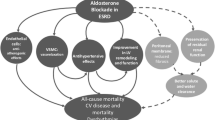
Abstract
Aldosterone, a steroid hormone with mineralocorticoid activity, is mainly recognized for its action on sodium reabsorption in the distal nephron of the kidney, which is mediated by the epithelial sodium channel (ENaC). Beyond this well-known action, however, aldosterone exerts other effects on the kidney, blood vessels and the heart, which can have pathophysiological consequences, particularly in the presence of a high salt intake. Aldosterone is implicated in renal inflammatory and fibrotic processes, as well as in podocyte injury and mesangial cell proliferation. In the cardiovascular system, aldosterone has specific hypertrophic and fibrotic effects and can alter endothelial function. Several lines of evidence support the existence of crosstalk between aldosterone and angiotensin II in vascular smooth muscle cells. The deleterious effects of aldosterone on the cardiovascular system require concomitant pathophysiological conditions such as a high salt diet, increased oxidative stress, or inflammation. Large interventional trials have confirmed the benefits of adding mineralocorticoid-receptor antagonists to standard therapy, in particular to angiotensin-converting-enzyme inhibitor and angiotensin II receptor blocker therapy, in patients with heart failure. Small interventional studies in patients with chronic kidney disease have shown promising results, with a significant reduction of proteinuria associated with aldosterone antagonism, but large interventional trials that test the efficacy and safety of mineralocorticoid-receptor antagonists in chronic kidney disease are needed.
Key Points
-
Beyond its role in the regulation of renal sodium reabsorption in the distal nephron, aldosterone may exert deleterious effects on the kidney and the cardiovascular system particularly in the presence of a high-salt diet
-
Aldosterone induces inflammation, fibrosis, mesangial cell proliferation and podocyte injury in the kidney
-
Aldosterone contributes to cardiovascular remodeling and fibrosis in animals on a high-salt diet
-
Large clinical trials have demonstrated the efficacy of mineralocorticoid-receptor antagonists in patients with heart failure
-
Despite encouraging results from small interventional studies, large interventional studies in patients with chronic kidney disease are needed to test the efficacy and safety of mineralocorticoid-receptor antagonists



Similar content being viewed by others
References
Loffing, J. & Korbmacher, C. Regulated sodium transport in the renal connecting tubule (CNT) via the epithelial sodium channel (ENaC). Pflugers Arch. 458, 111–135 (2009).
Bhalla, V., Soundararajan, R., Pao, A. C., Li, H. & Pearce, D. Disinhibitory pathways for control of sodium transport: regulation of ENaC by SGK1 and GILZ. Am. J. Physiol. Renal Physiol. 291, F714–F721 (2006).
Náray-Fejes-Tóth, A., Snyder, P. M. & Fejes-Tóth, G. The kidney-specific WNK1 isoform is induced by aldosterone and stimulates epithelial sodium channel-mediated Na+ transport. Proc. Natl Acad. Sci. USA 101, 17434–17439 (2004).
Rozansky, D. J. et al. Aldosterone mediates activation of the thiazide-sensitive Na-Cl cotransporter through an SGK1 and WNK4 signaling pathway. J. Clin. Invest. 119, 2601–2612 (2009).
Greene, E. L., Kren, S. & Hostetter, T. H. Role of aldosterone in the remnant kidney model in the rat. J. Clin. Invest. 98, 1063–1068 (1996).
Young, M., Fullerton, M., Dilley, R. & Funder, J. Mineralocorticoids, hypertension, and cardiac fibrosis. J. Clin. Invest. 93, 2578–2583 (1994).
Blasi, E. R. et al. Aldosterone/salt induces renal inflammation and fibrosis in hypertensive rats. Kidney Int. 63, 1791–1800 (2003).
Nishiyama, A. et al. Possible contributions of reactive oxygen species and mitogen-activated protein kinase to renal injury in aldosterone/salt-induced hypertensive rats. Hypertension 43, 841–848 (2004).
Funder, J. W., Pearce, P. T., Smith, R. & Campbell, J. Vascular type I aldosterone binding sites are physiological mineralocorticoid receptors. Endocrinology 125, 2224–2226 (1989).
