Key Points
-
Intraepithelial lymphocytes (IELs) that reside within the epithelium of the intestine form one of the main branches of the immune system.
-
IELs are almost exclusively antigen-experienced T cells that are heterogeneous in phenotype, ontogeny, antigen specificity and function.
-
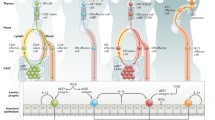
IELs consist of two main subtypes. The natural IELs consist of T cell receptor-αβ (TCRαβ)+ T cells and TCRγδ+ T cells that express CD8αα or are negative for both CD4 and CD8. These IELs acquire their activated and functional phenotype, in part, during self-agonist antigen-based selection in the thymus. Induced IELs consist of CD4+ and CD8αβ+ TCRαβ+ T cells that often co-express CD8αα. They are the progeny of naive T cells that are conventionally selected in the thymus and acquire their activated phenotype and functional differentiation post-thymically in response to cognate antigens encountered in the periphery.
-
IELs serve unique and dual functions. Their 'light side' is their ability to preserve the integrity of the epithelium and prevent damage induced by invading pathogens (protective immunity) or induced by excessive or aberrant inflammatory immune responses.
-
IELs also have a 'dark side': as they are located within the fragile, single cell layer of the epithelium and possess potent cytotoxic effector machinery, they can target the epithelium in a destructive way. Consequently, IELs may drive immunopathological responses in chronic inflammatory diseases, such as inflammatory bowel disease and coeliac disease.
Abstract
The intraepithelial lymphocytes (IELs) that reside within the epithelium of the intestine form one of the main branches of the immune system. As IELs are located at this critical interface between the core of the body and the outside environment, they must balance protective immunity with an ability to safeguard the integrity of the epithelial barrier: failure to do so would compromise homeostasis of the organism. In this Review, we address how the unique development and functions of intestinal IELs allow them to achieve this balance.



Similar content being viewed by others
References
Hooper, L. V. & Macpherson, A. J. Immune adaptations that maintain homeostasis with the intestinal microbiota. Nature Rev. Immunol. 10, 159–169 (2010).
Darlington, D. & Rogers, A. W. Epithelial lymphocytes in the small intestine of the mouse. J. Anat. 100, 813–830 (1966).
Bonneville, M. et al. Intestinal intraepithelial lymphocytes are a distinct set of γδ T cells. Nature 336, 479–481 (1988).
Goodman, T. & Lefrancois, L. Expression of the γ-δ T-cell receptor on intestinal CD8+ intraepithelial lymphocytes. Nature 333, 855–858 (1988). References 3 and 4 describe the high frequency of TCRγδ-expressing T cells among IELs.
Guy-Grand, D. et al. Two gut intraepithelial CD8+ lymphocyte populations with different T cell receptors: a role for the gut epithelium in T cell differentiation. J. Exp. Med. 173, 471–481 (1991).
Cheroutre, H. Starting at the beginning: new perspectives on the biology of mucosal T cells. Annu. Rev. Immunol. 22, 217–246 (2004).
Sugahara, S. et al. Extrathymic derivation of gut lymphocytes in parabiotic mice. Immunology 96, 57–65 (1999).
Suzuki, S. et al. Low level of mixing of partner cells seen in extrathymic T cells in the liver and intestine of parabiotic mice: its biological implication. Eur. J. Immunol. 28, 3719–3729 (1998).
Shires, J., Theodoridis, E. & Hayday, A. C. Biological insights into TCRγδ+ and TCRαβ+ intraepithelial lymphocytes provided by serial analysis of gene expression (SAGE). Immunity 15, 419–434 (2001). One of the first studies to describe the cytotoxic function of IELs against virus-infected intestinal epithelial cells.
Offit, P. A. & Dudzik, K. I. Rotavirus-specific cytotoxic T lymphocytes appear at the intestinal mucosal surface after rotavirus infection. J. Virol. 63, 3507–3512 (1989).
Tang, F. et al. Cytosolic PLA2 is required for CTL-mediated immunopathology of celiac disease via NKG2D and IL-15. J. Exp. Med. 206, 707–719 (2009).
Chardes, T., Buzoni-Gatel, D., Lepage, A., Bernard, F. & Bout, D. Toxoplasma gondii oral infection induces specific cytotoxic CD8α/β+ Thy-1+ gut intraepithelial lymphocytes, lytic for parasite-infected enterocytes. J. Immunol. 153, 4596–4603 (1994).
Muller, S., Buhler-Jungo, M. & Mueller, C. Intestinal intraepithelial lymphocytes exert potent protective cytotoxic activity during an acute virus infection. J. Immunol. 164, 1986–1994 (2000).
Roberts, A. I., O'Connell, S. M., Biancone, L., Brolin, R. E. & Ebert, E. C. Spontaneous cytotoxicity of intestinal intraepithelial lymphocytes: clues to the mechanism. Clin. Exp. Immunol. 94, 527–532 (1993).
Ebert, E. C. & Roberts, A. I. Lymphokine-activated killing by human intestinal lymphocytes. Cell. Immunol. 146, 107–116 (1993).
Guy-Grand, D., Malassis-Seris, M., Briottet, C. & Vassalli, P. Cytotoxic differentiation of mouse gut thymodependent and independent intraepithelial T lymphocytes is induced locally. Correlation between functional assays, presence of perforin and granzyme transcripts, and cytoplasmic granules. J. Exp. Med. 173, 1549–1552 (1991).
