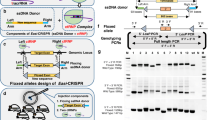
Abstract
CRISPR/Cas9-based genome editing can easily generate knockout mouse models by disrupting the gene sequence, but its efficiency for creating models that require either insertion of exogenous DNA (knock-in) or replacement of genomic segments is very poor. The majority of mouse models used in research involve knock-in (reporters or recombinases) or gene replacement (e.g., conditional knockout alleles containing exons flanked by LoxP sites). A few methods for creating such models have been reported that use double-stranded DNA as donors, but their efficiency is typically 1–10% and therefore not suitable for routine use. We recently demonstrated that long single-stranded DNAs (ssDNAs) serve as very efficient donors, both for insertion and for gene replacement. We call this method efficient additions with ssDNA inserts–CRISPR (Easi-CRISPR) because it is a highly efficient technology (efficiency is typically 30–60% and reaches as high as 100% in some cases). The protocol takes ∼2 months to generate the founder mice.







Similar content being viewed by others
References
Gurumurthy, C.B. et al. Validation of simple sequence length polymorphism regions of commonly used mouse strains for marker assisted speed congenics screening. Int. J. Genomics 2015, 735845 (2015).
Gurumurthy, C.B. et al. CRISPR: a versatile tool for both forward and reverse genetics research. Hum. Genet. 135, 971–976 (2016).
Quadros, R.M., Harms, D.W., Ohtsuka, M. & Gurumurthy, C.B. Insertion of sequences at the original provirus integration site of mouse ROSA26 locus using the CRISPR/Cas9 system. FEBS Open Bio 5, 191–197 (2015).
Inui, M. et al. Rapid generation of mouse models with defined point mutations by the CRISPR/Cas9 system. Sci. Rep. 4, 5396 (2014).
Horii, T. & Hatada, I. Challenges to increasing targeting efficiency in genome engineering. J. Reprod. Dev. 62, 7–9 (2016).
Yang, H. et al. One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154, 1370–1379 (2013).
Ma, Y. et al. Generating rats with conditional alleles using CRISPR/Cas9. Cell Res. 24, 122–125 (2014).
Miura, H., Gurumurthy, C.B., Sato, T., Sato, M. & Ohtsuka, M. CRISPR/Cas9-based generation of knockdown mice by intronic insertion of artificial microRNA using longer single-stranded DNA. Sci. Rep. 5, 12799 (2015).
Quadros, R.M. et al. Easi-CRISPR: a robust method for one-step generation of mice carrying conditional and insertion alleles using long ssDNA donors and CRISPR ribonucleoproteins. Genome Biol. 18, 92 (2017).
George, M. et al. Renal thrombotic microangiopathy in mice with combined deletion of endocytic recycling regulators EHD3 and EHD4. PLoS One 6, e17838 (2011).
Jaisser, F. Inducible gene expression and gene modification in transgenic mice. J. Am. Soc. Nephrol. 11, S95–S100 (2000).
Ohtsuka, M. et al. Fluorescent transgenic mice suitable for multi-color aggregation chimera studies. Cell Tissue Res. 350, 251–260 (2012).
Olou, A.A. et al. Mammalian ECD protein is a novel negative regulator of the PERK arm of the unfolded protein response. Mol. Cell Biol. 37, e00030–e00017 (2017).
Suda, T., Gao, X., Stolz, D.B. & Liu, D. Structural impact of hydrodynamic injection on mouse liver. Gene Ther. 14, 129–137 (2006).
Takahashi, G., Gurumurthy, C.B., Wada, K., Miura, H., Sato, M. & Ohtsuka, M. GONAD: genome-editing via Oviductal Nucleic Acids Delivery system: a novel microinjection independent genome engineering method in mice. Sci. Rep. 5, 11406 (2015).
Ohtsuka, M. et al. In situ genome editing method suitable for routine generation of germline modified animal models. Preprint at bioRxiv http://dx.doi.org/10.1101/172718. (2017).
Yu, Z. et al. Highly efficient genome modifications mediated by CRISPR/Cas9 in Drosophila. Genetics 195, 289–291 (2013).
Hwang, W.Y. et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 31, 227–229 (2013).
Li, D. et al. Heritable gene targeting in the mouse and rat using a CRISPR-Cas system. Nat. Biotechnol. 31, 681–683 (2013).