Caprio, M. et al. Functional mineralocorticoid receptors in human vascular endothelial cells regulate intercellular adhesion molecule-1 expression and promote leukocyte adhesion. Circ. Res. 102, 1359–1367 (2008).
Lombès, M. et al. Immunohistochemical and biochemical evidence for a cardiovascular mineralocorticoid receptor. Circ. Res. 71, 503–510 (1992).
Jaffe, I. Z. & Mendelsohn, M. E. Angiotensin II and aldosterone regulate gene transcription via functional mineralocortocoid receptors in human coronary artery smooth muscle cells. Circ. Res. 96, 643–650 (2005).
Nishiyama, A. et al. Involvement of aldosterone and mineralocorticoid receptors in rat mesangial cell proliferation and deformability. Hypertension 45, 710–716 (2005).
Johar, S., Cave, A. C., Narayanapanicker, A., Grieve, D. J. & Shah, A. M. Aldosterone mediates angiotensin II-induced interstitial cardiac fibrosis via a Nox2-containing NADPH oxidase. FASEB J. 20, 1546–1548 (2006).
Rocha, R., Chander, P. N., Zuckerman, A. & Stier, C. T. Jr. Role of aldosterone in renal vascular injury in stroke-prone hypertensive rats. Hypertension 33, 232–237 (1999).
Pitt, B. et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N. Engl. J. Med. 348, 1309–1321 (2003).
Pitt, B. et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 341, 709–717 (1999).
Ma, J. et al. Plasminogen activator inhibitor-1 deficiency protects against aldosterone-induced glomerular injury. Kidney Int. 69, 1064–1072 (2006).
Shibata, S., Nagase, M., Yoshida, S., Kawachi, H. & Fujita, T. Podocyte as the target for aldosterone: roles of oxidative stress and Sgk1. Hypertension 49, 355–364 (2007).
Du, J. et al. Mineralocorticoid receptor blockade and calcium channel blockade have different renoprotective effects on glomerular and interstitial injury in rats. Am. J. Physiol. Renal Physiol. 297, F802–F808 (2009).
Aldigier, J. C., Kanjanbuch, T., Ma, L. J., Brown, N. J. & Fogo, A. B. Regression of existing glomerulosclerosis by inhibition of aldosterone. J. Am. Soc. Nephrol. 16, 3306–3314 (2005).
Fujisawa, G. et al. Spironolactone prevents early renal injury in streptozotocin-induced diabetic rats. Kidney Int. 66, 1493–1502 (2004).
Ikeda, H. et al. Spironolactone suppresses inflammation and prevents L-NAME-induced renal injury in rats. Kidney Int. 75, 147–155 (2009).
Siragy, H. M. & Xue, C. Local renal aldosterone production induces inflammation and matrix formation in kidneys of diabetic rats. Exp. Physiol. 93, 817–824 (2008).
Terada, Y. et al. Aldosterone-stimulated SGK1 activity mediates profibrotic signaling in the mesangium. J. Am. Soc. Nephrol. 19, 298–309 (2008).
Riser, B. L. et al. Cyclic stretching of mesangial cells up-regulates intercellular adhesion molecule-1 and leukocyte adherence: a possible new mechanism for glomerulosclerosis. Am. J. Pathol. 158, 11–17 (2001).
Huang, S., Zhang, A., Ding, G. & Chen, R. Aldosterone-induced mesangial cell proliferation is mediated by EGF receptor transactivation. Am. J. Physiol. Renal Physiol. 296, F1323–F1333 (2009).
Virdis, A. et al. Spironolactone improves angiotensin-induced vascular changes and oxidative stress. Hypertension 40, 504–510 (2002).
Beswick, R. A., Dorrance, A. M., Leite, R. & Webb, R. C. NADH/NADPH oxidase and enhanced superoxide production in the mineralocorticoid hypertensive rat. Hypertension 38, 1107–1111 (2001).
Onozato, M. L. et al. Dual blockade of aldosterone and angiotensin II additively suppresses TGF-beta and NADPH oxidase in the hypertensive kidney. Nephrol. Dial. Transplant. 22, 1314–1322 (2007).
Beswick, R. A. et al. Long-term antioxidant administration attenuates mineralocorticoid hypertension and renal inflammatory response. Hypertension 37, 781–786 (2001).
