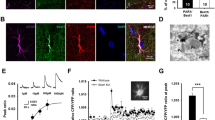
Abstract
The release of transmitters from glia influences synaptic functions. The modalities and physiological functions of glial release are poorly understood. Here we show that glutamate exocytosis from astrocytes of the rat hippocampal dentate molecular layer enhances synaptic strength at excitatory synapses between perforant path afferents and granule cells. The effect is mediated by ifenprodil-sensitive NMDA ionotropic glutamate receptors and involves an increase of transmitter release at the synapse. Correspondingly, we identify NMDA receptor 2B subunits on the extrasynaptic portion of excitatory nerve terminals. The receptor distribution is spatially related to glutamate-containing synaptic-like microvesicles in the apposed astrocytic processes. This glial regulatory pathway is endogenously activated by neuronal activity–dependent stimulation of purinergic P2Y1 receptors on the astrocytes. Thus, we provide the first combined functional and ultrastructural evidence for a physiological control of synaptic activity via exocytosis of glutamate from astrocytes.






Similar content being viewed by others
References
Parpura, V. et al. Glutamate-mediated astrocyte-neuron signalling. Nature 369, 744–747 (1994).
Bezzi, P. et al. Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 391, 281–285 (1998).
Araque, A., Li, N., Doyle, R.T. & Haydon, P.G. SNARE protein–dependent glutamate release from astrocytes. J. Neurosci. 20, 666–673 (2000).
Bezzi, P. et al. CXCR4-activated astrocyte glutamate release via TNFα: amplification by microglia triggers neurotoxicity. Nat. Neurosci. 4, 702–710 (2001).
Pasti, L., Zonta, M., Pozzan, T., Vicini, S. & Carmignoto, G. Cytosolic calcium oscillations in astrocytes may regulate exocytotic release of glutamate. J. Neurosci. 21, 477–484 (2001).
Bezzi, P. et al. Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat. Neurosci. 7, 613–620 (2004).
Domercq, M. et al. P2Y1 receptor–evoked glutamate exocytosis from astrocytes: control by TNF alpha and prostaglandins. J. Biol. Chem. 281, 30684–30696 (2006).
Crippa, D. et al. Synaptobrevin2-expressing vesicles in rat astrocytes: insights into molecular characterization, dynamics and exocytosis. J. Physiol. (Lond.) 570, 567–582 (2006).
Zhang, Q. et al. Fusion-related release of glutamate from astrocytes. J. Biol. Chem. 279, 12724–12733 (2004).
Volterra, A. & Meldolesi, J. Astrocytes, from brain glue to communication elements: the revolution continues. Nat. Rev. Neurosci. 6, 626–640 (2005).
Pasti, L., Volterra, A., Pozzan, T. & Carmignoto, G. Intracellular calcium oscillations in astrocytes: a highly plastic, bidirectional form of communication between neurons and astrocytes in situ. J. Neurosci. 17, 7817–7830 (1997).
Bowser, D.N. & Khakh, B.S. ATP excites interneurons and astrocytes to increase synaptic inhibition in neuronal networks. J. Neurosci. 24, 8606–8620 (2004).
Steinhauser, C., Berger, T., Frotscher, M. & Kettenmann, H. Heterogeneity in the membrane current pattern of identified glial cells in the hippocampal slice. Eur. J. Neurosci. 4, 472–484 (1992).
Shimamoto, K. et al. DL-threo-β-benzyloxyaspartate, a potent blocker of excitatory amino acid transporters. Mol. Pharmacol. 53, 195–201 (1998).
Kang, J., Jiang, L., Goldman, S.A. & Nedergaard, M. Astrocyte-mediated potentiation of inhibitory synaptic transmission. Nat. Neurosci. 1, 683–692 (1998).
Schiavo, G. et al. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 359, 832–835 (1992).
Rossetto, O. et al. Active-site mutagenesis of tetanus neurotoxin implicates Tyr-375 and Glu-271 in metalloproteolytic activity. Toxicon 39, 1151–1159 (2001).
Fellin, T. et al. Neuronal synchrony mediated by astrocytic glutamate through activation of extrasynaptic NMDA receptors. Neuron 43, 729–743 (2004).
Fellin, T., Gomez-Gonzalo, M., Gobbo, S., Carmignoto, G. & Haydon, P.G. Astrocytic glutamate is not necessary for the generation of epileptiform neuronal activity in hippocampal slices. J. Neurosci. 26, 9312–9322 (2006).
Morán-Jiménez, M.J. & Matute, C. Immunohistochemical localization of the P2Y1 purinergic receptor in neurons and glial cells of the central nervous system. Brain Res. Mol. Brain Res. 78, 50–58 (2000).
