Abstract
The major dietary sources of trans fatty acids (TFAs) in most countries are partially hydrogenated vegetable oils. TFA consumption is a modifiable dietary risk factor for metabolic syndrome, diabetes mellitus, and coronary heart disease. Here, we review the available data on various effects of TFAs, including metabolic and signaling pathways that mediate these effects, affected tissues, and relationships with clinical end points. TFA consumption causes metabolic dysfunction: it adversely affects circulating lipid levels, triggers systemic inflammation, induces endothelial dysfunction, and, according to some studies, increases visceral adiposity, body weight, and insulin resistance. Dietary TFAs influence the function of multiple cell types, including hepatocytes, adipocytes, macrophages and endothelial cells. Among dietary fats and nutrients, TFAs seem to have a unique cardiometabolic imprint that is linked to insulin-resistance and metabolic-syndrome pathways. Consistent with these adverse physiological effects, consumption of even small amounts of TFAs (2% of total energy intake) is consistently associated with a markedly increased incidence of coronary heart disease. Relationships between TFA consumption and diabetes mellitus have been less consistent, possibly owing to differences in study designs. Nevertheless, the documented adverse effects of TFAs underscore their potential to cause harm and the importance of policy measures to minimize consumption of industrially produced TFAs.
Key Points
-
Dietary trans fatty acids (TFAs) derive from industrial, partial hydrogenation of vegetable oils or from ruminant products
-
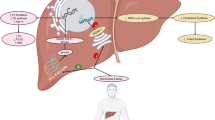
Dietary TFAs influence the function of multiple cell types, including hepatocytes, adipocytes, macrophages and endothelial cells
-
TFA consumption affects multiple metabolic risk factors, including lipid and lipoprotein levels, systemic inflammation, endothelial function, adiposity, and glucose–insulin homeostasis
-
TFA consumption increases risk of clinical coronary heart disease and likelihood of development of diabetes mellitus


Similar content being viewed by others
References
National Institute of Diabetes and Digestive and Kidney Diseases, NIH. Incidence of diagnosed diabetes among people aged 20 years or older, United States, 2007. National Diabetes Statistics [online] (2008).
American Heart Association. Heart Disease and Stroke Statistics—Update 2008. [online] (2008).
WHO. Obesity and Overweight. Fact Sheet No 311. [online] (2006).
Allison, D. B. et al. Estimated intakes of trans fatty and other fatty acids in the US population. J. Am. Diet. Assoc. 99, 166–174 (1999).
Mozaffarian, D., Abdollahi, M., Campos, H., Houshiarrad, A. & Willett, W. C. Consumption of trans fats and estimated effects on coronary heart disease in Iran. Eur. J. Clin. Nutr. 61, 1004–1010 (2007).
Stender, S., Dyerberg, J. & Astrup, A. High levels of industrially produced trans fat in popular fast foods. N. Engl. J. Med. 354, 1650–1652 (2006).
Dietary Guidelines Advisory Committee. Report of the Dietary Guidelines Advisory Committee on the dietary guidelines for Americans. US Department of Health Services Website [online] (2005).
Lock, A. L. & Bauman, D. E. Modifying milk fat composition of dairy cows to enhance fatty acids beneficial to human health. Lipids 39, 1197–1206 (2004).
Risérus, U., Vessby, B., Arnlöv, J. & Basu, S. Effects of cis-9, trans-11 conjugated linoleic acid supplementation on insulin sensitivity, lipid peroxidation, and proinflammatory markers in obese men. Am. J. Clin. Nutr. 80, 279–283 (2004).
Moloney, F., Yeow, T. P., Mullen, A., Nolan, J. J. & Roche, H. M. Conjugated linoleic acid supplementation, insulin sensitivity, and lipoprotein metabolism in patients with type 2 diabetes mellitus. Am. J. Clin. Nutr. 80, 887–895 (2004).
Smedman, A., Vessby, B. & Basu, S. Isomer-specific effects of conjugated linoleic acid on lipid peroxidation in humans: regulation by α-tocopherol and cyclo-oxygenase-2 inhibitor. Clin. Sci. (Lond.) 106, 67–73 (2004).
Nugent, A. P. et al. The effects of conjugated linoleic acid supplementation on immune function in healthy volunteers. Eur. J. Clin. Nutr. 59, 742–750 (2005).
