Abstract
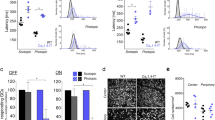
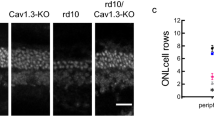
CaBP1–8 are neuronal Ca2+-binding proteins with similarity to calmodulin (CaM). Here we show that CaBP4 is specifically expressed in photoreceptors, where it is localized to synaptic terminals. The outer plexiform layer, which contains the photoreceptor synapses with secondary neurons, was thinner in the Cabp4−/− mice than in control mice. Cabp4−/− retinas also had ectopic synapses originating from rod bipolar and horizontal cells tha HJt extended into the outer nuclear layer. Responses of Cabp4−/− rod bipolars were reduced in sensitivity about 100-fold. Electroretinograms (ERGs) indicated a reduction in cone and rod synaptic function. The phenotype of Cabp4−/− mice shares similarities with that of incomplete congenital stationary night blindness (CSNB2) patients. CaBP4 directly associated with the C-terminal domain of the Cav1.4 α1-subunit and shifted the activation of Cav1.4 to hyperpolarized voltages in transfected cells. These observations indicate that CaBP4 is important for normal synaptic function, probably through regulation of Ca2+ influx and neurotransmitter release in photoreceptor synaptic terminals.







Similar content being viewed by others
References
Zhang, N. & Townes-Anderson, E. Regulation of structural plasticity by different channel types in rod and cone photoreceptors. J. Neurosci. 22, 7065–7079 (2002).
Nachman-Clewner, M., St Jules, R. & Townes-Anderson, E. L-type calcium channels in the photoreceptor ribbon synapse: localization and role in plasticity. J. Comp. Neurol. 415, 1–16 (1999).
Schmitz, F., Konigstorfer, A. & Sudhof, T.C. RIBEYE, a component of synaptic ribbons: a protein's journey through evolution provides insight into synaptic ribbon function. Neuron 28, 857–872 (2000).
Schmitz, Y. & Witkovsky, P. Dependence of photoreceptor glutamate release on a dihydropyridine-sensitive calcium channel. Neuroscience 78, 1209–1216 (1997).
Barnes, S. & Kelly, M.E.M. Calcium channels at the photoreceptor synapse. in. Photorecept. Calcium 514, 465–476 (2002).
Morgans, C.W. Localization of the α1F calcium channel subunit in the rat retina. Invest. Ophthalmol. Vis. Sci. 42, 2414–2418 (2001).
McRory, J.E. et al. The CACNA1F gene encodes an L-type calcium channel with unique biophysical properties and tissue distribution. J. Neurosci. 24, 1707–1718 (2004).
Baumann, L., Gerstner, A., Zong, X., Biel, M. & Wahl-Schott, C. Functional characterization of the L-type Ca2+ channel Cav1.4α1 from mouse retina. Invest. Ophthalmol. Vis. Sci. 45, 708–713 (2004).
Koschak, A. et al. Cav1.4α1 subunits can form slowly inactivating dihydropyridine-sensitive L-type Ca2+ channels lacking Ca2+-dependent inactivation. J. Neurosci. 23, 6041–6049 (2003).
Rieke, F. & Schwartz, E.A. A cGMP-gated current can control exocytosis at cone synapses. Neuron 13, 863–873 (1994).
Bech-Hansen, N.T. et al. Mutations in NYX, encoding the leucine-rich proteoglycan nyctalopin, cause X-linked complete congenital stationary night blindness. Nat. Genet. 26, 319–323 (2000).
Strom, T.M. et al. An L-type calcium-channel gene mutated in incomplete X-linked congenital stationary night blindness. Nat. Genet. 19, 260–263 (1998).
Ball, S.L. et al. Role of the β2 subunit of voltage-dependent calcium channels in the retinal outer plexiform layer. Invest. Ophthalmol. Vis. Sci. 43, 1595–1603 (2002).
Haeseleer, F. et al. Five members of a novel Ca2+-binding protein (CABP) subfamily with similarity to calmodulin. J. Biol. Chem. 275, 1247–1260 (2000).
Lee, A. et al. Differential modulation of Cav2.1 channels by calmodulin and Ca2+-binding protein 1. Nat. Neurosci. 5, 210–217 (2002).
Yang, J. et al. Identification of a family of calcium sensors as protein ligands of inositol trisphosphate receptor Ca2+ release channels. Proc. Natl Acad. Sci. USA 99, 7711–7716 (2002).