Nimmerjahn, A., Kirchhoff, F., Kerr, J.N.D. & Helmchen, F. Sulforhodamine 101 as a specific marker of astroglia in the neocortex in vivo. Nat. Methods 1, 31–37 (2004).
Boyer, J.L., Romero-Avila, T., Schachter, J.B. & Harden, T.K. Identification of competitive antagonists of the P2Y1 receptor. Mol. Pharmacol. 50, 1323–1329 (1996).
Von Kügelgen, I. & Wetter, A. Molecular pharmacology of P2Y-receptors. Naunyn Schmiedebergs Arch. Pharmacol. 362, 310–323 (2000).
Serrano, A., Haddjeri, N., Lacaille, J.C. & Robitaille, R. GABAergic network activation of glial cells underlies hippocampal heterosynaptic depression. J. Neurosci. 26, 5370–5382 (2006).
Jahn, R., Lang, T. & Sudhof, T.C. Membrane fusion. Cell 112, 519–533 (2003).
Zhang, Q., Fukuda, M., Van Bockstaele, E., Pascual, O. & Haydon, P.G. Synaptotagmin IV regulates glial glutamate release. Proc. Natl. Acad. Sci. USA 101, 9441–9446 (2004).
Panatier, A. et al. Glia-derived D-serine controls NMDA receptor activity and synaptic memory. Cell 125, 775–784 (2006).
Dalby, N.O. & Mody, I. Activation of NMDA receptors in rat dentate gyrus granule cells by spontaneous and evoked transmitter release. J. Neurophysiol. 90, 786–797 (2003).
Cull-Candy, S.G. & Leszkiewicz, D.N. Role of distinct NMDA receptor subtypes at central synapses. Sci. STKE 255, re16 (2004).
Karadottir, R., Cavelier, P., Bergersen, L.H. & Attwell, D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438, 1162–1166 (2005).
Parri, H.R., Gould, T.M. & Crunelli, V. Spontaneous astrocytic Ca2+ oscillations in situ drive NMDAR-mediated neuronal excitation. Nat. Neurosci. 4, 803–812 (2001).
Duguid, I.C. & Smart, T.G. Retrograde activation of presynaptic NMDA receptors enhances GABA release at cerebellar interneuron-Purkinje cell synapses. Nat. Neurosci. 7, 525–533 (2004).
Gitler, D. et al. Different presynaptic roles of synapsins at excitatory and inhibitory synapses. J. Neurosci. 24, 11368–11380 (2004).
Matsui, K. & Jahr, C.E. Ectopic release of synaptic vesicles. Neuron 40, 1173–1183 (2003).
Rusakov, D.A. & Lehre, K.P. Perisynaptic asymmetry of glia: new insights into glutamate signalling. Trends Neurosci. 25, 492–494 (2002).
Mori, M., Heuss, C., Gäwiler, B.H. & Gerber, U. Fast synaptic transmission mediated by P2X receptors in CA3 pyramidal cells of rat hippocampal slice cultures. J. Physiol. (Lond.) 535, 115–123 (2001).
Zhang, J.M. et al. ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression. Neuron 40, 971–982 (2003).
Mitchell, J.B., Lupica, C.R. & Dunwiddie, T.V. Activity-dependent release of endogenous adenosine modulates synaptic responses in the rat hippocampus. J. Neurosci. 13, 3439–3447 (1993).
Dunwiddie, T.V., Diao, L. & Proctor, W.R. Adenine nucleotides undergo rapid, quantitative conversion to adenosine in the extracellular space in rat hippocampus. J. Neurosci. 17, 7673–7682 (1997).
Pascual, O. et al. Astrocytic purinergic signaling coordinates synaptic networks. Science 310, 113–116 (2005).
Araque, A., Parpura, V., Sanzgiri, R.P. & Haydon, P.G. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 22, 208–215 (1999).
Jones, R.S.G. Entorhinal-hippocampal connections: a speculative view of their function. Trends Neurosci. 16, 58–64 (1993).
Gomez-Isla, T. et al. Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer's disease. J. Neurosci. 16, 4491–4500 (1996).
Rossi, D. et al. Defective tumor necrosis factor alpha–dependent control of astrocyte glutamate release in a transgenic mouse model of Alzheimer disease. J. Biol. Chem. 280, 42088–42096 (2005).
Rutecki, P.A., Grossman, R.G., Armstrong, D. & Irish-Loewen, S. Electrophysiological connections between the hippocampus and entorhinal cortex in patients with complex partial seizures. J. Neurosurg. 70, 667–675 (1989).
Staley, K.J. & Mody, I. Integrity of perforant path fibers and the frequency of action potential independent excitatory and inhibitory synaptic events in dentate gyrus granule cells. Synapse 9, 219–224 (1991).
