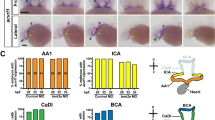
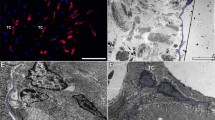
Abstract
Lymph vessels control fluid homeostasis, immunity and metastasis. Unraveling the molecular basis of lymphangiogenesis has been hampered by the lack of a small animal model that can be genetically manipulated. Here, we show that Xenopus tadpoles develop lymph vessels from lymphangioblasts or, through transdifferentiation, from venous endothelial cells. Lymphangiography showed that these lymph vessels drain lymph, through the lymph heart, to the venous circulation. Morpholino-mediated knockdown of the lymphangiogenic factor Prox1 caused lymph vessel defects and lymphedema by impairing lymphatic commitment. Knockdown of vascular endothelial growth factor C (VEGF-C) also induced lymph vessel defects and lymphedema, but primarily by affecting migration of lymphatic endothelial cells. Knockdown of VEGF-C also resulted in aberrant blood vessel formation in tadpoles. This tadpole model offers opportunities for the discovery of new regulators of lymphangiogenesis.





Similar content being viewed by others
References
Saharinen, P., Tammela, T., Karkkainen, M.J. & Alitalo, K. Lymphatic vasculature: development, molecular regulation and role in tumor metastasis and inflammation. Trends Immunol. 25, 387–395 (2004).
Jain, R.K. Angiogenesis and lymphangiogenesis in tumors: insights from intravital microscopy. Cold Spring Harb. Symp. Quant. Biol. 67, 239–248 (2002).
Alitalo, K. & Carmeliet, P. Molecular mechanisms of lymphangiogenesis in health and disease. Cancer Cell 1, 219–227 (2002).
Asellius, G. Asellii Cremonensis antomici ticiensis qua sententiae anatomicae multae, nel perperam receptae illustrantur. De Lacteibus sive lacteis venis Quarto Vasorum Mesaroicum genere novo invente Gasp. (Mediolani, Milan, 1627).
Lawson, N.D. & Weinstein, B.M. Arteries and veins: making a difference with zebrafish. Nat. Rev. Genet. 3, 674–682 (2002).
Daniels, C.B. et al. Regenerating lizard tails: a new model for investigating lymphangiogenesis. FASEB J. 17, 479–481 (2003).
Wilting, J., Schneider, M., Papoutski, M., Alitalo, K. & Christ, B. An avian model for studies of embryonic lymphangiogenesis. Lymphology 33, 81–94 (2000).
Olszewski, W. Regulation of water balance between blood and lymph in the frog, Rana pipiens. Lymphology 26, 154–155 (1993).
Liu, Z.Y. & Casley-Smith, J.R. The fine structure of the amphibian lymph sac. Lymphology 22, 31–35 (1989).
Hoyer, M. Untersuchungen ueber das Lymphgefaessystem der Froschlarven. Bull. Acad. Cracov. Teill II, 451–464 (1905).
Sabin, F.R. On the origin of the lymphatic system from the veins and the development of the lymph hearts and thoracic duct in the pig. Am. J. Anat. 1, 367–389 (1902).
Wigle, J.T. & Oliver, G. Prox1 function is required for the development of the murine lymphatic system. Cell 98, 769–778 (1999).
Wigle, J.T. et al. An essential role for Prox1 in the induction of the lymphatic endothelial cell phenotype. EMBO J. 21, 1505–1513 (2002).
Devic, E., Paquereau, L., Vernier, P., Knibiehler, B. & Audigier, Y. Expression of a new G protein-coupled receptor X-msr is associated with an endothelial lineage in Xenopus laevis . Mech. Dev. 59, 129–140 (1996).
Cleaver, O., Tonissen, K.F., Saha, M.S. & Krieg, P.A. Neovascularization of the Xenopus embryo. Dev. Dyn. 210, 66–77 (1997).
Levine, A.J., Munoz-Sanjuan, I., Bell, E., North, A.J. & Brivanlou, A.H. Fluorescent labeling of endothelial cells allows in vivo, continuous characterization of the vascular development of Xenopus laevis . Dev. Biol. 254, 50–67 (2003).
Karkkainen, M.J. et al. Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins. Nat. Immunol. 5, 74–80 (2004).
Baldwin, M.E. et al. Vascular endothelial growth factor D is dispensable for development of the lymphatic system. Mol. Cell. Biol. 25, 2441–2449 (2005).
Achen, M.G. et al. Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk1) and VEGF receptor 3 (Flt4). Proc. Natl. Acad. Sci. USA 95, 548–553 (1998).
Joukov, V. et al. A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. EMBO J. 15, 290–298 (1996).