Chardigny, J. M. et al. Do trans fatty acids from industrially produced sources and from natural sources have the same effect on cardiovascular disease risk factors in healthy subjects? Results of the Trans Fatty Acids Collaboration (TRANSFACT) study. Am. J. Clin. Nutr. 87, 558–566 (2008).
Motard-Belanger, A. et al. Study of the effect of trans fatty acids from ruminants on blood lipids and other risk factors for cardiovascular disease. Am. J. Clin. Nutr. 87, 593–599 (2008).
Willett, W. & Mozaffarian, D. Ruminant or industrial sources of trans fatty acids: public health issue or food label skirmish? Am. J. Clin. Nutr. 87, 515–516 (2008).
Oomen, C. M. et al. Association between trans fatty acid intake and 10-year risk of coronary heart disease in the Zutphen Elderly Study: a prospective population-based study. Lancet 357, 746–751 (2001).
Pietinen, P. et al. Intake of fatty acids and risk of coronary heart disease in a cohort of Finnish men. The α-tocopherol, β-carotene cancer prevention study. Am. J. Epidemiol. 145, 876–887 (1997).
Willett, W. C. et al. Intake of trans fatty acids and risk of coronary heart disease among women. Lancet 341, 581–585 (1993).
Jakobsen, M. U. et al. Intake of ruminant trans fatty acids and risk of coronary heart disease—an overview. Atheroscler. Suppl. 7, 9–11 (2006).
Jakobsen, M. U., Overvad, K., Dyerberg, J. & Heitmann, B. L. Intake of ruminant trans fatty acids and risk of coronary heart disease. Int. J. Epidemiol. 37, 173–182 (2008).
Mozaffarian, D. & Clarke, R. Quantitative effects on cardiovascular risk factors and coronary heart disease risk of replacing partially hydrogenated vegetable oils with other fats and oils. Eur. J. Clin. Nutr. (in press).
Mauger, J. F. et al. Effect of different forms of dietary hydrogenated fats on LDL particle size. Am. J. Clin. Nutr. 78, 370–375 (2003).
Mozaffarian, D., Aro, A. & Willett, W. C. Health effects of trans-fatty acids: experimental and observational evidence. Eur. J. Clin. Nutr. (in press).
Baer, D. J., Judd, J. T., Clevidende, B. A. & Tracy, R. P. Dietary fatty acids affect plasma markers of inflammation in healthy men fed controlled diets: a randomized crossover study. Am. J. Clin. Nutr. 79, 969–973 (2004).
Han, S. N. et al. Effect of hydrogenated and saturated, relative to polyunsaturated, fat on immune and inflammatory responses of adults with moderate hypercholesterolemia. J. Lipid Res. 43, 445–452 (2002).
Mozaffarian, D. et al. Dietary intake of trans fatty acids and systemic inflammation in women. Am. J. Clin. Nutr. 79, 606–612 (2004).
Lopez-Garcia, E. et al. Consumption of trans fatty acids is related to plasma biomarkers of inflammation and endothelial dysfunction. J. Nutr. 135, 562–566 (2005).
Ridker, P. M., Hennekens, C. H., Buring, J. E. & Rifai, N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N. Engl. J. Med. 342, 836–843 (2000).
de Roos, N. M., Bots, M. L. & Katan, M. B. Replacement of dietary saturated fatty acids by trans fatty acids lowers serum HDL cholesterol and impairs endothelial function in healthy men and women. Arterioscler. Thromb. Vasc. Biol. 21, 1233–1237 (2001).
Dyerberg, J. et al. Effects of trans- and n-3 unsaturated fatty acids on cardiovascular risk markers in healthy males. An 8 weeks dietary intervention study. Eur. J. Clin. Nutr. 58, 1062–1070 (2004).
Kavanagh, K. et al. Trans fat diet induces abdominal obesity and changes in insulin sensitivity in monkeys. Obesity (Silver Spring) 15, 1675–1684 (2007).
Koh-Banerjee, P. et al. Prospective study of the association of changes in dietary intake, physical activity, alcohol consumption, and smoking with 9-y gain in waist circumference among 16,587 US men. Am. J. Clin. Nutr. 78, 719–727 (2003).
Field, A. E., Willett, W. C., Lissner, L. & Colditz, G. A. Dietary fat and weight gain among women in the Nurses' Health Study. Obesity (Silver Spring) 15, 967–976 (2007).