Nagai, Y. et al. Aldosterone stimulates collagen gene expression and synthesis via activation of ERK1/2 in rat renal fibroblasts. Hypertension 46, 1039–1045 (2005).
Zhang, A., Jia, Z., Guo, X. & Yang, T. Aldosterone induces epithelial-mesenchymal transition via ROS of mitochondrial origin. Am. J. Physiol. Renal Physiol. 293, F723–F731 (2007).
Lai, L., Chen, J., Hao, C. M., Lin, S. & Gu, Y. Aldosterone promotes fibronectin production through a Smad2-dependent TGF-beta1 pathway in mesangial cells. Biochem. Biophys. Res. Commun. 348, 70–75 (2006).
Chun, T. Y., Chander, P. N., Kim, J. W., Pratt, J. H. & Stier, C. T. Jr. Aldosterone, but not angiotensin II, increases profibrotic factors in kidney of adrenalectomized stroke-prone spontaneously hypertensive rats. Am. J. Physiol. Endocrinol. Metab. 295, E305–E312 (2008).
Juknevicius, I., Segal, Y., Kren, S., Lee, R. & Hostetter, T. H. Effect of aldosterone on renal transforming growth factor-beta. Am. J. Physiol. Renal Physiol. 286, F1059–F1062 (2004).
Sun, Y., Zhang, J., Zhang, J. Q. & Ramires, F. J. Local angiotensin II and transforming growth factor-beta1 in renal fibrosis of rats. Hypertension 35, 1078–1084 (2000).
Han, K. H. et al. Spironolactone ameliorates renal injury and connective tissue growth factor expression in type II diabetic rats. Kidney Int. 70, 111–120 (2006).
Yuan, J., Jia, R. & Bao, Y. Aldosterone up-regulates production of plasminogen activator inhibitor-1 by renal mesangial cells. J. Biochem. Mol. Biol. 40, 180–188 (2007).
Brown, N. J. et al. Aldosterone modulates plasminogen activator inhibitor-1 and glomerulosclerosis in vivo. Kidney Int. 58, 1219–1227 (2000).
Huang, W. et al. Aldosterone and TGF-beta1 synergistically increase PAI-1 and decrease matrix degradation in rat renal mesangial and fibroblast cells. Am. J. Physiol. Renal Physiol. 294, F1287–F1295 (2008).
Shimokawa, H. & Rashid, M. Development of Rho-kinase inhibitors for cardiovascular medicine. Trends Pharmacol. Sci. 28, 296–302 (2007).
Sun, G. P. et al. Involvements of Rho-kinase and TGF-beta pathways in aldosterone-induced renal injury. J. Am. Soc. Nephrol. 17, 2193–2201 (2006).
Kretzler, M., Koeppen-Hagemann, I. & Kriz, W. Podocyte damage is a critical step in the development of glomerulosclerosis in the uninephrectomised-desoxycorticosterone hypertensive rat. Virchows Arch. 425, 181–193 (1994).
Nagase, M. et al. Podocyte injury underlies the glomerulopathy of Dahl salt-hypertensive rats and is reversed by aldosterone blocker. Hypertension 47, 1084–1093 (2006).
Whaley-Connell, A. et al. Mineralocorticoid receptor antagonism attenuates glomerular filtration barrier remodeling in the transgenic Ren2 rat. Am. J. Physiol. Renal Physiol. 296, F1013–F1022 (2009).
Shibata, S., Nagase, M., Yoshida, S., Kawachi, H. & Fujita, T. Podocyte as the target for aldosterone: roles of oxidative stress and Sgk1. Hypertension 49, 355–364 (2007).
Nagase, M., Matsui, H., Shibata, S., Gotoda, T. & Fujita, T. Salt-induced nephropathy in obese spontaneously hypertensive rats via paradoxical activation of the mineralocorticoid receptor: role of oxidative stress. Hypertension 50, 877–883 (2007).
Lee, S. H. et al. Activation of local aldosterone system within podocytes is involved in apoptosis under diabetic conditions. Am. J. Physiol. Renal Physiol. 297, F1381–F1390 (2009).
Shibata, S. et al. Modification of mineralocorticoid receptor function by Rac1 GTPase: implication in proteinuric kidney disease. Nat. Med. 14, 1370–1376 (2008).
