Abstract
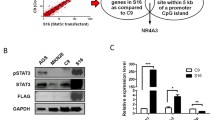
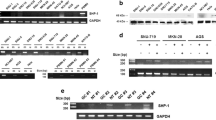
Hepatocellular carcinoma (HCC) is a major cause of cancer death, but the molecular mechanism for its development beyond its initiation has not been well characterized. Suppressor of cytokine signaling (SOCS-1; also known as JAB and SSI-1) switches cytokine signaling 'off' by means of its direct interaction with Janus kinase (JAK). We identified aberrant methylation in the CpG island of SOCS-1 that correlated with its transcription silencing in HCC cell lines. The incidence of aberrant methylation was 65% in the 26 human primary HCC tumor samples analyzed. Moreover, the restoration of SOCS-1 suppressed both growth rate and anchorage-independent growth of cells in which SOCS-1 was methylation-silenced and JAK2 was constitutively activated. This growth suppression was caused by apoptosis and was reproduced by AG490, a specific, chemical JAK2 inhibitor that reversed constitutive phosphorylation of STAT3 in SOCS-1 inactivated cells. The high prevalence of the aberrant SOCS-1 methylation and its growth suppression activity demonstrated the importance of the constitutive activation of the JAK/STAT pathway in the development of HCC. Our results also indicate therapeutic strategies for the treatment of HCC including use of SOCS-1 in gene therapy and inhibition of JAK2 by small molecules, such as AG490.





Similar content being viewed by others
References
Masuhara, M. et al. Cloning and characterization of novel CIS family genes. Biochem. Biophys. Res. Commun. 239, 439–446 (1997).
Hilton, D.J. et al. Twenty proteins containing a C-terminal SOCS box form five structural classes. Proc. Natl. Acad. Sci. USA 95, 114–119 (1998).
Starr, R. et al. A family of cytokine-inducible inhibitors of signalling. Nature 387, 917–921 (1997).
Endo, T.A. et al. A new protein containing an SH2 domain that inhibits JAK kinases. Nature 387, 921–924 (1997).
Naka, T. et al. Structure and function of a new STAT-induced STAT inhibitor. Nature 387, 924–929 (1997).
Kramer, J.A., Adams, M.D., Singh, G.B., Doggett, N.A. & Krawetz, S.A. Extended analysis of the region encompassing the PRM1→PRM2→TNP2 domain: genomic organization, evolution and gene identification. J. Exp. Zool. 282, 245–253 (1998).
Losman, J.A., Chen, X.P., Hilton, D. & Rothman, P. Cutting edge: SOCS-1 is a potent inhibitor of IL-4 signal transduction. J. Immunol. 162, 3770–3774 (1999).
Ihle, J.N. Cytokine receptor signalling. Nature 377, 591–594 (1995).
Ramponi, G. et al. Overexpression of a synthetic phosphotyrosine protein phosphatase gene inhibits normal and transformed cell growth. Int. J. Cancer 51, 652–656 (1992).
Li, J. et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275, 1943–1947 (1997).
Furnari, F.B., Lin, H., Huang, H.S. & Cavenee, W.K. Growth suppression of glioma cells by PTEN requires a functional phosphatase catalytic domain. Proc. Natl. Acad. Sci. USA 94, 12479–12484 (1997).
Shultz, L.D. et al. Mutations at the murine motheaten locus are within the hematopoietic cell protein-tyrosine phosphatase (Hcph) gene. Cell 73, 1445–1454 (1993).
Klingmuller, U., Lorenz, U., Cantley, L.C., Neel, B.G. & Lodish, H.F. Specific recruitment of SH-PTP1 to the erythropoietin receptor causes inactivation of JAK2 and termination of proliferative signals. Cell 80, 729–738 (1995).
Garcia, R. et al. Constitutive activation of Stat3 in fibroblasts transformed by diverse oncoproteins and in breast carcinoma cells. Cell Growth Differ. 8, 1267–1276 (1997).
Luo, H., Hanratty, W.P. & Dearolf, C.R. An amino acid substitution in the Drosophila hopTum-l Jak kinase causes leukemia-like hematopoietic defects. EMBO J. 14, 1412–1420 (1995).