Yamagata, T., Mathis, D. & Benoist, C. Self-reactivity in thymic double-positive cells commits cells to a CD8αα lineage with characteristics of innate immune cells. Nature Immunol. 5, 597–605 (2004).
Denning, T. L. et al. Mouse TCRαβ+CD8αα intraepithelial lymphocytes express genes that down-regulate their antigen reactivity and suppress immune responses. J. Immunol. 178, 4230–4239 (2007). References 17 and 18 suggest that CD8αα+TCRαβ+ IEL precursor cells acquire functional specialization during their differentiation in the thymus. Their unique functional and phenotypic differentiation is reflected in the gene signature that they display at the mature stage as CD8αα+TCRαβ+ IELs.
Bhagat, G. et al. Small intestinal CD8+TCRγδ+NKG2A+ intraepithelial lymphocytes have attributes of regulatory cells in patients with celiac disease. J. Clin. Invest. 118, 281–293 (2008).
Zhou, R., Wei, H., Sun, R., Zhang, J. & Tian, Z. NKG2D recognition mediates Toll-like receptor 3 signaling-induced breakdown of epithelial homeostasis in the small intestines of mice. Proc. Natl Acad. Sci. USA 104, 7512–7515 (2007).
Cepek, K. L. et al. Adhesion between epithelial cells and T lymphocytes mediated by E-cadherin and the αEβ7 integrin. Nature 372, 190–193 (1994).
Kilshaw, P. J. & Murant, S. J. A new surface antigen on intraepithelial lymphocytes in the intestine. Eur. J. Immunol. 20, 2201–2207 (1990).
Leishman, A. J. et al. Precursors of functional MHC class I- or class II-restricted CD8αα+ T cells are positively selected in the thymus by agonist self-peptides. Immunity 16, 355–364 (2002). The first report to describe the thymic agonist selection pathway for MHC class-I- and MHC class-II-restricted IELs using TCR transgenic models.
Gangadharan, D. et al. Identification of pre- and postselection TCRαβ+ intraepithelial lymphocyte precursors in the thymus. Immunity 25, 631–641 (2006).
Madakamutil, L. T. et al. CD8αα-mediated survival and differentiation of CD8 memory T cell precursors. Science 304, 590–593 (2004).
Hershberg, R. et al. Expression of the thymus leukemia antigen in mouse intestinal epithelium. Proc. Natl Acad. Sci. USA 87, 9727–9731 (1990).
Leishman, A. J. et al. T cell responses modulated through interaction between CD8αα and the nonclassical MHC class I molecule, TL. Science 294, 1936–1939 (2001). Identified TLA as a high-affinity ligand for CD8αα and showed functional effects of the interaction between TLA and CD8αα homodimers.
Lefrancois, L. Phenotypic complexity of intraepithelial lymphocytes of the small intestine. J. Immunol. 147, 1746–1751 (1991).
Mosley, R. L., Styre, D. & Klein, J. R. CD4+CD8+ murine intestinal intraepithelial lymphocytes. Int. Immunol. 2, 361–365 (1990).
Ohteki, T. & MacDonald, H. R. Expression of the CD28 costimulatory molecule on subsets of murine intestinal intraepithelial lymphocytes correlates with lineage and responsiveness. Eur. J. Immunol. 23, 1251–1255 (1993).
Van Houten, N., Mixter, P. F., Wolfe, J. & Budd, R. C. CD2 expression on murine intestinal intraepithelial lymphocytes is bimodal and defines proliferative capacity. Int. Immunol. 5, 665–672 (1993).
Lin, T. et al. CD3-CD8+ intestinal intraepithelial lymphocytes (IEL) and the extrathymic development of IEL. Eur. J. Immunol. 24, 1080–1087 (1994).
Wang, R., Wang-Zhu, Y. & Grey, H. Interactions between double positive thymocytes and high affinity ligands presented by cortical epithelial cells generate double negative thymocytes with T cell regulatory activity. Proc. Natl Acad. Sci. USA 99, 2181–2186 (2002).
Guy-Grand, D., Cuenod-Jabri, B., Malassis-Seris, M., Selz, F. & Vassalli, P. Complexity of the mouse gut T cell immune system: identification of two distinct natural killer T cell intraepithelial lineages. Eur. J. Immunol. 26, 2248–2256 (1996).
Huleatt, J. W. & Lefrancois, L. Antigen-driven induction of CD11c on intestinal intraepithelial lymphocytes and CD8+ T cells in vivo. J. Immunol. 154, 5684–5693 (1995).
Guy-Grand, D. et al. Different use of T cell receptor transducing modules in two populations of gut intraepithelial lymphocytes are related to distinct pathways of T cell differentiation. J. Exp. Med. 180, 673–679 (1994).
Ohno, H., Ono, S., Hirayama, N., Shimada, S. & Saito, T. Preferential usage of the Fc receptor γ chain in the T cell antigen receptor complex by γ/δ T cells localized in epithelia. J. Exp. Med. 179, 365–369 (1994).
Park, S. Y. et al. Differential contribution of the FcR γ chain to the surface expression of the T cell receptor among T cells localized in epithelia: analysis of FcR γ-deficient mice. Eur. J. Immunol. 25, 2107–2110 (1995).