Bushong, E.A., Martone, M.E. & Ellisman, M.H. Examination of the relationship between astrocyte morphology and laminar boundaries in the molecular layer of adult dentate gyrus. J. Comp. Neurol. 462, 241–251 (2003).
Gundersen, V. et al. Synaptic vesicular localization and exocytosis of L-aspartate in excitatory nerve terminals: a quantitative immunogold analysis in rat hippocampus. J. Neurosci. 18, 6059–6070 (1998).
Chaudhry, F.A. et al. Glutamate transporters in glial plasma membranes: highly differentiated localizations revealed by quantitative ultrastructural immunocytochemistry. Neuron 15, 711–720 (1995).
Lehre, K.P. & Danbolt, N.C. The number of glutamate transporter subtype molecules at glutamatergic synapses: chemical and stereological quantification in young adult rat brain. J. Neurosci. 18, 8751–8757 (1998).
Acknowledgements
We thank A. Pietropoli for initial electrophysiology experiments, T. Ivanova for testing the activity of TeNT in astrocytic membrane fractions, D. Attwell and J. Storm-Mathisen for critical comments to the present manuscript and D. Kullmann, D. Muller and S. Oliet for critical comments to previous versions, helpful discussions and suggestions. This work was supported by grants from the Swiss National Science Foundation (3100A0-100850) and Swiss State Secretariat for Education and Research (00.0553) to A.V. and from the Norwegian Research Council to L.H.B. and V.G.
Author information
Authors and Affiliations
Contributions
P.J. conducted most of the electrophysiology experiments and analyses of the electrophysiology data. L.H.B. conducted the immunogold experiments and the analysis of the immunogold data. K.B. conducted the Ca2+ imaging experiments and some of the electrophysiology experiments. P.B. supervised the Ca2+ imaging experiments and the analysis of the Ca2+ imaging data. M.S. conducted part of the analyses of the electrophysiology data. M.D. and C.M. conducted the light and electron microscopy experiments concerning P2Y1R localization. F.T. prepared TeNTLC and TeNTLCE271A. V.G. supervised the immunogold experiments as well as the analysis of the immunogold data and participated in writing the manuscript. A.V. supervised the project and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
The protocol of depolarizing pulses applied to astrocytes does not modify their electrical membrane properties. (PDF 155 kb)
Supplementary Fig. 2
The effect induced by astrocyte stimulation on sEPSC activity in GCs is not reproduced by stimulation of the surrounding neuropil. (PDF 155 kb)
Supplementary Fig. 3
The ifenprodil-sensitive increase in mEPSC frequency in GCs induced by astrocyte stimulation or by direct NMDA application is not accompanied by a change in the amplitude of the miniature events. (PDF 61 kb)
Supplementary Fig. 4
Internal perfusion of MK-801 in GCs effectively blocks GC NMDA receptors, but does not modify the ifenprodil-sensitive NMDA effect on mEPSCs. (PDF 236 kb)
Supplementary Fig. 5
The distribution of NR2B immunogold particles across the extrasynaptic parts of the terminal's membrane where it faces astrocytes suggests that the receptor is inserted in the plasma membrane of the terminal. (PDF 48 kb)
Supplementary Fig. 6
BAPTA diffusion through the astrocytic syncytium. (PDF 106 kb)
Supplementary Table 1
Depolarizing pulses for stimulating astrocytes or neuropil. (PDF 28 kb)
Supplementary Table 2
Astrocyte stimulation induces preferentially mEPSC frequency increase, not SICs, in GCs. (PDF 21 kb)
Supplementary Data
Astrocyte [Ca2+]i elevations in response to depolarizing stimuli: involvement of Ca2+ release from the internal stores and independence on Ca2+ entry. (PDF 25 kb)
Supplementary Note
About the functional connectivity between ML astrocytes and GC synapses. (PDF 22 kb)
Rights and permissions
About this article
Cite this article
Jourdain, P., Bergersen, L., Bhaukaurally, K. et al. Glutamate exocytosis from astrocytes controls synaptic strength. Nat Neurosci 10, 331–339 (2007). https://doi.org/10.1038/nn1849
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1849
- Springer Nature America, Inc.
This article is cited by
-
Astrocytes in the adult dentate gyrus—balance between adult and developmental tasks
Molecular Psychiatry (2024)
-
Timing to be precise? An overview of spike timing-dependent plasticity, brain rhythmicity, and glial cells interplay within neuronal circuits
Molecular Psychiatry (2023)
-
Revisiting the critical roles of reactive astrocytes in neurodegeneration
Molecular Psychiatry (2023)
-
Loss of neuron network coherence induced by virus-infected astrocytes: a model study
Scientific Reports (2023)
-
Cortical astrocytes modulate dominance behavior in male mice by regulating synaptic excitatory and inhibitory balance
Nature Neuroscience (2023)