Key Points
-
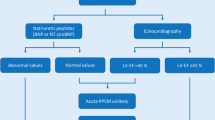
Peripartum cardiomyopathy (PPCM) is defined as idiopathic systolic dysfunction in peripartum women
-
To make a diagnosis of PPCM, other possible causes of heart failure in peripartum women, such as genetic forms of dilated cardiomyopathy, need to be excluded
-
The incidence and prognosis of PPCM vary according to socioeconomic and genetic factors
-
The aetiology of PPCM is unknown; risk factors might include pre-eclampsia, twin pregnancies, and African ethnicity
-
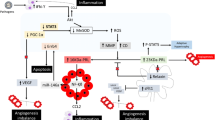
A possible pathophysiological mechanism for PPCM is the production of a 16 kDa fragment of prolactin; blocking prolactin is, therefore, a potential therapeutic target
Abstract
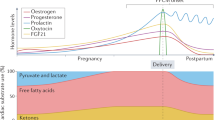
Cardiovascular diseases are a major cause of complications in pregnancy worldwide, and the number of patients who develop cardiac problems during pregnancy is increasing. Peripartum cardiomyopathy (PPCM) is a potentially life-threatening heart disease that emerges towards the end of pregnancy or in the first months postpartum, in previously healthy women. Symptoms and signs of PPCM are similar to those in patients with idiopathic dilated cardiomyopathy. The incidence varies geographically, most likely because of socioeconomic and genetic factors. The syndrome is associated with a high morbidity and mortality, and diagnosis is often delayed. Various mechanisms have been investigated, including the hypothesis that unbalanced peripartum or postpartum oxidative stress triggers the proteolytic cleavage of the nursing hormone prolactin into a potent antiangiogenic, proapoptotic, and proinflammatory 16 kDa fragment. This theory provides the basis for the discovery of disease-specific biomarkers and promising novel therapeutic targets. In this Review, we describe the latest understanding of the epidemiology, pathophysiology, and novel treatment strategies for patients with PPCM.


Similar content being viewed by others
References
Pearson, G. D. et al. Peripartum cardiomyopathy: National Heart, Lung, and Blood Institute and Office of Rare Diseases (National Institutes of Health) workshop recommendations and review. JAMA 283, 1183–1188 (2000).
Selle, T., Renger, I., Labidi, S., Bultmann, I. & Hilfiker-Kleiner, D. Reviewing peripartum cardiomyopathy: current state of knowledge. Future Cardiol. 5, 175–189 (2009).
Sliwa, K., Fett, J. & Elkayam, U. Peripartum cardiomyopathy. Lancet 368, 687–693 (2006).
Sliwa, K. et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of peripartum cardiomyopathy: a position statement from the Heart Failure Association of the European Society of Cardiology Working Group on Peripartum Cardiomyopathy. Eur. Heart J. 12, 767–778 (2010).
Demakis, J. G. & Rahimtoola, S. H. Peripartum cardiomyopathy. Circulation 44, 964–968 (1971).
Elkayam, U. et al. Pregnancy-associated cardiomyopathy: clinical characteristics and a comparison between early and late presentation. Circulation 111, 2050–2055 (2005).
Haghikia, A. et al. Phenotyping and outcome on contemporary management in a German cohort of patients with peripartum cardiomyopathy. Basic Res. Cardiol. 108, 366 (2013).
Goland, S. et al. Clinical profile and predictors of complications in peripartum cardiomyopathy. J. Card. Fail. 15, 645–650 (2009).
Sliwa, K. et al. Peripartum cardiomyopathy: inflammatory markers as predictors of outcome in 100 prospectively studied patients. Eur. Heart J. 27, 441–446 (2006).
Halkein, J. et al. MicroRNA-146a is a therapeutic target and biomarker for peripartum cardiomyopathy. J. Clin. Invest. 123, 2143–2154 (2013).
Hilfiker-Kleiner, D. et al. A cathepsin D-cleaved 16 kDa form of prolactin mediates postpartum cardiomyopathy. Cell 128, 589–600 (2007).
Patten, I. S. et al. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature 485, 333–338 (2012).