Arstila, T. et al. Identical T cell clones are located within the mouse gut epithelium and lamina propia and circulate in the thoracic duct lymph. J. Exp. Med. 191, 823–834 (2000).
Lefrancois, L. & Masopust, D. T cell immunity in lymphoid and non-lymphoid tissues. Curr. Opin. Immunol. 14, 503–508 (2002).
Mowat, A. M. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Rev. Immunol. 3, 331–341 (2003).
Neutra, M. R., Mantis, N. J. & Kraehenbuhl, J. P. Collaboration of epithelial cells with organized mucosal lymphoid tissues. Nature Immunol. 2, 1004–1009 (2001).
Cheroutre, H. & Lambolez, F. Doubting the TCR coreceptor function of CD8αα. Immunity 28, 149–159 (2008).
Regnault, A., Cumano, A., Vassalli, P., Guy-Grand, D. & Kourilsky, P. Oligoclonal repertoire of the CD8αα and the CD8αβ TCR-α/β murine intestinal intraepithelial T lymphocytes: evidence for the random emergence of T cells. J. Exp. Med. 180, 1345–1358 (1994). This study describes the oligoclonal repertoire of TCRαβ+ IELs in mice.
Cheroutre, H. & Lambolez, F. The thymus chapter in the life of gut-specific intra epithelial lymphocytes. Curr. Opin. Immunol. 20, 185–191 (2008).
Ishikawa, H. et al. Curriculum vitae of intestinal intraepithelial T cells: their developmental and behavioral characteristics. Immunol. Rev. 215, 154–165 (2007).
Lambolez, F., Kronenberg, M. & Cheroutre, H. Thymic differentiation of TCRαβ+ CD8αα+ IELs. Immunol. Rev. 215, 178–188 (2007).
Rocha, B. The extrathymic T-cell differentiation in the murine gut. Immunol. Rev. 215, 166–177 (2007).
Eberl, G. & Littman, D. R. Thymic origin of intestinal αβ T cells revealed by fate mapping of RORγt+ cells. Science 305, 248–251 (2004).
Cheroutre, H., Mucida, D. & Lambolez, F. The importance of being earnestly selfish. Nature Immunol. 10, 1047–1049 (2009).
Hogquist, K. A., Baldwin, T. A. & Jameson, S. C. Central tolerance: learning self-control in the thymus. Nature Rev. Immunol. 5, 772–782 (2005).
Jensen, K. D. et al. Thymic selection determines γδ T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon γ. Immunity 29, 90–100 (2008).
Carpenter, A. C. & Bosselut, R. Decision checkpoints in the thymus. Nature Immunol. 11, 666–673 (2010).
Collins, A., Littman, D. R. & Taniuchi, I. RUNX proteins in transcription factor networks that regulate T-cell lineage choice. Nature Rev. Immunol. 9, 106–115 (2009).
Singer, A., Adoro, S. & Park, J. H. Lineage fate and intense debate: myths, models and mechanisms of CD4- versus CD8-lineage choice. Nature Rev. Immunol. 8, 788–801 (2008).
Manzano, M., Abadia-Molina, A. C., Garcia-Olivares, E., Gil, A. & Rueda, R. Absolute counts and distribution of lymphocyte subsets in small intestine of BALB/c mice change during weaning. J. Nutr. 132, 2757–2762 (2002).
Helgeland, L., Brandtzaeg, P., Rolstad, B. & Vaage, J. T. Sequential development of intraepithelial γδ and αβ T lymphocytes expressing CD8αβ in neonatal rat intestine: requirement for the thymus. Immunology 92, 447–456 (1997).
Steege, J. C., Buurman, W. A. & Forget, P. P. The neonatal development of intraepithelial and lamina propria lymphocytes in the murine small intestine. Dev. Immunol. 5, 121–128 (1997).
Umesaki, Y., Setoyama, H., Matsumoto, S. & Okada, Y. Expansion of αβ T-cell receptor-bearing intestinal intraepithelial lymphocytes after microbial colonization in germ-free mice and its independence from thymus. Immunology 79, 32–37 (1993).
Latthe, M., Terry, L. & MacDonald, T. T. High frequency of CD8αα homodimer-bearing T cells in human fetal intestine. Eur. J. Immunol. 24, 1703–1705 (1994).
Vaz, N. M. & Faria, A. M. C. Guia Incompleto de Imunobiologia: Imunologia como se o Organismo Importasse. (COPMED, Belo Horizonte, 1993).
Mota-Santos, T. et al. Divergency in the specificity of the induction and maintenance of neonatal suppression. Eur. J. Immunol. 20, 1717–1721 (1990). References 59 and 62 describe the relevance of microbial colonization for the development of different IEL populations.
Pereira, P. et al. Autonomous activation of B and T cells in antigen-free mice. Eur. J. Immunol. 16, 685–688 (1986).
Hashimoto, K., Handa, H., Umehara, K. & Sasaki, S. Germfree mice reared on an “antigen-free” diet. Lab. Anim. Sci. 28, 38–45 (1978).
Menezes, J. S. et al. Stimulation by food proteins plays a critical role in the maturation of the immune system. Int. Immunol. 15, 447–455 (2003).
Staton, T. L. et al. CD8+ recent thymic emigrants home to and efficiently repopulate the small intestine epithelium. Nature Immunol. 7, 482–488 (2006).
