Abstract
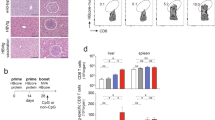
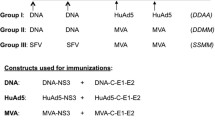
Three percent of the world's population is chronically infected with the hepatitis C virus (HCV) and at risk of developing liver cancer. Effective cellular immune responses are deemed essential for spontaneous resolution of acute hepatitis C and long-term protection. Here we describe a new T-cell HCV genetic vaccine capable of protecting chimpanzees from acute hepatitis induced by challenge with heterologous virus. Suppression of acute viremia in vaccinated chimpanzees occurred as a result of massive expansion of peripheral and intrahepatic HCV-specific CD8+ T lymphocytes that cross-reacted with vaccine and virus epitopes. These findings show that it is possible to elicit effective immunity against heterologous HCV strains by stimulating only the cellular arm of the immune system, and suggest a path for new immunotherapy against highly variable human pathogens like HCV, HIV or malaria, which can evade humoral responses.





Similar content being viewed by others
Accession codes
References
Seeff, L.B. Natural history of chronic hepatitis C. Hepatology 36, S35–S46 (2002).
Poynard, T., Yuen, M.F., Ratziu, V. & Lai, C.L. Viral hepatitis C. Lancet 362, 2095–2100 (2003).
Houghton M. & Abrignani S,. Prospects for a vaccine against the hepatitis C virus. Nature 436, 961–966 (2005).
Choo, Q.L. et al. Vaccination of chimpanzees against infection by the hepatitis C virus. Proc. Natl. Acad. Sci. USA 91, 1294–1298 (1994).
Farci, P. et al. Prevention of hepatitis C virus infection in chimpanzees by hyperimmune serum against the hypervariable region 1 of the envelope 2 protein. Proc. Natl. Acad. Sci. USA 93, 15394–15399 (1996).
Farci, P. et al. Lack of protective immunity against reinfection with hepatitis C virus. Science 258, 135–140 (1992).
Lai, M.E. et al. Hepatitis C virus in multiple episodes of acute hepatitis in polytransfused thalassaemic children. Lancet 343, 388–390 (1994).
Post, J.J. et al. Clearance of hepatitis C viremia associated with cellular immunity in the absence of seroconversion in the hepatitis C incidence and transmission in prisons study cohort. J. Infect. Dis. 189, 1846–1855 (2004).
Cooper, S. et al. Analysis of a successful immune response against hepatitis C virus. Immunity 4, 439–449 (1999).
Bowen, D.G. & Walker, C.M. Adaptive immune responses in acute and chronic hepatitis C virus infection. Nature 436, 946–952 (2005).
Shoukry, N.H. et al. Memory CD8+ T cells are required for protection from persistent hepatitis C virus infection. J. Exp. Med. 197, 1645–1655 (2003).
Grakoui, A. et al. HCV persistence and immune evasion in the absence of memory T cell help. Science 302, 659–662 (2003).
Lechner, F. et al. Analysis of successful immune responses in persons infected with hepatitis C virus. J. Exp. Med. 191, 1499–1512 (2000).
Spada, E. et al. Multi-specific T-cell response and negative HCV RNA tests during acute HCV infection are early prognostic factors of spontaneous clearance. Gut 53, 1673–1681 (2004).
Thimme, R. et al. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J. Exp. Med. 194, 1395–1406 (2001).
Thimme, R. et al. Viral and immunological determinants of hepatitis C virus clearance, persistence, and disease. Proc. Natl. Acad. Sci. USA 99, 15661–15668 (2002).
Wedemeyer, H. et al. Impaired effector function of hepatitis C virus-specific CD8+ T cells in chronic hepatitis C virus infection. J. Immunol. 169, 3447–3458 (2002).
Missale, G. et al. Different clinical behaviors of acute hepatitis C virus infection are associated with different vigor of the anti-viral cell-mediated immune response. J. Clin. Invest. 98, 706–714 (1996).
Diepolder, H.M. et al. Possible mechanism involving T-lymphocyte response to non-structural protein 3 in viral clearance in acute hepatitis C virus infection. Lancet 346, 1006–1007 (1995).
Gerlach, J.T. et al. Recurrence of hepatitis C virus after loss of virus-specific CD4+ T-cell response in acute hepatitis C. Gastroenterology 117, 933–941 (1999).
Cox, A.S. Comprehensive analyses of CD8+ T cell responses during longitudinal study of acute human hepatitis C. Hepatology 42, 104–112 (2005).
Lechner, F. et al. CD8+ T lymphocyte responses are induced during acute hepatitis C virus infection but are not sustained. Eur. J. Immunol. 30, 2479–2487 (2000).
Takaki, A. et al. Cellular immune responses persist and humoral responses decrease two decades after recovery from a single-source outbreak of hepatitis C. Nat. Med. 6, 578–582 (2000).
Bassett, S. et al. Protective immune response to hepatitis C virus in chimpanzees rechallenged following clearance of primary infection. Hepatology 33, 1479–1487 (2001).
Lanford, R.E. et al. Cross-genotype immunity to hepatitis C virus. J. Virol. 78, 1575–1581 (2004).
Mehta, S.H. et al. Protection against persistence of hepatitis C. Lancet 359, 1478–1483 (2002).
Takamizawa, A.C. et al. Structure and organization of the hepatitis C virus genome isolated from human carriers. J. Virol. 65, 1105–1113 (1991).