Otani, H. et al. Antagonistic effects of bone morphogenetic protein-4 and -7 on renal mesangial cell proliferation induced by aldosterone through MAPK activation. Am. J. Physiol. Renal Physiol. 292, F1513–F1525 (2007).
Terada, Y. et al. Aldosterone stimulates proliferation of mesangial cells by activating mitogen-activated protein kinase 1/2, cyclin D1, and cyclin A. J. Am. Soc. Nephrol. 16, 2296–2305 (2005).
Jackson, C. E. et al. Albuminuria in chronic heart failure: prevalence and prognostic importance. Lancet 374, 543–550 (2009).
Mercado, N. et al. Usefulness of proteinuria as a prognostic marker of mortality and cardiovascular events among patients undergoing percutaneous coronary intervention (data from the Evaluation of Oral Xemilofiban in Controlling Thrombotic Events [EXCITE] trial). Am. J. Cardiol. 102, 1151–1155 (2008).
Lambers Heerspink, H. J. et al. Albuminuria assessed from first-morning-void urine samples versus 24-hour urine collections as a predictor of cardiovascular morbidity and mortality. Am. J. Epidemiol. 168, 897–905 (2008).
Brantsma, A. H., Bakker, S. J., de Zeeuw, D., de Jong, P. E. & Gansevoort, R. T. Extended prognostic value of urinary albumin excretion for cardiovascular events. J. Am. Soc. Nephrol. 19, 1785–1791 (2008).
Eijkelkamp, W. B. et al. Albuminuria is a target for renoprotective therapy independent from blood pressure in patients with type 2 diabetic nephropathy: post hoc analysis from the Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL) trial. J. Am. Soc. Nephrol. 18, 1540–1546 (2007).
Wühl, E. et al. Strict blood-pressure control and progression of renal failure in children. N. Engl. J. Med. 361, 1639–1650 (2009).
Ruggenenti, P., Perna, A., Benini, R. & Remuzzi, G. Effects of dihydropyridine calcium channel blockers, angiotensin-converting enzyme inhibition, and blood pressure control on chronic, nondiabetic nephropathies. Gruppo Italiano di Studi Epidemiologici in Nefrologia (GISEN). J. Am. Soc. Nephrol. 9, 2096–2101 (1998).
Lewis, E. J., Hunsicker, L. G., Bain, R. P. & Rohde, R. D. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N. Engl. J. Med. 329, 1456–1462 (1993).
Maschio, G. et al. Effect of the angiotensin-converting-enzyme inhibitor benazepril on the progression of chronic renal insufficiency. The Angiotensin-Converting-Enzyme Inhibition in Progressive Renal Insufficiency Study Group. N. Engl. J. Med. 334, 939–945 (1996).
Ruggenenti, P. et al. Renal function and requirement for dialysis in chronic nephropathy patients on long-term ramipril: REIN follow-up trial. Gruppo Italiano di Studi Epidemiologici in Nefrologia (GISEN). Ramipril Efficacy in Nephropathy. Lancet 352, 1252–1256 (1998).
Brenner, B. M. et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 345, 861–869 (2001).
Bianchi, S., Bigazzi, R. & Campese, V. M. Long-term effects of spironolactone on proteinuria and kidney function in patients with chronic kidney disease. Kidney Int. 70, 2116–2123 (2006).
van den Meiracker, A. H. et al. Spironolactone in type 2 diabetic nephropathy: effects on proteinuria, blood pressure and renal function. J. Hypertens. 24, 2285–2292 (2006).
Schjoedt, K. J. et al. Beneficial impact of spironolactone in diabetic nephropathy. Kidney Int. 68, 2829–2836 (2005).
Rossing, K., Schjoedt, K. J., Smidt, U. M., Boomsma, F. & Parving, H. H. Beneficial effects of adding spironolactone to recommended antihypertensive treatment in diabetic nephropathy: a randomized, double-masked, cross-over study. Diabetes Care 28, 2106–2112 (2005).
Chrysostomou, A., Pedagogos, E., MacGregor, L. & Becker, G. J. Double-blind, placebo-controlled study on the effect of the aldosterone receptor antagonist spironolactone in patients who have persistent proteinuria and are on long-term angiotensin-converting enzyme inhibitor therapy, with or without an angiotensin II receptor blocker. Clin. J. Am. Soc. Nephrol. 1, 256–262 (2006).