Grueter, B. et al. Runx3 regulates integrin αE/CD103 and CD4 expression during development of CD4-/CD8+ T cells. J. Immunol. 175, 1694–1705 (2005).
Staton, T. L., Johnston, B., Butcher, E. C. & Campbell, D. J. Murine CD8+ recent thymic emigrants are αE integrin-positive and CC chemokine ligand 25 responsive. J. Immunol. 172, 7282–7288 (2004).
Kunisawa, J. et al. Sphingosine 1-phosphate dependence in the regulation of lymphocyte trafficking to the gut epithelium. J. Exp. Med. 204, 2335–2348 (2007).
Yu, S., Bruce, D., Froicu, M., Weaver, V. & Cantorna, M. T. Failure of T cell homing, reduced CD4/CD8αα intraepithelial lymphocytes, and inflammation in the gut of vitamin D receptor KO mice. Proc. Natl Acad. Sci. USA 105, 20834–20839 (2008).
Campbell, D. J. & Butcher, E. C. Rapid acquisition of tissue-specific homing phenotypes by CD4+ T cells activated in cutaneous or mucosal lymphoid tissues. J. Exp. Med. 195, 135–141 (2002).
Schulz, O. et al. Intestinal CD103+, but not CX3CR1+, antigen sampling cells migrate in lymph and serve classical dendritic cell functions. J. Exp. Med. 206, 3101–3114 (2009).
Bogunovic, M. et al. Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 (2009).
Ginhoux, F. et al. The origin and development of nonlymphoid tissue CD103+ DCs. J. Exp. Med. 206, 3115–3130 (2009).
Niess, J. H. et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 307, 254–258 (2005).
Agace, W. W. T-cell recruitment to the intestinal mucosa. Trends Immunol. 29, 514–522 (2008).
Iwata, M. et al. Retinoic acid imprints gut-homing specificity on T cells. Immunity 21, 527–538 (2004).
Mora, J. R. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature 424, 88–93 (2003). References 77 and 78 identified a role for retinoic acid-producing DCs in promoting T cell homing to the intestine.
McDermott, M. R. et al. Impaired intestinal localization of mesenteric lymphoblasts associated with vitamin A deficiency and protein-calorie malnutrition. Immunology 45, 1–5 (1982).
Hammerschmidt, S. I. et al. Stromal mesenteric lymph node cells are essential for the generation of gut-homing T cells in vivo. J. Exp. Med. 205, 2483–2490 (2008).
Edele, F. et al. Cutting edge: instructive role of peripheral tissue cells in the imprinting of T cell homing receptor patterns. J. Immunol. 181, 3745–3749 (2008).
Johansson-Lindbom, B. & Agace, W. W. Generation of gut-homing T cells and their localization to the small intestinal mucosa. Immunol. Rev. 215, 226–242 (2007).
El-Asady, R. et al. TGF-β-dependent CD103 expression by CD8+ T cells promotes selective destruction of the host intestinal epithelium during graft-versus-host disease. J. Exp. Med. 201, 1647–1657 (2005).
Ericsson, A., Svensson, M., Arya, A. & Agace, W. W. CCL25/CCR9 promotes the induction and function of CD103 on intestinal intraepithelial lymphocytes. Eur. J. Immunol. 34, 2720–2729 (2004).
Andrew, D. P., Rott, L. S., Kilshaw, P. J. & Butcher, E. C. Distribution of α4β7 and αEβ7 integrins on thymocytes, intestinal epithelial lymphocytes and peripheral lymphocytes. Eur. J. Immunol. 26, 897–905 (1996).
Poussier, P., Ning, T., Banerjee, D. & Julius, M. A unique subset of self-specific intraintestinal T cells maintains gut integrity. J. Exp. Med. 195, 1491–1497 (2002). Details a protective role for CD8αα+TCRαβ+ IELs in preventing inflammation induced by conventional CD4+ T cells in a model of induced colitis.
Kuhl, A. A. et al. Aggravation of intestinal inflammation by depletion/deficiency of γδ T cells in different types of IBD animal models. J. Leukoc. Biol. 81, 168–175 (2007).
Mucida, D. & Cheroutre, H. The many face-lifts of CD4 T helper cells. Adv. Immunol. 107, 139–152 (2010).
Roberts, S. J. et al. T-cell αβ+ and γδ+ deficient mice display abnormal but distinct phenotypes toward a natural, widespread infection of the intestinal epithelium. Proc. Natl Acad. Sci. USA 93, 11774–11779 (1996).
Komano, H. et al. Homeostatic regulation of intestinal epithelia by intraepithelial γδ T cells. Proc. Natl Acad. Sci. USA 92, 6147–6151 (1995).
Guy-Grand, D., DiSanto, J. P., Henchoz, P., Malassis-Seris, M. & Vassalli, P. Small bowel enteropathy: role of intraepithelial lymphocytes and of cytokines (IL-12, IFN-γ, TNF) in the induction of epithelial cell death and renewal. Eur. J. Immunol. 28, 730–744 (1998).
Boismenu, R. & Havran, W. L. Modulation of epithelial cell growth by intraepithelial γδ T cells. Science 266, 1253–1255 (1994). References 90–92 describe protective roles for TCRγδ+ IELs in intestinal epithelial cell growth and turnover and epithelium homeostasis.
