Abstract
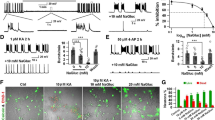
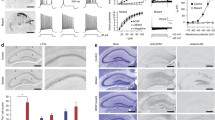
During development, activation of Cl−-permeable GABAA receptors (GABAA-R) excites neurons as a result of elevated intracellular Cl− levels and a depolarized Cl− equilibrium potential (ECl). GABA becomes inhibitory as net outward neuronal transport of Cl− develops in a caudal-rostral progression. In line with this caudal-rostral developmental pattern, GABAergic anticonvulsant compounds inhibit motor manifestations of neonatal seizures but not cortical seizure activity. The Na+-K+-2Cl− cotransporter (NKCC1) facilitates the accumulation of Cl− in neurons. The NKCC1 blocker bumetanide shifted ECl negative in immature neurons, suppressed epileptiform activity in hippocampal slices in vitro and attenuated electrographic seizures in neonatal rats in vivo. Bumetanide had no effect in the presence of the GABAA-R antagonist bicuculline, nor in brain slices from NKCC1-knockout mice. NKCC1 expression level versus expression of the Cl−-extruding transporter (KCC2) in human and rat cortex showed that Cl− transport in perinatal human cortex is as immature as in the rat. Our results provide evidence that NKCC1 facilitates seizures in the developing brain and indicate that bumetanide should be useful in the treatment of neonatal seizures.






Similar content being viewed by others
References
Ronen, G.M., Penney, S. & Andrews, W. The epidemiology of clinical neonatal seizures in Newfoundland: a population-based study. J. Pediatr. 134, 71–75 (1999).
Painter, M.J., Bergman, I. & Crumrine, P. Neonatal seizures. Pediatr. Clin. North Am. 33, 91–109 (1986).
Scher, M.S. et al. Electrographic seizures in preterm and full-term neonates: clinical correlates, associated brain lesions, and risk for neurologic sequelae. Pediatrics 91, 128–134 (1993).
Brunquell, P.J., Glennon, C.M., DiMario, F.J. Jr., Lerer, T. & Eisenfeld, L. Prediction of outcome based on clinical seizure type in newborn infants. J. Pediatr. 140, 707–712 (2002).
Jensen, F.E., Holmes, G.L., Lombroso, C.T., Blume, H.K. & Firkusny, I.R. Age-dependent changes in long-term seizure susceptibility and behavior after hypoxia in rats. Epilepsia 33, 971–980 (1992).
Baram, T.Z. Long-term neuroplasticity and functional consequences of single versus recurrent early-life seizures. Ann. Neurol. 54, 701–705 (2003).
Holmes, G.L. Effects of early seizures on later behavior and epileptogenicity. Ment. Retard. Dev. Disabil. Res. Rev. 10, 101–105 (2004).
Swann, J.W. The impact of seizures on developing hippocampal networks. Prog. Brain Res. 147, 347–354 (2005).
Sanchez, R.M., Dai, W., Levada, R.E., Lippman, J.J. & Jensen, F.E. AMPA/kainate receptor-mediated downregulation of GABAergic synaptic transmission by calcineurin after seizures in the developing rat brain. J. Neurosci. 25, 3442–3451 (2005).
Connell, J., Oozeer, R., de Vries, L., Dubowitz, L.M. & Dubowitz, V. Clinical and EEG response to anticonvulsants in neonatal seizures. Arch. Dis. Child. 64, 459–464 (1989).
Painter, M.J. et al. Phenobarbital compared with phenytoin for the treatment of neonatal seizures. N. Engl. J. Med. 341, 485–489 (1999).
Owens, D.F. & Kriegstein, A.R. Is there more to GABA than synaptic inhibition? Nat. Rev. Neurosci. 3, 715–727 (2002).
Ben Ari, Y. Excitatory actions of gaba during development: the nature of the nurture. Nat. Rev. Neurosci. 3, 728–739 (2002).
Dzhala, V.I. & Staley, K.J. Excitatory actions of endogenously released GABA contribute to initiation of ictal epileptiform activity in the developing hippocampus. J. Neurosci. 23, 1840–1846 (2003).
Khazipov, R. et al. Developmental changes in GABAergic actions and seizure susceptibility in the rat hippocampus. Eur. J. Neurosci. 19, 590–600 (2004).