Schjoedt, K. J. et al. Beneficial impact of spironolactone on nephrotic range albuminuria in diabetic nephropathy. Kidney Int. 70, 536–542 (2006).
Epstein, M. et al. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin. J. Am. Soc. Nephrol. 1, 940–951 (2006).
Furumatsu, Y. et al. Effect of renin-angiotensin-aldosterone system triple blockade on non-diabetic renal disease: addition of an aldosterone blocker, spironolactone, to combination treatment with an angiotensin-converting enzyme inhibitor and angiotensin II receptor blocker. Hypertens. Res. 31, 59–67 (2008).
Tylicki, L. et al. Triple pharmacological blockade of the renin-angiotensin-aldosterone system in nondiabetic CKD: an open-label crossover randomized controlled trial. Am. J. Kidney Dis. 52, 486–493 (2008).
Levey, A. S. et al. Proteinuria as a surrogate outcome in CKD: report of a scientific workshop sponsored by the National Kidney Foundation and the US Food and Drug Administration. Am. J. Kidney Dis. 54, 205–226 (2009).
de Zeeuw, D. Targeting proteinuria as a valid surrogate for individualized kidney protective therapy. Am. J. Kidney Dis. 51, 713–716 (2008).
Bomback, A. S., Kshirsagar, A. V. & Klemmer, P. J. Renal aspirin: will all patients with chronic kidney disease one day take spironolactone? Nat. Clin. Pract. Nephrol. 5, 74–75 (2009).
Brilla, C. G. & Weber, K. T. Mineralocorticoid excess, dietary sodium, and myocardial fibrosis. J. Lab. Clin. Med. 120, 893–901 (1992).
Lacolley, P. et al. Prevention of aortic and cardiac fibrosis by spironolactone in old normotensive rats. J. Am. Coll. Cardiol. 37, 662–667 (2001).
Benetos, A., Lacolley, P. & Safar, M. E. Prevention of aortic fibrosis by spironolactone in spontaneously hypertensive rats. Arterioscler. Thromb. Vasc. Biol. 17, 1152–1156 (1997).
Rizzoni, D. et al. Vascular hypertrophy and remodeling in secondary hypertension. Hypertension 28, 785–790 (1996).
Callera, G. E. et al. Aldosterone activates vascular p38MAP kinase and NADPH oxidase via c-Src. Hypertension 45, 773–779 (2005).
Iwashima, F. et al. Aldosterone induces superoxide generation via Rac1 activation in endothelial cells. Endocrinology 149, 1009–1014 (2008).
Sun, Y. et al. Aldosterone-induced inflammation in the rat heart: role of oxidative stress. Am. J. Pathol. 161, 1773–1781 (2002).
Hayashi, H. et al. Aldosterone nongenomically produces NADPH oxidase-dependent reactive oxygen species and induces myocyte apoptosis. Hypertens. Res. 31, 363–375 (2008).
Petry, A. et al. NOX2 and NOX4 mediate proliferative response in endothelial cells. Antioxid. Redox Signal. 8, 1473–1484 (2006).
Stas, S. et al. Mineralocorticoid receptor blockade attenuates chronic overexpression of the renin-angiotensin-aldosterone system stimulation of reduced nicotinamide adenine dinucleotide phosphate oxidase and cardiac remodeling. Endocrinology 148, 3773–3780 (2007).
Nakano, S., Kobayashi, N., Yoshida, K., Ohno, T. & Matsuoka, H. Cardioprotective mechanisms of spironolactone associated with the angiotensin-converting enzyme/epidermal growth factor receptor/extracellular signal-regulated kinases, NAD(P)H oxidase/lectin-like oxidized low-density lipoprotein receptor-1, and Rho-kinase pathways in aldosterone/salt-induced hypertensive rats. Hypertens. Res. 28, 925–936 (2005).
Nakamura, T. et al. Critical role of apoptosis signal-regulating kinase 1 in aldosterone/salt-induced cardiac inflammation and fibrosis. Hypertension 54, 544–551 (2009).