Haynes, L.P., Tepikin, A.V. & Burgoyne, R.D. Calcium-binding protein 1 is an inhibitor of agonist-evoked, inositol 1,4,5-trisphosphate-mediated calcium signaling. J. Biol. Chem. 279, 547–555 (2004).
Koulen, P., Fletcher, E.L., Craven, S.E., Bredt, D.S. & Wassle, H. Immunocytochemical localization of the postsynaptic density protein PSD-95 in the mammalian retina. J. Neurosci. 18, 10136–10149 (1998).
Van Hooser, J.P. et al. Rapid restoration of visual pigment and function with oral retinoid in a mouse model of childhood blindness. Proc. Natl Acad. Sci. USA 97, 8623–8628 (2000).
Grunert, U., Haverkamp, S., Fletcher, E.L. & Wassle, H. Synaptic distribution of ionotropic glutamate receptors in the inner plexiform layer of the primate retina. J. Comp. Neurol. 447, 138–151 (2002).
Nomura, A. et al. Developmentally regulated postsynaptic localization of a metabotropic glutamate receptor in rat rod bipolar cells. Cell 77, 361–369 (1994).
Dick, O. et al. The presynaptic active zone protein bassoon is essential for photoreceptor ribbon synapse formation in the retina. Neuron 37, 775–786 (2003).
Baylor, D.A., Lamb, T.D. & Yau, K.W. Responses of retinal rods to single photons. J. Physiol. (Lond.) 288, 613–634 (1979).
Nakatani, K. & Yau, K.W. Calcium and magnesium fluxes across the plasma membrane of the toad rod outer segment. J. Physiol. (Lond.) 395, 695–729 (1988).
Field, G.D. & Rieke, F. Nonlinear signal transfer from mouse rods to bipolar cells and implications for visual sensitivity. Neuron 34, 773–785 (2002).
Slaughter, M.M. & Miller, R.F. 2-amino-4-phosphonobutyric acid: a new pharmacological tool for retina research. Science 211, 182–185 (1981).
Sampath, A.P. & Rieke, F. Selective transmission of single photon responses by saturation at the rod-to-rod bipolar synapse. Neuron 41, 431–443 (2004).
Pitt, G.S. et al. Molecular basis of calmodulin tethering and Ca2+-dependent inactivation of L-type Ca2+ channels. J. Biol. Chem. 276, 30794–30802 (2001).
Erickson, M.G., Alseikhan, B.A., Peterson, B.Z. & Yue, D.T. Preassociation of calmodulin with voltage-gated Ca2+ channels revealed by FRET in single living cells. Neuron 31, 973–985 (2001).
Zuhlke, R.D., Pitt, G.S., Deisseroth, K., Tsien, R.W. & Reuter, H. Calmodulin supports both inactivation and facilitation of L-type calcium channels. Nature 399, 159–162 (1999).
Zhou, H. et al. Ca2+-binding protein-1 facilitates and forms a postsynaptic complex with Cav1.2 (L-type) Ca2+ channels. J. Neurosci. 24, 4698–4708 (2004).
Baylor, D.A. & Fuortes, M.G. Electrical responses of single cones in the retina of the turtle. J. Physiol. (Lond.) 207, 77–92 (1970).
Pate, P. et al. Determinants for calmodulin binding on voltage-dependent Ca2+ channels. J. Biol. Chem. 275, 39786–39792 (2000).
Peterson, B.Z., DeMaria, C.D. & Yue, D.T. Calmodulin is the Ca2+ sensor for Ca2+-dependent inactivation of 1-type calcium channels. Neuron 22, 549–558 (1999).
Seidenbecher, C.I., Reissner, C. & Kreutz, M.R. Caldendrins in the inner retina. in. Photorecept. Calcium 514, 451–463 (2002).
Dolmetsch, R. Excitation-transcription coupling: signaling by ion channels to the nucleus. Sci. STKE 2003, PE4 (2003).
Deisseroth, K., Heist, E.K. & Tsien, R.W. Translocation of calmodulin to the nucleus supports CREB phosphorylation in hippocampal neurons. Nature 392, 198–202 (1998).
Bading, H., Ginty, D.D. & Greenberg, M.E. Regulation of gene expression in hippocampal neurons by distinct calcium signaling pathways. Science 260, 181–186 (1993).
Mao, Z.X., Bonni, A., Xia, F., Nadal-Vicens, M. & Greenberg, M.E. Neuronal activity-dependent cell survival mediated by transcription factor MEF2. Science 286, 785–790 (1999).