Mengel, J. et al. Anti-γδ T cell antibody blocks the induction and maintenance of oral tolerance to ovalbumin in mice. Immunol. Lett. 48, 97–102 (1995).
Fujihashi, K. et al. γδ T cells regulate mucosally induced tolerance in a dose-dependent fashion. Int. Immunol. 11, 1907–1916 (1999).
Chen, Y., Chou, K., Fuchs, E., Havran, W. L. & Boismenu, R. Protection of the intestinal mucosa by intraepithelial γδ T cells. Proc. Natl Acad. Sci. USA 99, 14338–14343 (2002).
Inagaki-Ohara, K. et al. Intestinal intraepithelial lymphocytes sustain the epithelial barrier function against Eimeria vermiformis infection. Infect. Immun. 74, 5292–5301 (2006).
Ismail, A. S., Behrendt, C. L. & Hooper, L. V. Reciprocal interactions between commensal bacteria and γδ intraepithelial lymphocytes during mucosal injury. J. Immunol. 182, 3047–3054 (2009).
Mombaerts, P., Arnoldi, J., Russ, F., Tonegawa, S. & Kaufmann, S. H. Different roles of αβ and γδ T cells in immunity against an intracellular bacterial pathogen. Nature 365, 53–56 (1993). Shows a protective role for TCRαβ+ and TCRγδ+ IELs against intracellular bacterial infections in the intestine.
Hamada, S. et al. Importance of murine Vδ1γδ T cells expressing interferon-γ and interleukin-17A in innate protection against Listeria monocytogenes infection. Immunology 125, 170–177 (2008).
Andrew, E. M. et al. Delineation of the function of a major γδ T cell subset during infection. J. Immunol. 175, 1741–1750 (2005).
Tsuchiya, T. et al. Role of γδ T cells in the inflammatory response of experimental colitis mice. J. Immunol. 171, 5507–5513 (2003).
Witherden, D. A. et al. The junctional adhesion molecule JAML is a costimulatory receptor for epithelial γδ T cell activation. Science 329, 1205–1210 (2010).
Lepage, A. C., Buzoni-Gatel, D., Bout, D. T. & Kasper, L. H. Gut-derived intraepithelial lymphocytes induce long term immunity against Toxoplasma gondii. J. Immunol. 161, 4902–4908 (1998). Describes a role for IFNγ-producing TCRαβ+CD8αα+IELs in the protection against an intestinal infection with parasites.
Roark, C. L., Simonian, P. L., Fontenot, A. P., Born, W. K. & O'Brien, R. L. γδ T cells: an important source of IL-17. Curr. Opin. Immunol. 20, 353–357 (2008).
Simpson, S. J. et al. Expression of pro-inflammatory cytokines by TCRαβ+ and TCRγδ+ T cells in an experimental model of colitis. Eur. J. Immunol. 27, 17–25 (1997).
Takagaki, Y., DeCloux, A., Bonneville, M. & Tonegawa, S. Diversity of γδ T-cell receptors on murine intestinal intra-epithelial lymphocytes. Nature 339, 712–714 (1989).
Kyes, S., Carew, E., Carding, S. R., Janeway, C. A. Jr & Hayday, A. Diversity in T-cell receptor γ gene usage in intestinal epithelium. Proc. Natl Acad. Sci. USA 86, 5527–5531 (1989). Together with reference 103, references 106 and 107describe the restricted repertoire of TCRγδ+ IELs.
Tanaka, Y., Morita, C. T., Nieves, E., Brenner, M. B. & Bloom, B. R. Natural and synthetic non-peptide antigens recognized by human γδ T cells. Nature 375, 155–158 (1995).
O'Brien, R. L. et al. Stimulation of a major subset of lymphocytes expressing T cell receptor γδ by an antigen derived from Mycobacterium tuberculosis. Cell 57, 667–674 (1989).
Yamagata, T., Benoist, C. & Mathis, D. A shared gene-expression signature in innate-like lymphocytes. Immunol. Rev. 210, 52–66 (2006).
Mixter, P. F. et al. A model for the origin of TCR-αβ+ CD4-CD8- B220+ cells based on high affinity TCR signals. J. Immunol. 162, 5747–5756 (1999).
Sewell, A. K. et al. Antagonism of cytotoxic T-lymphocyte activation by soluble CD8. Nature Med. 5, 399–404 (1999).
Cawthon, A. G., Lu, H. & Alexander-Miller, M. A. Peptide requirement for CTL activation reflects the sensitivity to CD3 engagement: correlation with CD8αβ versus CD8αα expression. J. Immunol. 167, 2577–2584 (2001).
Ma, C. S., Nichols, K. E. & Tangye, S. G. Regulation of cellular and humoral immune responses by the SLAM and SAP families of molecules. Annu. Rev. Immunol. 25, 337–379 (2007).
Masopust, D., Vezys, V., Marzo, A. L. & Lefrancois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001). Details preferential effector memory cell differentiation and cytotoxic function of intestinal CD8+TCRαβ+ IELs in response to infections.
Hansen, S. G. et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nature Med. 15, 293–299 (2009).
Dharakul, T. et al. Immunization with baculovirus-expressed recombinant rotavirus proteins VP1, VP4, VP6, and VP7 induces CD8+ T lymphocytes that mediate clearance of chronic rotavirus infection in SCID mice. J. Virol. 65, 5928–5932 (1991).
