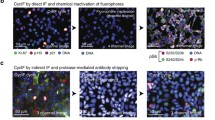
Abstract
Here we present a detailed protocol for molecular profiling of individual cultured mammalian cells using multicolor multicycle immunofluorescence with quantum dot probes. It includes instructions for cell culture growth and processing (2 h + 48–72 h for cell growth), preparation and characterization of universal quantum dot probes (4.5 h + overnight incubation), cyclic cell staining (∼4.5 h per cycle) and image analysis (varies by application). The use of quantum dot fluorescent probes enables highly multiplexed, robust quantitative molecular imaging with a conventional fluorescence microscopy setup, whereas the probe preparation methodology, using a self-assembly between protein A–decorated universal quantum dots and intact primary antibodies, offers a fast, simple and purification-free route for an on-demand preparation of antibody-functionalized quantum dot libraries. As a result, this protocol can be used by biomedical researchers for a variety of cell staining applications, and, with further optimization, for staining of other biological specimens (e.g., clinical tissue sections).





Similar content being viewed by others
References
de Souza, N. Single-cell methods. Nat. Methods 9, 35 (2012).
Chaurand, P., Sanders, M.E., Jensen, R.A. & Caprioli, R.M. Proteomics in diagnostic pathology: profiling and imaging proteins directly in tissue sections. Am. J. Pathol. 165, 1057–1068 (2004).
Liotta, L. & Petricoin, E. Molecular profiling of human cancer. Nat. Rev. Genet. 1, 48–56 (2000).
Gorg, A., Weiss, W. & Dunn, M.J. Current two-dimensional electrophoresis technology for proteomics. Proteomics 4, 3665–3685 (2004).
Aebersold, R. & Mann, M. Mass spectrometry-based proteomics. Nature 422, 198–207 (2003).
Schwamborn, K. & Caprioli, R.M. Molecular imaging by mass spectrometry–looking beyond classical histology. Nat. Rev. Cancer 10, 639–646 (2010).
Wollscheid, B. et al. Mass-spectrometric identification and relative quantification of N-linked cell surface glycoproteins. Nat. Biotechnol. 27, 378–386 (2009).
Caldwell, R.L. & Caprioli, R.M. Tissue profiling by mass spectrometry–A review of methodology and applications. Mol. Cell. Proteomics 4, 394–401 (2005).
Chattopadhyay, P.K. et al. Quantum dot semiconductor nanocrystals for immunophenotyping by polychromatic flow cytometry. Nat. Med. 12, 972–977 (2006).
Bendall, S.C. et al. Single-cell mass cytometry of differential immune and drug responses across a human hematopoietic continuum. Science 332, 687–696 (2011).
Ma, C. et al. A clinical microchip for evaluation of single immune cells reveals high functional heterogeneity in phenotypically similar T cells. Nat. Med. 17, 738–743 (2011).
Salehi-Reyhani, A. et al. A first step towards practical single cell proteomics: a microfluidic antibody capture chip with TIRF detection. Lab Chip 11, 1256–1261 (2011).
Umemura, S. & Osamura, R.Y. Utility of immunohistochemistry in breast cancer practice. Breast Cancer 11, 334–338 (2004).
True, L.D. Quantitative immunohistochemistry: a new tool for surgical pathology? Am. J. Clin. Pathol. 90, 324–325 (1988).
Micheva, K.D., Busse, B., Weiler, N.C., O'Rourke, N. & Smith, S.J. Single-synapse analysis of a diverse synapse population: proteomic imaging methods and markers. Neuron 68, 639–653 (2010).
Schubert, W. et al. Analyzing proteome topology and function by automated multidimensional fluorescence microscopy. Nat. Biotechnol. 24, 1270–1278 (2006).
Medintz, I.L. & Mattoussi, H. Quantum dot-based resonance energy transfer and its growing application in biology. Phys. Chem. Chem. Phys. 11, 17–45 (2009).
Medintz, I.L., Mattoussi, H. & Clapp, A.R. Potential clinical applications of quantum dots. Int. J. Nanomedicine 3, 151–167 (2008).
Misra, R.D. Quantum dots for tumor-targeted drug delivery and cell imaging. Nanomedicine 3, 271–274 (2008).
Resch-Genger, U., Grabolle, M., Cavaliere-Jaricot, S., Nitschke, R. & Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 5, 763–775 (2008).