Christiansen, E., Schnider, S., Palmvig, B., Tauber-Lassen, E. & Pedersen, O. Intake of a diet high in trans monounsaturated fatty acids or saturated fatty acids. Effects on postprandial insulinemia and glycemia in obese patients with NIDDM. Diabetes Care 20, 881–887 (1997).
Vega-Lopez, S., Ausman, L. M., Jalbert, S. M., Erkkilä, A. T & Lichtenstein, A. H. Palm and partially hydrogenated soybean oils adversely alter lipoprotein profiles compared with soybean and canola oils in moderately hyperlipidemic subjects. Am. J. Clin. Nutr. 84, 54–62 (2006).
Lefevre, M. et al. Comparison of the acute response to meals enriched with cis- or trans-fatty acids on glucose and lipids in overweight individuals with differing FABP2 genotypes. Metabolism 54, 1652–1658 (2005).
Lovejoy, J. C. et al. Effects of diets enriched in saturated (palmitic), monounsaturated (oleic), or trans (elaidic) fatty acids on insulin sensitivity and substrate oxidation in healthy adults. Diabetes Care 25, 1283–1288 (2002).
Louheranta, A. M., Turpeinen, A. K., Vidgren, H. M, Schwab, U. S. & Uusitupa, M. I. A high-trans fatty acid diet and insulin sensitivity in young healthy women. Metabolism 48, 870–875 (1999).
Axen, K. V., Dikeakos, A. & Sclafani, A. High dietary fat promotes syndrome X in nonobese rats. J. Nutr. 133, 2244–2249 (2003).
Ibrahim, A., Natrajan, S. & Ghafoorunissa, R. Dietary trans-fatty acids alter adipocyte plasma membrane fatty acid composition and insulin sensitivity in rats. Metabolism 54, 240–246 (2005).
Natarajan, S., Ghafoorunissa, R. & Ibrahim, A. Dietary trans fatty acids alter diaphragm phospholipid fatty acid composition, triacylglycerol content and glucose transport in rats. Br. J. Nutr. 93, 829–833 (2005).
Albuquerque, K. T. et al. Intake of trans fatty acid-rich hydrogenated fat during pregnancy and lactation inhibits the hypophagic effect of central insulin in the adult offspring. Nutrition 22, 820–829 (2006).
Pisani, L. P. et al. Hydrogenated fat diet intake during pregnancy and lactation modifies the PAI-1 gene expression in white adipose tissue of offspring in adult life. Lipids Health Dis. 7, 13 (2008).
Osso, F. S. et al. Trans fatty acids in maternal milk lead to cardiac insulin resistance in adult offspring. Nutrition 24, 727–732 (2008).
Assumpcão, R. P., dos Santos, F. D., de Mattos Machado Andrade, P., Barreto, G. F. & das Graças Tavares do Carmo, M. Effect of variation of trans-fatty acid in lactating rats' diet on lipoprotein lipase activity in mammary gland, liver, and adipose tissue. Nutrition 20, 806–811 (2004).
Innis, S. M. & King, D. J. Trans fatty acids in human milk are inversely associated with concentrations of essential all-cis n-6 and n-3 fatty acids and determine trans, but not n-6 and n-3, fatty acids in plasma lipids of breast-fed infants. Am. J. Clin. Nutr. 70, 383–390 (1999).
Mozaffarian, D. & Willett, W. C. Trans fatty acids and cardiovascular risk: a unique cardiometabolic imprint? Curr. Atheroscler. Rep. 9, 486–493 (2007).
Dashti, N., Feng, Q., Freeman, M. R., Gandhi, M. & Franklin, F. A. Trans polyunsaturated fatty acids have more adverse effects than saturated fatty acids on the concentration and composition of lipoproteins secreted by human hepatoma HepG2 cells. J. Nutr. 132, 2651–2659 (2002).
Mitmesser, S. H. & Carr, T. P. Trans fatty acids alter the lipid composition and size of apo-B100-containing lipoproteins secreted by HepG2 cells. J. Nutr. Biochem. 16, 178–183 (2005).
Matthan, N. R. et al. Hydrogenated fat consumption affects acylation-stimulating protein levels and cholesterol esterification rates in moderately hypercholesterolemic women. J. Lipid Res. 42, 1841–1848 (2001).
Abbey, M. & Nestel, P. J. Plasma cholesteryl ester transfer protein activity is increased when trans-elaidic acid is substituted for cis-oleic acid in the diet. Atherosclerosis 106, 99–107 (1994).