Forster, O. et al. Reversal of IFN-γ, oxLDL and prolactin serum levels correlate with clinical improvement in patients with peripartum cardiomyopathy. Eur. J. Heart Fail. 10, 861–868 (2008).
Ricke-Hoch, M. et al. Opposing roles of Akt and STAT3 in protection of the maternal heart from peripartum stress. Cardiovasc. Res. 101, 587–596 (2014).
Kamiya, C. A. et al. Different characteristics of peripartum cardiomyopathy between patients complicated with and without hypertensive disorders: results from the Japanese Nationwide survey of peripartum cardiomyopathy. Circ. J. 75, 1975–1981 (2011).
Brar, S. S. et al. Incidence, mortality, and racial differences in peripartum cardiomyopathy. Am. J. Cardiol. 100, 302–304 (2007).
Fett, J. D. Peripartum cardiomyopathy: insights from Haiti regarding a disease of unknown etiology. Minn. Med. 85, 46–48 (2002).
Fett, J. D., Christie, L. G., Carraway, R. D. & Murphy, J. G. Five-year prospective study of the incidence and prognosis of peripartum cardiomyopathy at a single institution. Mayo Clin. Proc. 80, 1602–1606 (2005).
Kuklina, E. V. & Callaghan, W. M. Cardiomyopathy and other myocardial disorders among hospitalizations for pregnancy in the United States: 2004–2006. Obstet. Gynecol. 115, 93–100 (2010).
Fett, J. D. et al. Unrecognized peripartum cardiomyopathy in Haitian women. Int. J. Gynaecol. Obstet. 90, 161–166 (2005).
Desai, D., Moodley, J. & Naidoo, D. Peripartum cardiomyopathy: experiences at King Edward VIII Hospital, Durban, South Africa and a review of the literature. Trop. Doct. 25, 118–123 (1995).
Chapa, J. B. et al. Prognostic value of echocardiography in peripartum cardiomyopathy. Obstet. Gynecol. 105, 1303–1308 (2005).
Gentry, M. B. et al. African-American women have a higher risk for developing peripartum cardiomyopathy. J. Am. Coll. Cardiol. 55, 654–659 (2010).
Modi, K. A., Illum, S., Jariatul, K., Caldito, G. & Reddy, P. C. Poor outcome of indigent patients with peripartum cardiomyopathy in the United States. Am. J. Obstet. Gynecol. 201, 171.e1–e5 (2009).
Goland, S., Modi, K., Hatamizadeh, P. & Elkayam, U. Differences in clinical profile of African–American women with peripartum cardiopmyopathy in the United States. J. Card. Fail. 19, 214–218 (2013).
Blauwet, L. A. et al. Predictors of outcome in 176 South African patients with peripartum cardiomyopathy. Heart 99, 308–313 (2013).
Mielniczuk, L. M. et al. Frequency of peripartum cardiomyopathy. Am. J. Cardiol. 97, 1765–1768 (2006).
European Society of Cardiology. PeriPartum CardioMyopathy (PPCM) Registry [online], (2014).
Elkayam, U. Clinical characteristics of peripartum cardiomyopathy in the United States: diagnosis, prognosis, and management. J. Am. Coll. Cardiol. 58, 659–670 (2011).
Safirstein, J. G. et al. Predictors of left ventricular recovery in a cohort of peripartum cardiomyopathy patients recruited via the internet. Int. J. Cardiol. 154, 27–31 (2012).
Sliwa, K. et al. EURObservational Research Programme: a worldwide registry on peripartum cardiomyopathy (PPCM) in conjunction with the Heart Failure Association of the European Society of Cardiology Working Group on PPCM. Eur. J. Heart Fail. http://dx.doi.org/10.1002/ejhf.68.
Pearl, W. Familial occurrence of peripartum cardiomyopathy. Am. Heart J. 129, 421–422 (1995).
Pierce, J. A., Price, B. O. & Joyce, J. W. Familial occurrence of postpartal heart failure. Arch. Intern. Med. 111, 651–655 (1963).
Meyer, G. P. et al. Bromocriptine treatment associated with recovery from peripartum cardiomyopathy in siblings: two case reports. J. Med. Case Reports 4, 80 (2010).