Kanwar, S. S., Ganguly, N. K., Walia, B. N. & Mahajan, R. C. Direct and antibody dependent cell mediated cytotoxicity against Giardia lamblia by splenic and intestinal lymphoid cells in mice. Gut 27, 73–77 (1986).
Masopust, D., Vezys, V., Wherry, E. J., Barber, D. L. & Ahmed, R. Cutting edge: gut microenvironment promotes differentiation of a unique memory CD8 T cell population. J. Immunol. 176, 2079–2083 (2006).
Masopust, D., Jiang, J., Shen, H. & Lefrancois, L. Direct analysis of the dynamics of the intestinal mucosa CD8 T cell response to systemic virus infection. J. Immunol. 166, 2348–2356 (2001).
Pope, C. et al. Organ-specific regulation of the CD8 T cell response to Listeria monocytogenes infection. J. Immunol. 166, 3402–3409 (2001).
Huleatt, J. W., Pilip, I., Kerksiek, K. & Pamer, E. G. Intestinal and splenic T cell responses to enteric Listeria monocytogenes infection: distinct repertoires of responding CD8 T lymphocytes. J. Immunol. 166, 4065–4073 (2001).
Mehandru, S. et al. Primary HIV-1 infection is associated with preferential depletion of CD4+ T lymphocytes from effector sites in the gastrointestinal tract. J. Exp. Med. 200, 761–770 (2004).
Li, Q. et al. Peak SIV replication in resting memory CD4+ T cells depletes gut lamina propria CD4+ T cells. Nature 434, 1148–1152 (2005).
Raffatellu, M. et al. Simian immunodeficiency virus-induced mucosal interleukin-17 deficiency promotes Salmonella dissemination from the gut. Nature Med. 14, 421–428 (2008).
Prendergast, A. et al. HIV-1 infection is characterized by profound depletion of CD161+ Th17 cells and gradual decline in regulatory T cells. AIDS 24, 491–502 (2010).
Epple, H. J. et al. Acute HIV infection induces mucosal infiltration with CD4+ and CD8+ T cells, epithelial apoptosis, and a mucosal barrier defect. Gastroenterology 139, 1289–1300 (2010).
Sugimoto, K. et al. IL-22 ameliorates intestinal inflammation in a mouse model of ulcerative colitis. J. Clin. Invest. 118, 534–544 (2008).
McGeachy, M. J. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell-mediated pathology. Nature Immunol. 8, 1390–1397 (2007).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Mucida, D. et al. Retinoic acid can directly promote TGF-β-mediated Foxp3+ Treg cell conversion of naive T cells. Immunity 30, 471–472; author reply 472–473 (2009).
Coombes, J. L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Sun, C. M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
Curtis, M. M. & Way, S. S. Interleukin-17 in host defence against bacterial, mycobacterial and fungal pathogens. Immunology 126, 177–185 (2009).
Conti, H. R. et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. J. Exp. Med. 206, 299–311 (2009).
O'Shea, J. J. & Paul, W. E. Mechanisms underlying lineage commitment and plasticity of helper CD4+ T cells. Science 327, 1098–1102 (2010).
Hershberg, R. M. et al. Highly polarized HLA class II antigen processing and presentation by human intestinal epithelial cells. J. Clin. Invest. 102, 792–803 (1998).
Khanna, R. et al. Class I processing-defective Burkitt's lymphoma cells are recognized efficiently by CD4+ EBV-specific CTLs. J. Immunol. 158, 3619–3625 (1997).
Alcami, A. & Koszinowski, U. H. Viral mechanisms of immune evasion. Immunol. Today 21, 447–455 (2000).
Giacomelli, R. et al. Increase of circulating γ/δ T lymphocytes in the peripheral blood of patients affected by active inflammatory bowel disease. Clin. Exp. Immunol. 98, 83–88 (1994).
Kanazawa, H., Ishiguro, Y., Munakata, A. & Morita, T. Multiple accumulation of Vδ2+ γδ T-cell clonotypes in intestinal mucosa from patients with Crohn's disease. Dig. Dis. Sci. 46, 410–416 (2001).
Yeung, M. M. et al. Characterisation of mucosal lymphoid aggregates in ulcerative colitis: immune cell phenotype and TcR-γδ expression. Gut 47, 215–227 (2000).
Kawaguchi-Miyashita, M. et al. An accessory role of TCRγδ+ cells in the exacerbation of inflammatory bowel disease in TCRα mutant mice. Eur. J. Immunol. 31, 980–988 (2001).
Mizoguchi, A. et al. Role of the CD5 molecule on TCR γδ T cell-mediated immune functions: development of germinal centers and chronic intestinal inflammation. Int. Immunol. 15, 97–108 (2003).
Park, S. G. et al. T regulatory cells maintain intestinal homeostasis by suppressing γδ T cells. Immunity 33, 791–803 (2010).
Guehler, S. R., Finch, R. J., Bluestone, J. A. & Barrett, T. A. Increased threshold for TCR-mediated signaling controls self reactivity of intraepithelial lymphocytes. J. Immunol. 160, 5341–5346 (1998).
Cheroutre, H. In IBD eight can come before four. Gastroenterology 131, 667–670 (2006).