Aste-Amezaga, M. et al. Quantitative adenovirus neutralization assays based on the secreted alkaline phosphatase reporter gene: application in epidemiologic studies and in the design of adenovector vaccines. Hum. Gene Ther. 15, 293–304 (2004).
Capone, S. et al. A novel Ad6 based HCV vector that overcomes pre-existing anti-Ad5 immunity and induces potent and broad cellular immune responses in Rhesus Macaques. J Virol. (in the press).
Caulfield, M.J. et al. Sustained peptide-specific gamma interferon T-cell response in rhesus macaques immunized with human immunodeficiency virus gag DNA vaccines. J. Virol. 76, 10038–10043 (2002).
Forns, X. et al. Vaccination of chimpanzees with plasmid DNA encoding the hepatitis C virus (HCV) envelope E2 protein modified the infection after challenge with homologous monoclonal HCV. Hepatology 32, 618–625 (2000).
Rollier, C. et al. Control of heterologous hepatitis C virus infection in chimpanzees is associated with the quality of vaccine-induced peripheral T-helper immune response. J. Virol. 78, 187–196 (2004).
Puig, M., Major, M.E., Mihalik, K. & Feinstone, S.M. Immunization of chimpanzees with an envelope protein-based vaccine enhances specific humoral and cellular immune responses that delay hepatitis C virus infection. Vaccine 22, 991–1000 (2004).
Kuiken, C., Yusim, K., Boykin, L. & Richardson, R . The Los Alamos hepatitis C sequence database. Bioinformatics 21, 379–384 (2005).
Tatsis, N. & Ertl, H.C. Adenoviruses as vaccine vectors. Mol. Ther. 10, 4616–4629 (2004).
Shiver, J.W. & Emini, E.A. Recent advances in the development of HIV-1 vaccines using replication-incompetent adenovirus vectors. Annu. Rev. Med. 55, 355–372 (2004).
Zampaglione, I. et al. In vivo DNA gene electro-transfer: a systematic analysis of different electrical parameters. J. Gene Med. 7, 1475–1481 (2005).
Zucchelli, S. et al. Enhancing B- and T-cell immune response to a hepatitis C virus E2 DNA vaccine by intramuscular electrical gene transfer. J. Virol. 74, 11598–11607 (2000).
Major, M.E. et al. Hepatitis C virus kinetics and host responses associated with disease and outcome of infection in chimpanzees. Hepatology 39, 1709–1720 (2004).
Bassett, S., Brasky, K.M. & Lanford, R.E. Analysis of hepatitis C virus-inoculated chimpanzees reveals unexpected clinical profiles. J. Virol. 72, 2589–2599 (1998).
Alter, M.J. Hepatitis C virus infection in the United States. J. Hepatol. 31, 88–91 (1999).
Palitzsch, K.D. et al. Prevalence of antibodies against hepatitis C virus in the adult German population. Eur. J. Gastroenterol. Hepatol. 11, 1215–1220 (1999).
Riestra, S. et al. Prevalence of hepatitis C virus infection in the general population of northern Spain. Eur. J. Gastroenterol. Hepatol. 13, 477–481 (2001).
Casimiro, D.R. et al. Heterologous human immunodeficiency virus type 1 priming-boosting immunization strategies involving replication-defective adenovirus and poxvirus vaccine vectors. J. Virol. 78, 11434–11438 (2004).
Guglietta, S. et al. Positive selection of cytotoxic T lymphocyte escape variants during acute hepatitis C virus infection. Eur J. Immunology 35, 2627–2637 (2005).
Acknowledgements
We acknowledge R.H. Purcell and J. Bukh for provision of the H77 viral inoculum; J. Burns and L. Kierstead for assistance with the intrahepatic lymphocyte cultures; P. Sczerba for clinical chemistry; C.M. Walker and R.H. Purcell for discussion; J. Fontenot, D. Hasselschwert, N. Smith and the staff at the New Iberia Research Center for the care of chimpanzees and samples and M. Emili for artwork. We also thank C.M. Rice, D. Mathis, J.W. Mattaj and G. Migliaccio for critical reading of the manuscript. This paper is dedicated to the memory of Elisabetta Sporeno, who will be missed by all her colleagues. This work was supported in part by Ministero dell'Istruzione, dell'Università e della Ricerca-MIUR (project “Vaccino per l'epatite C”, Legge n. 488/1992).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Representative IFN-γ ICS with PBMCs from two vaccinated chimpanzees. (PDF 166 kb)
Supplementary Fig. 2
HCV-specific T-cell proliferation by BrdU assay in vaccinated chimpanzees. (PDF 27 kb)
Supplementary Fig. 3
Kinetics of HCV RNA, amino acid substitution and epitope recognition for chimpanzee 96A022. (PDF 108 kb)
Rights and permissions
About this article
Cite this article
Folgori, A., Capone, S., Ruggeri, L. et al. A T-cell HCV vaccine eliciting effective immunity against heterologous virus challenge in chimpanzees. Nat Med 12, 190–197 (2006). https://doi.org/10.1038/nm1353
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1353
- Springer Nature America, Inc.
This article is cited by
-
Development of therapeutic vaccines for the treatment of diseases
Molecular Biomedicine (2022)
-
Vaccination at the forefront of the fight against hepatitis B and C
Nature Reviews Gastroenterology & Hepatology (2022)
-
Hepatitis C virus DNA vaccines: a systematic review
Virology Journal (2021)
-
Optimising T cell (re)boosting strategies for adenoviral and modified vaccinia Ankara vaccine regimens in humans
npj Vaccines (2020)
-
Protective cellular immune response against hepatitis C virus elicited by chimeric protein formulations in BALB/c mice
Archives of Virology (2020)