Fett, J. D., Sundstrom, B. J., Etta King, M. & Ansari, A. A. Mother–daughter peripartum cardiomyopathy. Int. J. Cardiol. 86, 331–332 (2002).
van Spaendonck-Zwarts, K. et al. Peripartum cardiomyopathy as part of familial dilated cardiomyopathy. Circulation 121, 2169–2175 (2010).
Morales, A. et al. Rare variant mutations in pregnancy-associated or peripartum cardiomyopathy. Circulation 121, 2176–2182 (2010).
van Spaendonck-Zwarts, K. Y. et al. Titin gene mutations are common in families with both peripartum cardiomyopathy and dilated cardiomyopathy. Eur. Heart J. http://dx.doi.org/10.1093/eurheartj/ehu050.
Bahl, A., Swamy, A., Sharma, Y. & Kumar, N. Isolated noncompaction of left ventricle presenting as peripartum cardiomyopathy. Int. J. Cardiol. 109, 422–423 (2006).
Lea, B., Bailey, A. L., Wiisanen, M. E., Attili, A. & Rajagopalan, N. Left ventricular noncompaction presenting as peripartum cardiomyopathy. Int. J. Cardiol. 154, e65–e66 (2012).
Rehfeldt, K. H., Pulido, J. N., Mauermann, W. J. & Click, R. L. Left ventricular hypertrabeculation/noncompaction in a patient with peripartum cardiomyopathy. Int. J. Cardiol. 139, e18–e20 (2010).
Hilfiker-Kleiner, D., Struman, I., Hoch, M., Podewski, E. & Sliwa, K. 16-kDa prolactin and bromocriptine in postpartum cardiomyopathy. Curr. Heart Fail. Rep. 9, 174–182 (2012).
Toescu, V., Nuttall, S. L., Martin, U., Kendall, M. J. & Dunne, F. Oxidative stress and normal pregnancy. Clin. Endocrinol. (Oxf.) 57, 609–613 (2002).
Lkhider, M., Castino, R., Bouguyon, E., Isidoro, C. & Ollivier-Bousquet, M. Cathepsin D released by lactating rat mammary epithelial cells is involved in prolactin cleavage under physiological conditions. J. Cell Sci. 117, 5155–5164 (2004).
Ferrara, N., Clapp, C. & Weiner, R. The 16K fragment of prolactin specifically inhibits basal or fibroblast growth factor stimulated growth of capillary endothelial cells. Endocrinology 129, 896–900 (1991).
Piwnica, D. et al. Cathepsin D processes human prolactin into multiple 16K-like N-terminal fragments: study of their antiangiogenic properties and physiological relevance. Mol. Endocrinol. 18, 2522–2542 (2004).
Macotela, Y. et al. Matrix metalloproteases from chondrocytes generate an antiangiogenic 16 kDa prolactin. J. Cell Sci. 119, 1790–1800 (2006).
Tabruyn, S. P., Nguyen, N. Q., Cornet, A. M., Martial, J. A. & Struman, I. The antiangiogenic factor, 16-kDa human prolactin, induces endothelial cell cycle arrest by acting at both the G0–G1 and the G2–M phases. Mol. Endocrinol. 19, 1932–1942 (2005).
D'Angelo, G. et al. 16K human prolactin inhibits vascular endothelial growth factor-induced activation of Ras in capillary endothelial cells. Mol. Endocrinol. 13, 692–704 (1999).
Tabruyn, S. P. et al. The antiangiogenic factor 16K human prolactin induces caspase-dependent apoptosis by a mechanism that requires activation of nuclear factor-κB. Mol. Endocrinol. 17, 1815–1823 (2003).
Lee, S. H., Kunz, J., Lin, S. H. & Yu-Lee, L. Y. 16-kDa prolactin inhibits endothelial cell migration by down-regulating the Ras–Tiam1–Rac1–Pak1 signaling pathway. Cancer Res. 67, 11045–11053 (2007).
Gonzalez, C. et al. 16K-prolactin inhibits activation of endothelial nitric oxide synthase, intracellular calcium mobilization, and endothelium-dependent vasorelaxation. Endocrinology 145, 5714–5722 (2004).