Leopold, J. A. et al. Aldosterone impairs vascular reactivity by decreasing glucose-6-phosphate dehydrogenase activity. Nat. Med. 13, 189–197 (2007).
Iglarz, M., Touyz, R. M., Viel, E. C., Amiri, F. & Schiffrin, E. L. Involvement of oxidative stress in the profibrotic action of aldosterone. Interaction wtih the renin-angiotension system. Am. J. Hypertens. 17, 597–603 (2004).
Park, Y. M., Park, M. Y., Suh, Y. L. & Park, J. B. NAD(P)H oxidase inhibitor prevents blood pressure elevation and cardiovascular hypertrophy in aldosterone-infused rats. Biochem. Biophys. Res. Commun. 313, 812–817 (2004).
Michea, L. et al. Mineralocorticoid receptor antagonism attenuates cardiac hypertrophy and prevents oxidative stress in uremic rats. Hypertension 52, 295–300 (2008).
Rocha, R. et al. Aldosterone induces a vascular inflammatory phenotype in the rat heart. Am. J. Physiol. Heart Circ. Physiol 283, H1802–H1810 (2002).
Rocha, R. et al. Selective aldosterone blockade prevents angiotensin II/salt-induced vascular inflammation in the rat heart. Endocrinology 143, 4828–4836 (2002).
Rickard, A. J. et al. Deletion of mineralocorticoid receptors from macrophages protects against deoxycorticosterone/salt-induced cardiac fibrosis and increased blood pressure. Hypertension 54, 537–543 (2009).
Min, L. J. et al. Aldosterone and angiotensin II synergistically induce mitogenic response in vascular smooth muscle cells. Circ. Res. 97, 434–442 (2005).
Nakamura, Y. et al. MDM2: a novel mineralocorticoid-responsive gene involved in aldosterone-induced human vascular structural remodeling. Am. J. Pathol. 169, 362–371 (2006).
Jeong, Y. et al. Aldosterone activates endothelial exocytosis. Proc. Natl Acad. Sci. USA 106, 3782–3787 (2009).
Irita, J. et al. Plasma osteopontin levels are higher in patients with primary aldosteronism than in patients with essential hypertension. Am. J. Hypertens. 19, 293–297 (2006).
Luther, J. M. et al. Angiotensin II induces interleukin-6 in humans through a mineralocorticoid receptor-dependent mechanism. Hypertension 48, 1050–1057 (2006).
Savoia, C., Touyz, R. M., Amiri, F. & Schiffrin, E. L. Selective mineralocorticoid receptor blocker eplerenone reduces resistance artery stiffness in hypertensive patients. Hypertension 51, 432–439 (2008).
Robert, V. et al. Angiotensin AT1 receptor subtype as a cardiac target of aldosterone: role in aldosterone-salt-induced fibrosis. Hypertension 33, 981–986 (1999).
Schiffrin, E. L., Thomé, F. S. & Genest, J. Vascular angiotensin II receptors in renal and DOCA-salt hypertensive rats. Hypertension 5, V16–V21 (1983).
Schiffrin, E. L., Gutkowska, J. & Genest, J. Effect of angiotensin II and deoxycorticosterone infusion on vascular angiotensin II receptors in rats. Am. J. Physiol. 246, H608–H614 (1984).
Schiffrin, E. L., Franks, D. J. & Gutkowska, J. Effect of aldosterone on vascular angiotensin II receptors in the rat. Can. J. Physiol. Pharmacol. 63, 1522–1527 (1985).
Lemarié, C. A. et al. Aldosterone-induced activation of signaling pathways requires activity of angiotensin type 1a receptors. Circ. Res. 105, 852–859 (2009).
Min, L. J. et al. Cross-talk between aldosterone and angiotensin II in vascular smooth muscle cell senescence. Cardiovasc. Res. 76, 506–516 (2007).
Montezano, A. C. et al. Aldosterone and angiotensin II synergistically stimulate migration in vascular smooth muscle cells through c-Src-regulated redox-sensitive RhoA pathways. Arterioscler. Thromb. Vasc. Biol. 28, 1511–1518 (2008).
Blanco-Rivero, J. et al. Participation of prostacyclin in endothelial dysfunction induced by aldosterone in normotensive and hypertensive rats. Hypertension 46, 107–112 (2005).