Smith, A.M., Duan, H., Mohs, A.M. & Nie, S. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv. Drug Deliv. Rev. 60, 1226–1240 (2008).
Tholouli, E. et al. Quantum dots light up pathology. J. Pathol. 216, 275–285 (2008).
Xing, Y. & Rao, J. Quantum dot bioconjugates for in vitro diagnostics and in vivo imaging. Cancer Biomark. 4, 307–319 (2008).
Zrazhevskiy, P. & Gao, X. Multifunctional quantum dots for personalized medicine. Nano Today 4, 414–428 (2009).
Zrazhevskiy, P., Sena, M. & Gao, X.H. Designing multifunctional quantum dots for bioimaging, detection, and drug delivery. Chem. Soc. Rev. 39, 4326–4354 (2010).
Yezhelyev, M.V. et al. In situ molecular profiling of breast cancer biomarkers with multicolor quantum dots. Adv. Mater. 19, 3146–3151 (2007).
Matsuno, A., Itoh, J., Takekoshi, S., Nagashima, T. & Osamura, R.Y. Three-dimensional imaging of the intracellular localization of growth hormone and prolactin and their mRNA using nanocrystal (Quantum dot) and confocal laser scanning microscopy techniques. J. Histochem. Cytochem. 53, 833–838 (2005).
Shi, C. et al. Quantum dots-based multiplexed immunohistochemistry of protein expression in human prostate cancer cells. Eur. J. Histochem. 52, 127–134 (2008).
Liu, J., Lau, S.K., Varma, V.A., Kairdolf, B.A. & Nie, S. Multiplexed detection and characterization of rare tumor cells in Hodgkin's lymphoma with multicolor quantum dots. Anal. Chem. 82, 6237–6243 (2010).
Liu, J. et al. Molecular mapping of tumor heterogeneity on clinical tissue specimens with multiplexed quantum dots. ACS Nano 4, 2755–2765 (2010).
Chen, C. et al. Quantum-dot–based immunofluorescent imaging of HER2 and ER provides new insights into breast cancer heterogeneity. Nanotechnology 21, 095101 (2010).
Sweeney, E. et al. Quantitative multiplexed quantum dot immunohistochemistry. Biochem. Biophys. Res. Commun. 374, 181–186 (2008).
Fountaine, T.J., Wincovitch, S.M., Geho, D.H., Garfield, S.H. & Pittaluga, S. Multispectral imaging of clinically relevant cellular targets in tonsil and lymphoid tissue using semiconductor quantum dots. Mod. Pathol. 19, 1181–1191 (2006).
Huang, D.H. et al. Comparison and optimization of multiplexed quantum dot–based immunohistofluorescence. Nano Res. 3, 61–68 (2010).
Xing, Y. et al. Bioconjugated quantum dots for multiplexed and quantitative immunohistochemistry. Nat. Protoc. 2, 1152–1165 (2007).
Zrazhevskiy, P. & Gao, X. Quantum dot imaging platform for single-cell molecular profiling. Nat. Commun. 4, 1619 (2013).
Denysenko, T. et al. Glioblastoma cancer stem cells: heterogeneity, microenvironment and related therapeutic strategies. Cell Biochem. Funct. 28, 343–351 (2010).
Sachs, K., Perez, O., Pe'er, D., Lauffenburger, D.A. & Nolan, G.P. Causal protein-signaling networks derived from multiparameter single-cell data. Science 308, 523–529 (2005).
Ciftlik, A.T., Lehr, H.-A. & Gijs, M.A.M. Microfluidic processor allows rapid HER2 immunohistochemistry of breast carcinomas and significantly reduces ambiguous (2+) read-outs. Proc. Natl. Acad. Sci. USA 110, 5363–5368 (2013).
Chang, K.H. et al. Novel 16-minute technique for evaluating melanoma resection margins during Mohs surgery. J. Am. Acad. Dermatol. 64, 107–112 (2011).
Goldman, E.R. et al. Avidin: a natural bridge for quantum dot-antibody conjugates. J. Am. Chem. Soc. 124, 6378–6382 (2002).
Jaiswal, J.K., Mattoussi, H., Mauro, J.M. & Simon, S.M. Long-term multiple color imaging of live cells using quantum dot bioconjugates. Nat. Biotechnol. 21, 47–51 (2003).