Yang, D. et al. Effective gene targeting in rabbits using RNA-guided Cas9 nucleases. J. Mol. Cell Biol. 6, 97–99 (2014).
Gao, Y. et al. Single Cas9 nickase induced generation of NRAMP1 knockin cattle with reduced off-target effects. Genome Biol. 18, 13 (2017).
Hongbing, H. et al. One-step generation of myostatin gene knockout sheep via the CRISPR/Cas9 system. Front. Agric. Sci. Eng. 1, 2 (2014).
Ni, W. et al. Efficient gene knockout in goats using CRISPR/Cas9 system. PLoS One 9, e106718 (2014).
Hai, T., Teng, F., Guo, R., Li, W. & Zhou, Q. One-step generation of knockout pigs by zygote injection of CRISPR/Cas system. Cell Res. 24, 372–375 (2014).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Skarnes, W.C. Is mouse embryonic stem cell technology obsolete? Genome Biol. 16, 109 (2015).
Cohen, J. 'Any idiot can do it.' Genome editor CRISPR could put mutant mice in everyone's reach. Science 354, 538–542 (2016).
Maruyama, T. et al. Increasing the efficiency of precise genome editing with CRISPR-Cas9 by inhibition of nonhomologous end joining. Nat. Biotechnol. 33, 538–542 (2015).
Nakao, H. et al. A possible aid in targeted insertion of large DNA elements by CRISPR/Cas in mouse zygotes. Genesis 54, 65–77 (2016).
Wang, B. et al. Highly efficient CRISPR/HDR-mediated knock-in for mouse embryonic stem cells and zygotes. BioTechniques 59, 201–202 (2015).
Aida, T. et al. Gene cassette knock-in in mammalian cells and zygotes by enhanced MMEJ. BMC Genomics 17, 979 (2016).
Aida, T. et al. Cloning-free CRISPR/Cas system facilitates functional cassette knock-in in mice. Genome Biol. 16, 87 (2015).
Bishop, K.A. et al. CRISPR/Cas9-mediated insertion of loxP sites in the mouse Dock7 gene provides an effective alternative to use of targeted embryonic stem cells. G3 (Bethesda) 6, 2051–2061 (2016).
Lee, A.Y. & Lloyd, K.C.K. Conditional targeting of Ispd using paired Cas9 nickase and a single DNA template in mice. FEBS Open Bio 4, 637–642 (2014).
Ellefson, J.W. Synthetic evolutionary origin of a proofreading reverse transcriptase. Science 352, 1590–1593 (2016).
Harms, D.W. et al. Mouse genome editing using the CRISPR/Cas system. Curr. Protoc. Hum. Genet. 83, 15.7.1–15.7.27 (2014).
Jacobi, A.M et al. Simplified CRISPR tools for efficient genome editing and streamlined protocols for their delivery into mammalian cells and mouse zygotes. Methods 121–122, 16–28 (2017).
Yoshimi, K. et al. ssODN-mediated knock-in with CRISPR-Cas for large genomic regions in zygotes. Nat. Commun. 7, 10431 (2016).
Schilit, S.L.P., Ohtsuka, M., Quadros, R.M. & Gurumurthy, C.B. Pronuclear injection-based targeted transgenesis. Curr. Protoc. Hum. Genet. 15.10.1–15.10.28 2016.
Behringer, R., Gertsenstein, M., Nagy, K.V. & Nagy, A. Manipulating the mouse embryo: a laboratory manual 4th edn. Cold Spring Harbor Laboratory Press (2014).
Hendel, A. et al. Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat. Biotechnol. 33, 985–989 (2015).
Raveux, A., Vandormael-Pournin, S. & Cohen-Tannoudji, M. Optimization of the production of knock-in alleles by CRISPR/Cas9 microinjection into the mouse zygote. Sci. Rep. 7, 42661 (2017).
Chu, V.T. et al. Efficient generation of Rosa26 knock-in mice using CRISPR/Cas9 in C57BL/6 zygotes. BMC Biotechnol. 16, 4 (2016).
Ménoret, S. et al. Homology-directed repair in rodent zygotes using Cas9 and TALEN engineered proteins. Sci. Rep. 5, 14410 (2015).
Wang, L. et al. Large genomic fragment deletion and functional gene cassette knock-in via Cas9 protein mediated genome editing in one-cell rodent embryos. Sci. Rep. 5, 17517 (2015).