Tajima, M. et al. IL-6-dependent spontaneous proliferation is required for the induction of colitogenic IL-17-producing CD8+ T cells. J. Exp. Med. 205, 1019–1027 (2008).
Nancey, S. et al. CD8+ cytotoxic T cells induce relapsing colitis in normal mice. Gastroenterology 131, 485–496 (2006).
Meresse, B. et al. Coordinated induction by IL15 of a TCR-independent NKG2D signaling pathway converts CTL into lymphokine-activated killer cells in celiac disease. Immunity 21, 357–366 (2004).
Hue, S. et al. A direct role for NKG2D/MICA interaction in villous atrophy during celiac disease. Immunity 21, 367–377 (2004). Together with reference 148, references 151 and 152 describe a pathway involving IL-15 that induces pathogenic cytotoxic IELs through NKG2D signalling.
Jabri, B. & Sollid, L. M. Tissue-mediated control of immunopathology in coeliac disease. Nature Rev. Immunol. 9, 858–870 (2009).
Jabri, B. et al. Selective expansion of intraepithelial lymphocytes expressing the HLA-E-specific natural killer receptor CD94 in celiac disease. Gastroenterology 118, 867–879 (2000).
Bodd, M. et al. HLA-DQ2-restricted gluten-reactive T cells produce IL-21 but not IL-17 or IL-22. Mucosal Immunol. 3, 594–601 (2010).
Monteleone, I. et al. Characterization of IL-17A-producing cells in celiac disease mucosa. J. Immunol. 184, 2211–2218 (2010).
Benahmed, M. et al. Inhibition of TGF-β signaling by IL-15: a new role for IL-15 in the loss of immune homeostasis in celiac disease. Gastroenterology 132, 994–1008 (2007).
Depaolo, R. W. et al. Co-adjuvant effects of retinoic acid and IL-15 induce inflammatory immunity to dietary antigens. Nature 471, 220–224 (2011). Reports an alternative role for retinoic acid, showing that, in the presence of IL-15, retinoic acid induces pro-inflammatory DCs, leading to the generation of pathogenic T cells in the gut.
Abraham, C. & Cho, J. H. Inflammatory bowel disease. N. Engl. J. Med. 361, 2066–2078 (2009).
Xavier, R. J. & Podolsky, D. K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 448, 427–434 (2007).
Uhlig, H. H. et al. Differential activity of IL-12 and IL-23 in mucosal and systemic innate immune pathology. Immunity 25, 309–318 (2006).
Hue, S. et al. Interleukin-23 drives innate and T cell-mediated intestinal inflammation. J. Exp. Med. 203, 2473–2483 (2006).
Duerr, R. H. et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 314, 1461–1463 (2006).
Targan, S. R. & Karp, L. C. Defects in mucosal immunity leading to ulcerative colitis. Immunol. Rev. 206, 296–305 (2005).
Saenz, S. A., Noti, M. & Artis, D. Innate immune cell populations function as initiators and effectors in Th2 cytokine responses. Trends Immunol. 31, 407–413 (2010).
Veillette, A., Bookman, M. A., Horak, E. M. & Bolen, J. B. The CD4 and CD8 T cell surface antigens are associated with the internal membrane tyrosine-protein kinase p56lck. Cell 55, 301–308 (1988).
Arcaro, A. et al. Essential role of CD8 palmitoylation in CD8 coreceptor function. J. Immunol. 165, 2068–2076 (2000).
Crooks, M. E. & Littman, D. R. Disruption of T lymphocyte positive and negative selection in mice lacking the CD8β chain. Immunity 1, 277–285 (1994).
Hayday, A. & Gibbons, D. Brokering the peace: the origin of intestinal T cells. Mucosal Immunol. 1, 172–174 (2008).
Kanamori, Y. et al. Identification of novel lymphoid tissues in murine intestinal mucosa where clusters of c-kit+ IL-7R+ Thy1+ lympho-hemopoietic progenitors develop. J. Exp. Med. 184, 1449–1459 (1996).
Saito, H. et al. Generation of intestinal T cells from progenitors residing in gut cryptopatches. Science 280, 275–278 (1998).
Lambolez, F. et al. Characterization of T cell differentiation in the murine gut. J. Exp. Med. 195, 437–449 (2002).
De Geus, B. et al. Phenotype of intraepithelial lymphocytes in euthymic and athymic mice: implications for differentiation of cells bearing a CD3-associated γδ T cell receptor. Eur. J. Immunol. 20, 291–298 (1990).
Bandeira, A. et al. Extrathymic origin of intestinal intraepithelial lymphocytes bearing T-cell antigen receptor γδ. Proc. Natl Acad. Sci. USA 88, 43–47 (1991).
Naito, T., Shiohara, T., Hibi, T., Suematsu, M. & Ishikawa, H. RORγt is dispensable for the development of intestinal mucosal T cells. Mucosal Immunol. 1, 198–207 (2008).
Lambolez, F. et al. The thymus exports long-lived fully committed T cell precursors that can colonize primary lymphoid organs. Nature Immunol. 7, 76–82 (2006).