Imanishi, T. et al. Addition of eplerenone to an angiotensin-converting enzyme inhibitor effectively improves nitric oxide bioavailability. Hypertension 51, 734–741 (2008).
Rajagopalan, S., Duquaine, D., King, S., Pitt, B. & Patel, P. Mineralocorticoid receptor antagonism in experimental atherosclerosis. Circulation 105, 2212–2216 (2002).
Garnier, A. et al. Cardiac specific increase in aldosterone production induces coronary dysfunction in aldosterone synthase-transgenic mice. Circulation 110, 1819–1825 (2004).
Xavier, F. E. et al. Aldosterone induces endothelial dysfunction in resistance arteries from normotensive and hypertensive rats by increasing thromboxane A2 and prostacyclin. Br. J. Pharmacol. 154, 1225–1235 (2008).
Farquharson, C. A. & Struthers, A. D. Spironolactone increases nitric oxide bioactivity, improves endothelial vasodilator dysfunction, and suppresses vascular angiotensin I/angiotensin II conversion in patients with chronic heart failure. Circulation 101, 594–597 (2000).
Peng, X., Haldar, S., Deshpande, S., Irani, K. & Kass, D. A. Wall stiffness suppresses Akt/eNOS and cytoprotection in pulse-perfused endothelium. Hypertension 41, 378–381 (2003).
Kidoaki, S. & Matsuda, T. Shape-engineered vascular endothelial cells: nitric oxide production, cell elasticity, and actin cytoskeletal features. J. Biomed. Mater. Res. A 81, 728–735 (2007).
Oberleithner, H. et al. Human endothelium: target for aldosterone. Hypertension 43, 952–956 (2004).
Hillebrand, U. et al. Dose-dependent endothelial cell growth and stiffening by aldosterone: endothelial protection by eplerenone. J. Hypertens. 25, 639–647 (2007).
Oberleithner, H. et al. Plasma sodium stiffens vascular endothelium and reduces nitric oxide release. Proc. Natl Acad. Sci. USA 104, 16281–16286 (2007).
Mutoh, A., Isshiki, M. & Fujita, T. Aldosterone enhances ligand-stimulated nitric oxide production in endothelial cells. Hypertens. Res. 31, 1811–1820 (2008).
Liu, S. L., Schmuck, S., Chorazcyzewski, J. Z., Gros, R. & Feldman, R. D. Aldosterone regulates vascular reactivity: short-term effects mediated by phosphatidylinositol 3-kinase-dependent nitric oxide synthase activation. Circulation 108, 2400–2406 (2003).
Nietlispach, F. et al. Influence of acute and chronic mineralocorticoid excess on endothelial function in healthy men. Hypertension 50, 82–88 (2007).
Farquharson, C. A. & Struthers, A. D. Aldosterone induces acute endothelial dysfunction in vivo in humans: evidence for an aldosterone-induced vasculopathy. Clin. Sci. (Lond.) 103, 425–431 (2002).
Romagni, P., Rossi, F., Guerrini, L., Quirini, C. & Santiemma, V. Aldosterone induces contraction of the resistance arteries in man. Atherosclerosis 166, 345–349 (2003).
Schmidt, B. M. et al. Short term cardiovascular effects of aldosterone in healthy male volunteers. J. Clin. Endocrinol. Metab. 84, 3528–3533 (1999).
Schmidt, B. M. et al. Rapid nongenomic effects of aldosterone on human forearm vasculature. Hypertension 42, 156–160 (2003).
Arima, S. et al. Nongenomic vascular action of aldosterone in the glomerular microcirculation. J. Am. Soc. Nephrol. 14, 2255–2263 (2003).
Wehling, M., Neylon, C. B., Fullerton, M., Bobik, A. & Funder, J. W. Nongenomic effects of aldosterone on intracellular Ca2+ in vascular smooth muscle cells. Circ. Res. 76, 973–979 (1995).
Calò, L. A. et al. Absence of vascular remodelling in a high angiotensin-II state (Bartter's and Gitelman's syndromes): implications for angiotensin II signalling pathways. Nephrol. Dial. Transplant. 23, 2804–2809 (2008).