Lee, J. Y. & Carr, T. P. Dietary fatty acids regulate acyl-CoA:cholesterol acyltransferase and cytosolic cholesteryl ester hydrolase in hamsters. J. Nutr. 134, 3239–3244 (2004).
Kummerow, F. A., Mahfouz, M. M. & Zhou, Q. Trans fatty acids in partially hydrogenated soybean oil inhibit prostacyclin release by endothelial cells in presence of high level of linoleic acid. Prostaglandins Other Lipid Mediat. 84, 138–153 (2007).
Diamond, F. B. Jr & Eichler, D. C. Leptin and the adipocyte endocrine system. Crit. Rev. Clin. Lab. Sci. 39, 499–525 (2002).
Niu, S. L. et al. Reduced G protein-coupled signaling efficiency in retinal rod outer segments in response to n-3 fatty acid deficiency. J. Biol. Chem. 279, 31098–31104 (2004).
Niu, S. L., Mitchell, D. C. & Litman, B. J. Trans fatty acid derived phospholipids show increased membrane cholesterol and reduced receptor activation as compared to their cis analogs. Biochemistry 44, 4458–4465 (2005).
Saravanan, N., Haseeb, A., Ehtesham, N. Z. & Ghafoorunissa, R. Differential effects of dietary saturated and trans-fatty acids on expression of genes associated with insulin sensitivity in rat adipose tissue. Eur. J. Endocrinol. 153, 159–165 (2005).
Mozaffarian, D., Katan, M. B., Ascherio, A., Stampfer, M. J. & Willett, W. C. Trans fatty acids and cardiovascular disease. N. Engl. J. Med. 354, 1601–1613 (2006).
Mensink, R. P., Zock, P. L., Kester, A. D. & Katan, M. B. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials. Am. J. Clin. Nutr. 77, 1146–1155 (2003).
van Dam, R. M., Willett, W. C., Rimm, E. B., Stampfer, M. J. & Hu, F. B. Dietary fat and meat intake in relation to risk of type 2 diabetes in men. Diabetes Care 25, 417–424 (2002).
Meyer, K. A., Kushi, L. H., Jacobs, D. R. Jr & Folsom, A. R. Dietary fat and incidence of type 2 diabetes in older Iowa women. Diabetes Care 24, 1528–1535 (2001).
Hu, F. B. et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N. Engl. J. Med. 345, 790–797 (2001).
Salmerón, J. et al. Dietary fat intake and risk of type 2 diabetes in women. Am. J. Clin. Nutr. 73, 1019–1026 (2001).
Radesky, J. S. et al. Diet during early pregnancy and development of gestational diabetes. Paediatr. Perinat. Epidemiol. 22, 47–59 (2008).
WHO. Diet, nutrition and the prevention of chronic diseases: report of a joint WHO/FAO expert consultation. World Health Organ Tech. Rep. Ser. 916, 1–49 (2003).
Leth, T., Jensen, H. G., Mikkelsen, A. A. & Bysted, A. The effect of the regulation on trans fatty acid content in Danish food. Atheroscler. Suppl. 7, 53–56 (2006).
New York City Department of Health and Mental Hygiene. Healthy heart—avoid trans fat. Cardiovascular Disease Prevention [online] (2009).
Acknowledgements
The authors' research was supported by grants from the NIH, National Heart, Lung and Blood Institute (R01 HL 085710-01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Micha, R., Mozaffarian, D. Trans fatty acids: effects on metabolic syndrome, heart disease and diabetes. Nat Rev Endocrinol 5, 335–344 (2009). https://doi.org/10.1038/nrendo.2009.79
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2009.79
- Springer Nature Limited
This article is cited by
-
Association of systemic immunity-inflammation index with metabolic syndrome in U.S. adult: a cross-sectional study
BMC Geriatrics (2024)
-
Enzyme engineering for functional lipids synthesis: recent advance and perspective
Bioresources and Bioprocessing (2024)
-
Nutrient patterns in relation to metabolic health status and serum levels of brain-derived neurotrophic factor (BDNF) and adropin in adults
Scientific Reports (2024)
-
Adherence to diet with higher dietary diabetes risk reduction score is associated with reduced risk of type 2 diabetes incident in Iranian adults
BMC Public Health (2023)
-
The effects of trans fat diet intake on metabolic parameters and pancreatic tissue in offspring of prenatal bisphenol A exposed rats
Scientific Reports (2023)