Acknowledgements
This is the manuscript number 1364 of the La Jolla Institute for Allergy and Immunology, California, USA. We thank F. van Wijk for helpful discussions and M. Cheroutre for her contribution. Work in the H.C. laboratory is supported by the National Institutes of Health (RO1 AI050265-06) and the La Jolla Institute for Allergy and Immunology. Work in the D.M. laboratory is supported by The Rockefeller University, New York, USA, and by the Crohn's & Colitis Foundation of America.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Pathogens
-
Opportunistic organisms that cause acute or chronic disease following host infection. Derived from the Greek word 'pathos', which means 'suffering'.
- Intraepithelial lymphocytes
-
(IELs). These lymphocyte populations consist mostly of T cells and are found within the epithelial layer of mammalian mucosal linings, such as the gastrointestinal tract and reproductive tract. However, unlike conventional naive T cells, IELs are antigen- experienced T cells and, on encountering antigens, they immediately release cytokines or mediate killing of infected target cells.
- Thymus leukaemia antigen
-
(TLA). A non-polymorphic, non-classical MHC class I molecule (MHC class I-b family) with a restricted expression pattern. It is constitutively expressed on intestinal epithelial cells and can be induced on antigen-presenting cells. TLA is structurally incapable of binding or presenting peptide antigens and it does not engage with T cell receptors. However, the α3 extracellular domain of TLA interacts with CD8α. TLA displays stronger affinity for CD8αα homodimers compared with CD8αβ heterodimers, and CD8αα expression can be detected with TLA-specific tetramers.
- Lamina propria
-
Connective tissue that underlies the epithelium of the mucosa and contains various myeloid and lymphoid cells, including macrophages, dendritic cells, T cells and B cells.
- Microbiota
-
The microorganisms present in normal, healthy individuals. These microorganisms live mostly in the digestive tract but are also found in some other tissues.
- Germ-free mice
-
Mice born and raised in sterile isolators. They are devoid of colonizing microorganisms, but after they have been experimentally colonized by known bacteria, they are said to be gnotobiotic.
- Gut-associated lymphoid tissues
-
Lymphoid structures and aggregates associated with the intestinal mucosa, specifically the tonsils, Peyer's patches, lymphoid follicles, appendix and caecal patch. Enriched in lymphocytes and specialized dendritic cell and macrophage subsets.
- Peyer's patches
-
Groups of lymphoid nodules present in the small intestine (usually the ileum). They occur in the intestinal wall, opposite the line of attachment of the mesentery. They consist of a dome area, B cell follicles and interfollicular T cell areas. High endothelial venules are present mainly in the interfollicular areas.
- Mesenteric lymph nodes
-
Lymph nodes, located at the base of the mesentery, that collect lymph (including cells and antigens) draining from the intestinal mucosa.
- Microfold cells
-
(M cells). Specialized antigen-sampling cells that are located in the follicle-associated epithelium of the organized mucosa-associated lymphoid tissues. M cells deliver antigens by transepithelial vesicular transport from the aero-digestive lumen directly to subepithelial lymphoid tissues of nasopharynx-associated lymphoid tissue and Peyer's patches.
- NKG2D
-
(Natural killer group 2, member D). A lectin-type activating receptor that is encoded by the NK complex and is expressed at the surface of NK cells, NKT cells, natural and induced intraepithelial lymphocytes and conventional T cell receptor-γδ (TCRγδ) T cells, as well as some conventional cytolytic CD8αβ+TCRαβ+ T cells. The ligands for NKG2D are MHC class I polypeptide-related sequence A (MICA) and MICB in humans, and retinoic acid early transcript 1 (RAE1) and H60 in mice. Such ligands are generally expressed at the surface of infected, stressed or transformed cells.
- Inflammatory bowel disease
-
A chronic condition of the intestine that is characterized by severe inflammation and mucosal tissue destruction. The most common forms in humans are ulcerative colitis and Crohn's disease.
- Coeliac disease
-
Coeliac disease is a condition that damages the lining of the small intestine and interferes with nutrient absorption. The damage is due to an aberrant immune response to gluten-derived antigens, which are found in wheat, barley, rye and possibly oats.
- Crohn's disease
-
A form of chronic inflammatory bowel disease that can affect the entire gastrointestinal tract but is most common in the colon and terminal ileum. It is characterized by transmural inflammation, strictures and granuloma formation, and it is thought to result from an abnormal T cell-mediated immune response to commensal bacteria.
- Ulcerative colitis
-
A chronic disease that is characterized by inflammation of the mucosa and sub-mucosa tissues, mainly of the large intestine.
Rights and permissions
About this article
Cite this article
Cheroutre, H., Lambolez, F. & Mucida, D. The light and dark sides of intestinal intraepithelial lymphocytes. Nat Rev Immunol 11, 445–456 (2011). https://doi.org/10.1038/nri3007
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3007
- Springer Nature Limited
This article is cited by
-
Activation of mucosal insulin receptor exacerbates intestinal inflammation by promoting tissue resident memory T cells differentiation through EZH2
Journal of Translational Medicine (2024)
-
p53 suppresses MHC class II presentation by intestinal epithelium to protect against radiation-induced gastrointestinal syndrome
Nature Communications (2024)
-
The transcription factor Aiolos restrains the activation of intestinal intraepithelial lymphocytes
Nature Immunology (2024)
-
TGF-β controls development of TCRγδ+CD8αα+ intestinal intraepithelial lymphocytes
Cell Discovery (2023)
-
Gut microbial fatty acid isomerization modulates intraepithelial T cells
Nature (2023)