Zhang, L. et al. Large genomic fragment deletions and insertions in mouse using CRISPR/Cas9. PLoS One 10, e0120396 (2015).
Acknowledgements
This work was supported in part by a Grant-in-Aid for Young Scientists (B) (16K18821) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) to H.M.; and by 2014 Tokai University School of Medicine Research Aid, the MEXT-Supported Program for the Strategic Research Foundation at Private Universities 2015–2019 to Tokai University, the Research and Study Project of Tokai University General Research Organization, a Grant-in-Aid for Scientific Research (16H04685) from the MEXT, and funding from 2016–2017 Tokai University School of Medicine Project Research to M.O.; and by an Institutional Development Award (principal investigator: S. Smith) P20GM103471 (to C.B.G. and R.M.Q.). We thank A. Koesters, University of Nebraska Medical Center, for her editorial contribution and J.M. Miano, University of Rochester, for his helpful comments on the manuscript. We also gratefully acknowledge the contribution of the staff of the Support Center for Medical Research and Education, Tokai University, for sequencing and microinjection.
Author information
Authors and Affiliations
Contributions
M.O. conceived the idea of using long ssDNAs as donors in genome editing, which was further tested and improved upon by the other three authors. All authors contributed equally in writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
C.B.G., M.O., and H.M. have filed a patent application relating to the work described in this paper with international application number PCT/US2016/035660, filed June 3, 2016 (DNA editing using single-stranded DNA).
Integrated supplementary information
Supplementary Figure 1 Comparison of RNA yield between kits.
1μg DNA was used for RNA synthesis according to each manufacturer's protocol. The RNA samples in lanes 1 and 2 were synthesized using kits from two different vendors. 1% of the eluates, from purification using the MEGAclear columns, were loaded. This figure illustrates that kits can vary in their efficiencies. It should be noted that the identity of the products is not an important point: we have observed a little better performance of a different batch from vendor 1, and somewhat poorer performance of another batch of kit from vendor 2.
Supplementary Figure 2 Comparison of cDNA yield among different kits.
cDNAs (ssDNA) were synthesized using reverse transcriptases from three different vendors (1 to 3). 1.7μg RNA was used for cDNA synthesis according to each manufactures protocol and 50% of the reaction volumes were loaded. The cDNA yield from all three transcriptases are comparable.
Supplementary Figure 3 Comparison of cDNA synthesis parameters.
1.7μg (lane 1 and 2) or 5μg (lanes 3 and 4) of RNA was used for cDNA synthesis and the reactions were incubated for 10 minutes (lanes 1 and 3) or 2 hours (lanes 2 and 4). 15% of the reaction volume was loaded. The band intensities suggest that 5μg RNA input produces optimal yields of cDNA.
Supplementary Figure 4 Gel extraction of bands from cDNA preparations.
(a) Typical smear-like appearance of the cDNA (ssDNA) preparations in an agarose gel showing a prominent band (box # 1) and a less prominent band (box # 2). (b and c) Gel slices excised for DNA extraction. (d) The purified ssDNAs loaded in another gel showing both bands migrate similarly. Either of the gel preparations (1 or 2) can be used for microinjection, or both preparations can be pooled before use.
Supplementary Figure 5 Verification of ssDNA using S1 nuclease.
About 150 ng of ~0.5kb cDNA was incubated with or without S1 nuclease at 37°C for 15 minutes.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1. (PDF 492 kb)
Rights and permissions
About this article
Cite this article
Miura, H., Quadros, R., Gurumurthy, C. et al. Easi-CRISPR for creating knock-in and conditional knockout mouse models using long ssDNA donors. Nat Protoc 13, 195–215 (2018). https://doi.org/10.1038/nprot.2017.153
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2017.153
- Springer Nature Limited
This article is cited by
-
A beneficial adaptive role for CHOP in driving cell fate selection during ER stress
EMBO Reports (2024)
-
Maternal RNA transcription in Dlk1-Dio3 domain is critical for proper development of the mouse placental vasculature
Communications Biology (2024)
-
An Update on the Application of CRISPR Technology in Clinical Practice
Molecular Biotechnology (2024)
-
Bridging biological samples to functional nucleic acid biosensor applications: current enzymatic-based strategies for single-stranded DNA generation
Analytical Sciences (2024)
-
Muc4 loss mitigates epidermal growth factor receptor activity essential for PDAC tumorigenesis
Oncogene (2023)