Endemann, D. H., Touyz, R. M., Iglarz, M., Savoia, C. & Schiffrin, E. L. Eplerenone prevents salt-induced vascular remodeling and cardiac fibrosis in stroke-prone spontaneously hypertensive rats. Hypertension 43, 1252–1257 (2004).
Nagata, K. et al. Mineralocorticoid receptor antagonism attenuates cardiac hypertrophy and failure in low-aldosterone hypertensive rats. Hypertension 47, 656–664 (2006).
Funder, J. W. Reconsidering the roles of the mineralocorticoid receptor. Hypertension 53, 286–290 (2009).
Young, M. & Funder, J. W. Eplerenone, but not steroid withdrawal, reverses cardiac fibrosis in deoxycorticosterone/salt-treated rats. Endocrinology 145, 3153–3157 (2004).
Young, M. J., Moussa, L., Dilley, R. & Funder, J. W. Early inflammatory responses in experimental cardiac hypertrophy and fibrosis: effects of 11 beta-hydroxysteroid dehydrogenase inactivation. Endocrinology 144, 1121–1125 (2003).
Jorde, U. P. et al. Elevated plasma aldosterone levels despite complete inhibition of the vascular angiotensin-converting enzyme in chronic heart failure. Circulation 106, 1055–1057 (2002).
Iraqi, W. et al. Extracellular cardiac matrix biomarkers in patients with acute myocardial infarction complicated by left ventricular dysfunction and heart failure: insights from the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS) study. Circulation 119, 2471–2479 (2009).
Edwards, N. C., Steeds, R. P., Stewart, P. M., Ferro, C. J. & Townend, J. N. Effect of spironolactone on left ventricular mass and aortic stiffness in early-stage chronic kidney disease: a randomized controlled trial. J. Am. Coll. Cardiol. 54, 505–512 (2009).
Pitt, B., Bakris, G., Ruilope, L. M., DiCarlo, L. & Mukherjee, R. Serum potassium and clinical outcomes in the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS). Circulation 118, 1643–1650 (2008).
Juurlink, D. N. et al. Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study. N. Engl. J. Med. 351, 543–551 (2004).
Masoudi, F. A. et al. Adoption of spironolactone therapy for older patients with heart failure and left ventricular systolic dysfunction in the United States, 1998–2001. Circulation 112, 39–47 (2005).
Bozkurt, B., Agoston, I. & Knowlton, A. A. Complications of inappropriate use of spironolactone in heart failure: when an old medicine spirals out of new guidelines. J. Am. Coll. Cardiol. 41, 211–214 (2003).
Lea, W. B. et al. Aldosterone antagonism or synthase inhibition reduces end-organ damage induced by treatment with angiotensin and high salt. Kidney Int. 75, 936–944 (2009).
Acknowledgements
The authors' work described in this Review was supported by the Canadian Institutes of Health Research (CIHR) grants 37917 and 82790, a Canada Research Chair (CRC) on Hypertension and Vascular Research from the CIHR/CRC Program of the Government of Canada, and the Canada Fund for Innovation (all to E. L. S.). M. B. was supported by the Heart and Stroke Foundation of Canada.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
E. L. Schiffrin receives grant/research support from the Canadian Institutes of Health Research and receives grant/research support from and is a consultant for Pfizer Canada. M. Briet declares no competing interests.
Rights and permissions
About this article
Cite this article
Briet, M., Schiffrin, E. Aldosterone: effects on the kidney and cardiovascular system. Nat Rev Nephrol 6, 261–273 (2010). https://doi.org/10.1038/nrneph.2010.30
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2010.30
- Springer Nature Limited
This article is cited by
-
Mineralocorticoid receptor overactivation: targeting systemic impact with non-steroidal mineralocorticoid receptor antagonists
Diabetologia (2024)
-
Advancing Guideline-Directed Medical Therapy in Heart Failure: Overcoming Challenges and Maximizing Benefits
American Journal of Cardiovascular Drugs (2024)
-
The renin angiotensin aldosterone system
Pflügers Archiv - European Journal of Physiology (2024)
-
Higher plasma aldosterone concentrations in patients with aortic diseases and hypertension: a retrospective observational study
European Journal of Medical Research (2023)
-
Diagnostic accuracy of using multiple cytokines to predict aldosterone-producing adenoma
Scientific Reports (2023)