Gonzalez, C. et al. Elevated vasoinhibins may contribute to endothelial cell dysfunction and low birth weight in preeclampsia. Lab. Invest. 87, 1009–1017 (2007).
Nguyen, N. Q. et al. Inhibition of tumor growth and metastasis establishment by adenovirus-mediated gene transfer delivery of the antiangiogenic factor 16K hPRL. Mol. Ther. 15, 2094–2100 (2007).
Habedank, D. et al. Recovery from peripartum cardiomyopathy after treatment with bromocriptine. Eur. J. Heart Fail. 10, 1149–1151 (2008).
Hilfiker-Kleiner, D. et al. Recovery from postpartum cardiomyopathy in 2 patients by blocking prolactin release with bromocriptine. J. Am. Coll. Cardiol. 50, 2354–2355 (2007).
Sliwa, K. et al. Evaluation of bromocriptine in the treatment of acute severe peripartum cardiomyopathy: a proof-of-concept pilot study. Circulation 121, 1465–1473 (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
Bello, N., Rendon, I. S. & Arany, Z. The relationship between pre-eclampsia and peripartum cardiomyopathy: a systematic review and meta-analysis. J. Am. Coll. Cardiol. 62, 1715–1723 (2013).
Rana, S. et al. Angiogenic factors and the risk of adverse outcomes in women with suspected preeclampsia. Circulation 125, 911–919 (2012).
Shahul, S. et al. Subclinical left ventricular dysfunction in preeclamptic women with preserved left ventricular ejection fraction: a 2D speckle-tracking imaging study. Circ. Cardiovasc. Imaging 5, 734–739 (2012).
Powe, C. E., Levine, R. J. & Karumanchi, S. A. Preeclampsia, a disease of the maternal endothelium: the role of antiangiogenic factors and implications for later cardiovascular disease. Circulation 123, 2856–2869 (2011).
Fett, J. D. Earlier detection can help avoid many serious complications of peripartum cardiomyopathy. Future Cardiol. 9, 809–816 (2013).
Elkayam, U. et al. Maternal and fetal outcomes of subsequent pregnancies in women with peripartum cardiomyopathy. N. Engl. J. Med. 344, 1567–1571 (2001).
Chung, E., Yeung, F. & Leinwand, L. A. Akt and MAPK signaling mediate pregnancy-induced cardiac adaptation. J. Appl. Physiol. (1985) 112, 1564–1575 (2012).
Eghbali, M. et al. Molecular and functional signature of heart hypertrophy during pregnancy. Circ. Res. 96, 1208–1216 (2005).
Walenta, K. et al. Circulating microparticles as indicators of peripartum cardiomyopathy. Eur. Heart J. 33, 1469–1479 (2012).
Acknowledgements
We thank Sylvia Dennis (Hatter Institute for Cardiovascular Research in Africa, University of Cape Town, South Africa) for proofreading the manuscript. The authors are supported by the Deutsche Forschungs Gesellschaft (DFG), the Bundesministeriums für Bildung und Forschung (BMBF), the National Research Foundation South Africa, and the Medical Research Foundation South Africa.
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, contributed substantially to discussion of its content, wrote the manuscript, and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Hilfiker-Kleiner, D., Sliwa, K. Pathophysiology and epidemiology of peripartum cardiomyopathy. Nat Rev Cardiol 11, 364–370 (2014). https://doi.org/10.1038/nrcardio.2014.37
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2014.37
- Springer Nature Limited
This article is cited by
-
Imaging of heart disease in women: review and case presentation
European Journal of Nuclear Medicine and Molecular Imaging (2022)
-
Peripartum cardiomyopathy: a review
Heart Failure Reviews (2021)
-
Left ventricular function recovery in peripartum cardiomyopathy: a cardiovascular magnetic resonance study by myocardial T1 and T2 mapping
Journal of Cardiovascular Magnetic Resonance (2020)
-
Vasoinhibin reduces joint inflammation, bone loss, and the angiogenesis and vasopermeability of the pannus in murine antigen-induced arthritis
Laboratory Investigation (2020)
-
Understanding Health Disparities in Cardiovascular Diseases in Pregnancy Among Black Women: Prevalence, Preventive Care, and Peripartum Support Networks
Current Cardiovascular Risk Reports (2020)