1945 (2008).
Voskas, D. et al. A cyclosporine-sensitive psoriasis-like disease produced in Tie2 transgenic mice. Am. J. Pathol. 166, 843–855 (2005).
Vassar, R. & Fuchs, E. Transgenic mice provide new insights into the role of TGF-alpha during epidermal development and differentiation. Genes Dev. 5, 714–727 (1991).
Ishihara, K. & Hirano, T. IL-6 in autoimmune disease and chronic inflammatory proliferative disease. Cytokine Growth Factor Rev. 13, 357–368 (2002).
Guo, L., Yu, Q. C. & Fuchs, E. Targeting expression of keratinocyte growth factor to keratinocytes elicits striking changes in epithelial differentiation in transgenic mice. EMBO J. 12, 973–986 (1993).
Groves, R. W., Mizutani, H., Kieffer, J. D. & Kupper, T. S. Inflammatory skin disease in transgenic mice that express high levels of interleukin 1 alpha in basal epidermis. Proc. Natl Acad. Sci. USA 92, 11874–11878 (1995).
Blumberg, H. et al. Interleukin 20: discovery, receptor identification, and role in epidermal function. Cell 104, 9–19 (2001).
Xia, Y. P. et al. Transgenic delivery of VEGF to mouse skin leads to an inflammatory condition resembling human psoriasis. Blood 102, 161–168 (2003).
Cook, P. W., Brown, J. R., Cornell, K. A. & Pittelkow, M. R. Suprabasal expression of human amphiregulin in the epidermis of transgenic mice induces a severe, early-onset, psoriasis-like skin pathology: expression of amphiregulin in the basal epidermis is also associated with synovitis. Exp. Dermatol. 13, 347–356 (2004).
Blessing, M., Schirmacher, P. & Kaiser, S. Overexpression of bone morphogenetic protein-6 (BMP-6) in the epidermis of transgenic mice: inhibition or stimulation of proliferation depending on the pattern of transgene expression and formation of psoriatic lesions. J. Cell. Biol. 135, 227–239 (1996).
Carroll, J. M., Romero, M. R. & Watt, F. M. Suprabasal integrin expression in the epidermis of transgenic mice results in developmental defects and a phenotype resembling psoriasis. Cell 83, 957–968 (1995).
Cook, P. W. et al. Transgenic expression of the human amphiregulin gene induces a psoriasis-like phenotype. J. Clin. Invest. 100, 2286–2294 (1997).
Carroll, J. M., Crompton, T., Seery, J. P. & Watt, F. M. Transgenic mice expressing IFN-gamma in the epidermis have eczema, hair hypopigmentation, and hair loss. J. Invest. Dermatol. 108, 412–422 (1997).
Hobbs, R. M., Silva-Vargas, V., Groves, R. & Watt, F. M. Expression of activated MEK1 in differentiating epidermal cells is sufficient to generate hyperproliferative and inflammatory skin lesions. J. Invest. Dermatol. 123, 503–515 (2004).
Hida, S. et al. CD8(+) T cell-mediated skin disease in mice lacking IRF-2, the transcriptional attenuator of interferon-alpha/beta signaling. Immunity 13, 643–655 (2000).
Schön, M. P., Schön, M., Warren, H. B., Donohue, J. P. & Parker, C. M. Cutaneous inflammatory disorder in integrin alphaE (CD103)-deficient mice. J. Immunol. 165, 6583–6589 (2000).
Rebholz, B. et al. Crosstalk between keratinocytes and adaptive immune cells in an IkappaBalpha protein-mediated inflammatory disease of the skin. Immunity 27, 296–307 (2007).
Acknowledgements
The authors are very grateful to T. Luger and P. Lipsky for critically reading the manuscript and providing valuable suggestions. This work was supported by the European Research Council Advanced Grant (ERC FCK/2008/37) and the Banco Bilbao Vizcaya Argentaria (BBVA) Foundation.
Author information
Authors and Affiliations
Contributions
All authors contributed equally in researching data for the article, discussion of the content, writing the article, and both reviewing and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Wagner, E., Schonthaler, H., Guinea-Viniegra, J. et al. Psoriasis: what we have learned from mouse models. Nat Rev Rheumatol 6, 704–714 (2010). https://doi.org/10.1038/nrrheum.2010.157
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2010.157
- Springer Nature Limited
This article is cited by
-
Delivery of a sebum modulator by an engineered skin microbe in mice
Nature Biotechnology (2024)
-
A simple tool for evaluation of inflammation in psoriasis: Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio as markers in psoriasis patients and related murine models of psoriasis-like skin disease
Journal of Molecular Medicine (2024)
-
Elevated cholesteryl ester transfer and phospholipid transfer proteins aggravated psoriasis in imiquimod-induced mouse models
Lipids in Health and Disease (2022)
-
Single cell transcriptional zonation of human psoriasis skin identifies an alternative immunoregulatory axis conducted by skin resident cells
Cell Death & Disease (2021)
-
Immune responses and therapeutic options in psoriasis
Cellular and Molecular Life Sciences (2021)