Morgans, C.W., El Far, O., Berntson, A., Wassle, H. & Taylor, W.R. Calcium extrusion from mammalian photoreceptor terminals. J. Neurosci. 18, 2467–2474 (1998).
Morgans, C.W. Calcium channel heterogeneity among cone photoreceptors in the tree shrew retina. Eur. J. Neurosci. 11, 2989–2993 (1999).
Berntson, A., Taylor, W.R. & Morgans, C.W. Molecular identity, synaptic localization, and physiology o0f calcium channels in retinal bipolar cells. J. Neurosci. Res. 71, 146–151 (2003).
Bech-Hansen, N.T. et al. Loss-of-function mutations in a calcium-channel α1-subunit gene in Xp11.23 cause incomplete X-linked congenital stationary night blindness. Nat. Genet. 19, 264–267 (1998).
Haeseleer, F., Imanishi, Y., Saperstein, D.A. & Palczewski, K. Gene transfer mediated by recombinant baculovirus into mouse eye. Invest. Ophthalmol. Vis. Sci. 42, 3294–3300 (2001).
Ellis, S.B. et al. Sequence and expression of messenger RNAs encoding the α1-subunit and α2-subunit of a Dhp-sensitive calcium channel. Science 241, 1661–1664 (1988).
Perezreyes, E. et al. Cloning and expression of a cardiac brain β-subunit of the L-type calcium channel. J. Biol. Chem. 267, 1792–1797 (1992).
Haeseleer, F. et al. Molecular characterization of a third member of the guanylyl cyclase–activating protein subfamily. J. Biol. Chem. 274, 6526–6535 (1999).
Imanishi, Y. et al. Characterization of retinal guanylate cyclase–activating protein 3 (GCAP3) from zebrafish to man. Eur. J. Neurosci. 15, 63–78 (2002).
Batten, M.L. et al. Lecithin-retinol acyltransferase is essential for accumulation of all-trans-retinyl esters in the eye and in the liver. J. Biol. Chem. 279, 10422–10432 (2004).
Maeda, T. et al. Evaluation of the role of the retinal G protein–coupled receptor (RGR) in the vertebrate retina in vivo. J. Neurochem. 85, 944–956 (2003).
Acknowledgements
We thank W. Baehr, P. Detwiler, T. Doan, S. Bajjalieh and A. Polans for comments on the manuscript. We thank Y. Kim, M. Batten, A. Alekseev and M. Kalnoky for technical assistance. Anti-mGluR6 and anti-SV2 antibodies were gifts from S. Nakanishi and S.M. Bajjalieh, respectively. This research was supported by National Institutes of Health grants EY09339 to K.P., EY014561 to F.H., EY11850 to F.R. and NS044922 to A.L., the Whitehall Foundation to A.L., a grant from Research to Prevent Blindness (RPB), Inc. to the Department of Ophthalmology at the University of Washington and a grant from the E.K. Bishop Foundation. K.P. is a RPB Senior Investigator. D.P. was supported by a Vision Core Grant EY01730.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
The mouse Cabp4 gene and the targeting construct. (PDF 251 kb)
Supplementary Fig. 2
Cross-section of retinas analyzed by transmission EM. (PDF 2395 kb)
Supplementary Fig. 3
Retina histology of six to eight-month-old Cabp4−/− and Cabp4+/+ mice. (PDF 403 kb)
Supplementary Fig. 4
ERG responses from Cabp4−/− and Cabp4+/+ mice. (PDF 100 kb)
Rights and permissions
About this article
Cite this article
Haeseleer, F., Imanishi, Y., Maeda, T. et al. Essential role of Ca2+-binding protein 4, a Cav1.4 channel regulator, in photoreceptor synaptic function. Nat Neurosci 7, 1079–1087 (2004). https://doi.org/10.1038/nn1320
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1320
- Springer Nature America, Inc.
This article is cited by
-
Unusual OCT findings in a patient with CABP4-associated cone–rod synaptic disorder
Documenta Ophthalmologica (2024)
-
Caldendrin represses neurite regeneration and growth in dorsal root ganglion neurons
Scientific Reports (2023)
-
Function of cone and cone-related pathways in CaV1.4 IT mice
Scientific Reports (2021)
-
Transmission at rod and cone ribbon synapses in the retina
Pflügers Archiv - European Journal of Physiology (2021)
-
Presynaptic calcium channels: specialized control of synaptic neurotransmitter release
Nature Reviews Neuroscience (2020)