Stein, V., Hermans-Borgmeyer, I., Jentsch, T.J. & Hubner, C.A. Expression of the KCl cotransporter KCC2 parallels neuronal maturation and the emergence of low intracellular chloride. J. Comp. Neurol. 468, 57–64 (2004).
Scher, M.S., Alvin, J., Gaus, L., Minnigh, B. & Painter, M.J. Uncoupling of EEG-clinical neonatal seizures after antiepileptic drug use. Pediatr. Neurol. 28, 277–280 (2003).
Staley, K., Smith, R., Schaack, J., Wilcox, C. & Jentsch, T.J. Alteration of GABAA receptor function following gene transfer of the CLC-2 chloride channel. Neuron 17, 543–551 (1996).
Delpire, E. Cation-chloride cotransporters in neuronal communication. News Physiol. Sci. 15, 309–312 (2000).
Payne, J.A., Rivera, C., Voipio, J. & Kaila, K. Cation-chloride co-transporters in neuronal communication, development and trauma. Trends Neurosci. 26, 199–206 (2003).
Yamada, J. et al. Cl- uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J. Physiol. (Lond.) 557, 829–841 (2004).
Isenring, P., Jacoby, S.C., Payne, J.A. & Forbush, B. III. Comparison of Na-K-Cl cotransporters. NKCC1, NKCC2, and the HEK cell Na-L-Cl cotransporter. J. Biol. Chem. 273, 11295–11301 (1998).
Hannaert, P., Alvarez-Guerra, M., Pirot, D., Nazaret, C. & Garay, R.P. Rat NKCC2/NKCC1 cotransporter selectivity for loop diuretic drugs. Naunyn Schmiedebergs Arch. Pharmacol. 365, 193–199 (2002).
Plotkin, M.D., Snyder, E.Y., Hebert, S.C. & Delpire, E. Expression of the Na-K-2Cl cotransporter is developmentally regulated in postnatal rat brains: a possible mechanism underlying GABA's excitatory role in immature brain. J. Neurobiol. 33, 781–795 (1997).
Wang, C. et al. Developmental changes in KCC1, KCC2, and NKCC1 mRNA expressions in the rat brain. Brain Res. Dev. Brain Res. 139, 59–66 (2002).
Clayton, G.H., Owens, G.C., Wolff, J.S. & Smith, R.L. Ontogeny of cation-Cl– cotransporter expression in rat neocortex. Brain Res. Dev. Brain Res. 109, 281–292 (1998).
Rivera, C. et al. The K+/Cl- co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 397, 251–255 (1999).
Sullivan, J.E., Witte, M.K., Yamashita, T.S., Myers, C.M. & Blumer, J.L. Pharmacokinetics of bumetanide in critically ill infants. Clin. Pharmacol. Ther. 60, 405–413 (1996).
Lopez-Samblas, A.M., Adams, J.A., Goldberg, R.N. & Modi, M.W. The pharmacokinetics of bumetanide in the newborn infant. Biol. Neonate 72, 265–272 (1997).
Lu, J., Karadsheh, M. & Delpire, E. Developmental regulation of the neuronal-specific isoform of K-Cl cotransporter KCC2 in postnatal rat brains. J. Neurobiol. 39, 558–568 (1999).
Sankar, R. & Painter, M.J. Neonatal seizures: after all these years we still love what doesn't work. Neurology 64, 776–777 (2005).
Twyman, R.E., Rogers, C.J. & Macdonald, R.L. Differential regulation of gamma-aminobutyric acid receptor channels by diazepam and phenobarbital. Ann. Neurol. 25, 213–220 (1989).
Staley, K. Enhancement of the excitatory actions of GABA by barbiturates and benzodiazepines. Neurosci. Lett. 146, 105–107 (1992).
Dzhala, V.I. & Staley, K.J. Transition from interictal to ictal activity in limbic networks in vitro. J. Neurosci. 23, 7873–7880 (2003).
Dzhala, V.I. & Staley, K.J. Mechanisms of fast ripples in the hippocampus. J. Neurosci. 24, 8896–8906 (2004).
Ben Ari, Y., Cherubini, E., Corradetti, R. & Gaiarsa, J.L. Giant synaptic potentials in immature rat CA3 hippocampal neurones. J. Physiol. (Lond.) 416, 303–325 (1989).
Khalilov, I., Dzhala, V., Ben Ari, Y. & Khazipov, R. Dual role of GABA in the neonatal rat hippocampus. Dev. Neurosci. 21, 310–319 (1999).
Leinekugel, X. et al. Correlated bursts of activity in the neonatal hippocampus in vivo. Science 296, 2049–2052 (2002).