Saaristo, A. et al. Adenoviral VEGF-C overexpression induces blood vessel enlargement, tortuosity, and leakiness but no sprouting angiogenesis in the skin or mucous membranes. FASEB J. 16, 1041–1049 (2002).
Ober, E.A. et al. VEGF-C is required for vascular development and endoderm morphogenesis in zebrafish. EMBO Rep. 5, 78–84 (2004).
Wilting, J. et al. Development of the avian lymphatic system. Microsc. Res. Tech. 55, 81–91 (2001).
Newport, J. & Kirschner, M. A major developmental transition in early Xenopus embryos: II. Control of the onset of transcription. Cell 30, 687–696 (1982).
Nieuwkoop, P.J. & Faber, J. Normal table of Xenopus laevis (Daudin): A systematical and chronological survey of the development from the fertilized egg till the end of metamorphosis (Garand Publishing, New York, 1994).
Harland, R.M. In situ hybridization: an improved whole-mount method for Xenopus embryos. Methods Cell Biol. 36, 685–695 (1991).
Carmeliet, P. et al. Urokinase-generated plasmin activates matrix metalloproteinases during aneurysm formation. Nat. Genet. 17, 439–444 (1997).
Padera, T.P. et al. Lymphatic metastasis in the absence of functional intratumor lymphatics. Science 296, 1883–1886 (2002).
Acknowledgements
A.N. is sponsored by a EU Sixth Framework Programme Marie Curie Intra European Fellowship; E.N. by the Fund for Scientific Research-Flanders (FWO); M.K. by the FCT (grant SFRH/BD/9349/2002, Portugal); M.S. and C.F. by the Deutsche Forschungsgemeinschaft (Germany). This work is supported, in part, by grant G.0567.05 from the FWO, by an unrestricted Bristol-Myers-Squibb grant, by a grant GOA2001/09 from Concerted Research Activities, Belgium and by grant CA-85140 from the US National Cancer Institute. The authors thank S. Tomarev and A. Ciau-Uitz for providing the rabbit anti-mouse Prox1 antibody and the Xenopus Fli and Msr cDNA, respectively, and K. Brepoels, A. Bouché, A. Claeys, M. De Mol, B. Hermans, S. Jansen, L. Kieckens, S. Louwette, A. Manderveld, W. Martens, L. Notebaert, A. Van Nuffelen, B. Vanwetswinkel (Leuven) and D. Fukumura (Boston) for their contribution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Development of the rostral lymph sac and lymph heart. (PDF 560 kb)
Supplementary Fig. 2
Scheme of lymphatic cell migration (PDF 211 kb)
Supplementary Fig. 3
Identity and functionality of lymph vessels and heart. (PDF 277 kb)
Supplementary Fig. 4
Morpholinos block translation of Prox1 and VEGF-C. (PDF 290 kb)
Supplementary Fig. 5
Dose-dependence of Prox1 or VEGF-C knockdown. (PDF 83 kb)
Supplementary Fig. 6
Prox1 knockdown using the splice site morpholino. (PDF 401 kb)
Supplementary Movie 1
Intravital video-microscopy of VCLV and PCV. The first half of the movie shows the ventral caudal lymph vessel (VCLV), visualized by the progressive drainage of FITC-dextran after lymphangiography. In the second half of the movie, the microscope filters were switched to phase-contrast microscopy, revealing the posterior cardinal vein (PCV), containing flowing red blood cells. The movie was made using the same tadpole. Top of movie: dorsal side of the tadpole. (MOV 3125 kb)
Supplementary Movie 2
Intravital video-microscopy of lymph heart. Lymphangiography with FITC-dextran, showing the lymph heart, pumping the green fluorescent dye into the venous circulation through a set of valves. (AVI 2201 kb)
Rights and permissions
About this article
Cite this article
Ny, A., Koch, M., Schneider, M. et al. A genetic Xenopus laevis tadpole model to study lymphangiogenesis. Nat Med 11, 998–1004 (2005). https://doi.org/10.1038/nm1285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1285
- Springer Nature America, Inc.
This article is cited by
-
Lymphatic vessel: origin, heterogeneity, biological functions, and therapeutic targets
Signal Transduction and Targeted Therapy (2024)
-
The lymphatic vascular system: much more than just a sewer
Cell & Bioscience (2022)
-
Vascular endothelial cell specification in health and disease
Angiogenesis (2021)
-
Non-canonical WNT-signaling controls differentiation of lymphatics and extension lymphangiogenesis via RAC and JNK signaling
Scientific Reports (2019)
-
Multipotent Adult Progenitor Cells Support Lymphatic Regeneration at Multiple Anatomical Levels during Wound Healing and Lymphedema
Scientific Reports (2018)