Jin, T. et al. Antibody-protein A conjugated quantum dots for multiplexed imaging of surface receptors in living cells. Mol. BioSyst. 6, 2325–2331 (2010).
Mattoussi, H. et al. Self-assembly of CdSe-ZnS quantum dot bioconjugates using an engineered recombinant protein. J. Am. Chem. Soc. 122, 12142–12150 (2000).
Levenson, R.M. Spectral imaging and pathology: seeing more. Lab Med. 35, 244–251 (2004).
True, L.D. & Gao, X. Quantum dots for molecular pathology: their time has arrived. J. Mol. Diagn. 9, 7–11 (2007).
Byers, R.J. et al. Semiautomated multiplexed quantum dot-based in situ hybridization and spectral deconvolution. J. Mol. Diagnostics 9, 20–29 (2007).
Ghazani, A.A. et al. High-throughput quantification of protein expression of cancer antigens in tissue microarray using quantum dot nanocrystals. Nano Lett. 6, 2881–2886 (2006).
Toth, Z.E. & Mezey, E. Simultaneous visualization of multiple antigens with tyramide signal amplification using antibodies from the same species. J. Histochem. Cytochem. 55, 545–554 (2007).
Glass, G., Papin, J.A. & Mandell, J.W. SIMPLE: a sequential immunoperoxidase labeling and erasing method. J. Histochem. Cytochem. 57, 899–905 (2009).
Pirici, D. et al. Antibody elution method for multiple immunohistochemistry on primary antibodies raised in the same species and of the same subtype. J. Histochem. Cytochem. 57, 567–575 (2009).
Wahlby, C., Erlandsson, F., Bengtsson, E. & Zetterberg, A. Sequential immunofluorescence staining and image analysis for detection of large numbers of antigens in individual cell nuclei. Cytometry 47, 32–41 (2002).
Acknowledgements
This work was supported in part by the US National Institutes of Health (R01CA131797 to X.G.; P50CA097186 to L.D.T.), the US National Science Foundation (NSF) (0645080), US Department of Defense–Congressionally Directed Medical Research Programs (DoD-CDMRP) (W81XWH0710117), the Coulter foundation and the Department of Bioengineering at the University of Washington. X.G. thanks the NSF for a Faculty Early Career Development award (CAREER). P.Z. thanks the University of Washington Center for Nanotechnology for a University Initiatives Fund Fellowship, the NSF for a Graduate Research Fellowship (DGE-0718124) and the National Cancer Institute for a T32 Fellowship (T32CA138312). We are also grateful to R. Vessella and P. Nelson for fruitful discussions on clinical diagnostics and molecular pathology research and to J. Li, C. Probst and J. Shang for valuable comments on the manuscript.
Author information
Authors and Affiliations
Contributions
P.Z., L.D.T. and X.G. contributed to the experiment design and data analysis. P.Z. performed the experiments. P.Z. and X.G. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
Examination of expanded molecular target sets through multicolor multicycle staining (PDF 266 kb)
Supplementary Figure 2
Achieving QDot probe intra-nuclear access with Proteinase K digestion (PDF 305 kb)
Supplementary Figure 3
Hyperspectral imaging of cells labeled by multicolor QDot probes (PDF 317 kb)
Supplementary Figure 4
Building a reference QDot spectral library with HSI (PDF 311 kb)
Rights and permissions
About this article
Cite this article
Zrazhevskiy, P., True, L. & Gao, X. Multicolor multicycle molecular profiling with quantum dots for single-cell analysis. Nat Protoc 8, 1852–1869 (2013). https://doi.org/10.1038/nprot.2013.112
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.112
- Springer Nature Limited
This article is cited by
-
Oligonucleotide conjugated antibody strategies for cyclic immunostaining
Scientific Reports (2021)
-
Quantitative microimmunohistochemistry for the grading of immunostains on tumour tissues
Nature Biomedical Engineering (2019)
-
Multiplexed in-gel microfluidic immunoassays: characterizing protein target loss during reprobing of benzophenone-modified hydrogels
Scientific Reports (2019)
-
Simultaneous detection of multiple viruses in their co-infected cells using multicolour imaging with self-assembled quantum dot probes
Microchimica Acta (2017)
-
Direct metabolomics for plant cells by live single-cell mass spectrometry
Nature Protocols (2015)