Delpire, E., Lu, J., England, R., Dull, C. & Thorne, T. Deafness and imbalance associated with inactivation of the secretory Na-K-2Cl co-transporter. Nat. Genet. 22, 192–195 (1999).
Sung, K.W., Kirby, M., McDonald, M.P., Lovinger, D.M. & Delpire, E. Abnormal GABAA receptor-mediated currents in dorsal root ganglion neurons isolated from Na-K-2Cl cotransporter null mice. J. Neurosci. 20, 7531–7538 (2000).
Kanaka, C. et al. The differential expression patterns of messenger RNAs encoding K-Cl cotransporters (KCC1,2) and Na-K-2Cl cotransporter (NKCC1) in the rat nervous system. Neuroscience 104, 933–946 (2001).
Owens, D.F., Boyce, L.H., Davis, M.B. & Kriegstein, A.R. Excitatory GABA responses in embryonic and neonatal cortical slices demonstrated by gramicidin perforated-patch recordings and calcium imaging. J. Neurosci. 16, 6414–6423 (1996).
Staley, K.J. & Mody, I. Shunting of excitatory input to dentate gyrus granule cells by a depolarizing GABAA receptor-mediated postsynaptic conductance. J. Neurophysiol. 68, 197–212 (1992).
Jin, X., Huguenard, J.R. & Prince, D.A. Impaired Cl− extrusion in layer V pyramidal neurons of chronically injured epileptogenic neocortex. J. Neurophysiol. 93, 2117–2126 (2005).
Hochman, D.W., Baraban, S.C., Owens, J.W. & Schwartzkroin, P.A. Dissociation of synchronization and excitability in furosemide blockade of epileptiform activity. Science 270, 99–102 (1995).
Stringer, J.L. & Pan, E. Effect of seizures and diuretics on the osmolality of the cerebrospinal fluid. Brain Res. 745, 328–330 (1997).
Haglund, M.M. & Hochman, D.W. Furosemide and mannitol suppression of epileptic activity in the human brain. J. Neurophysiol. 94, 907–918 (2005).
Hesdorffer, D.C., Stables, J.P., Hauser, W.A., Annegers, J.F. & Cascino, G. Are certain diuretics also anticonvulsants? Ann. Neurol. 50, 458–462 (2001).
Cohen, I., Navarro, V., Clemenceau, S., Baulac, M. & Miles, R. On the origin of interictal activity in human temporal lobe epilepsy in vitro. Science 298, 1418–1421 (2002).
Ulfig, N., Nickel, J. & Bohl, J. Monoclonal antibodies SMI 311 and SMI 312 as tools to investigate the maturation of nerve cells and axonal patterns in human fetal brain. Cell Tissue Res. 291, 433–443 (1998).
Acknowledgements
This work was supported by the US National Institutes of Health and National Institute of Neurological Disorders and Stroke grants NS34360, NS34700, and NS40109 (to K.J.S.), NS36758 (to E.D.), NS31718 (to F.E.J.), NS38475 (to F.E.J. and D.M.T.) and the Hearst Foundation (to D.M.T.). Some tissue samples were provided by the University of Maryland Brain Tissue Bank for Developmental Disorders, Baltimore, Maryland, USA supported by N01-HD-43368.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Dzhala, V., Talos, D., Sdrulla, D. et al. NKCC1 transporter facilitates seizures in the developing brain. Nat Med 11, 1205–1213 (2005). https://doi.org/10.1038/nm1301
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1301
- Springer Nature America, Inc.
This article is cited by
-
Acute symptomatic seizures in newborns: a narrative review
Acta Epileptologica (2024)
-
Emergence of consciousness from anesthesia through ubiquitin degradation of KCC2 in the ventral posteromedial nucleus of the thalamus
Nature Neuroscience (2023)
-
Imbalanced expression of cation-chloride cotransporters as a potential therapeutic target in an Angelman syndrome mouse model
Scientific Reports (2023)
-
New GABA-Targeting Therapies for the Treatment of Seizures and Epilepsy: I. Role of GABA as a Modulator of Seizure Activity and Recently Approved Medications Acting on the GABA System
CNS Drugs (2023)
-
A Phase III Study of Bumetanide Oral Liquid Formulation for the Treatment of Children and Adolescents Aged Between 7 and 17 Years with Autism Spectrum Disorder (SIGN 1 Trial): Participant Baseline Characteristics
Child Psychiatry & Human Development (2023)