Harrison, D.A., Binari, R., Nahreini, T.S., Gilman, M. & Perrimon, N. Activation of a Drosophila Janus kinase (JAK) causes hematopoietic neoplasia and developmental defects. EMBO J. 14, 2857–2865 (1995).
Lacronique, V. et al. A TEL-JAK2 fusion protein with constitutive kinase activity in human leukemia. Science 278, 1309–1312 (1997).
Bromberg, J.F. et al. Stat3 as an oncogene. Cell 98, 295–303 (1999).
Catlett-Falcone, R. et al. Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity 10, 105–115 (1999).
Herman, J.G. et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc. Natl. Acad. Sci. USA 91, 9700–9704 (1994).
Herman, J.G. et al. Inactivation of the CDKN2/p16/MTS1 gene is frequently associated with aberrant DNA methylation in all common human cancers. Cancer Res. 55, 4525–4530 (1995).
Esteller, M., Hamilton, S.R., Burger, P.C., Baylin, S.B. & Herman, J.G. Inactivation of the DNA repair gene O6-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer Res. 59, 793–797 (1999).
Graff, J.R. et al. E-cadherin expression is silenced by DNA hypermethylation in human breast and prostate carcinomas. Cancer Res. 55, 5195–5199 (1995).
Katzenellenbogen, R.A., Baylin, S.B. & Herman, J.G. Hypermethylation of the DAP-kinase CpG island is a common alteration in B-cell malignancies. Blood 93, 4347–4353 (1999).
Herman, J.G. et al. Incidence and functional consequences of hMLH1 promoter hypermethylation in colorectal carcinoma. Proc. Natl. Acad. Sci. USA 95, 6870–6875 (1998).
Nagai, H. et al. Aberration of genomic DNA in association with human hepatocellular carcinomas detected by 2-dimensional gel analysis. Cancer Res. 54, 1545–1550 (1994).
Yoshikawa, H. et al. Chromosome assignment of aberrant NotI restriction DNA fragments in primary hepatocellular carcinoma. Gene 197, 129–135 (1997).
Paulson, J.R. & Laemmli, U.K. The structure of histone-depleted metaphase chromosomes. Cell 12, 817–828 (1977).
Vogelstein, B., Pardoll, D.M. & Coffey, D.S. Supercoiled loops and eucaryotic DNA replication. Cell 22, 79–85 (1980).
Zlatanova, J.S. & van Holde, K.E. Chromatin loops and transcriptional regulation. Crit. Rev. Euk. Gene Exp. 2, 211–224 (1992).
Meydan, N. et al. Inhibition of acute lymphoblastic leukaemia by a Jak-2 inhibitor. Nature 379, 645–648 (1996).
Migone, T.S. et al. Constitutively activated Jak–STAT pathway in T cells transformed with HTLV-I. Science 269, 79–81 (1995).
Danial, N.N., Pernis, A. & Rothman, P.B. Jak–STAT signaling induced by the v-abl oncogene. Science 269, 1875–1877 (1995).
Weber-Nordt, R.M. et al. Constitutive activation of STAT proteins in primary lymphoid and myeloid leukemia cells and in Epstein-Barr virus (EBV)-related lymphoma cell lines. Blood 88, 809–816 (1996).
Cressman, D.E., Diamond, R.H. & Taub, R. Rapid activation of the Stat3 transcription complex in liver regeneration. Hepatology 21, 1443–1449 (1995).
Trautwein, C., Rakemann, T., Niehof, M., Rose-John, S. & Manns, M.P. Acute-phase response factor, increased binding, and target gene transcription during liver regeneration. Gastroenterology 110, 1854–1862 (1996).
Kishimoto, T., Taga, T. & Akira, S. Cytokine signal transduction. Cell 76, 253–262 (1994).
Darnell, J.E., Jr., Kerr, I.M. & Stark, G.R. Jak–STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 264, 1415–1421 (1994).
Starr, R. et al. Liver degeneration and lymphoid deficiencies in mice lacking suppressor of cytokine signaling-1. Proc. Natl. Acad. Sci. USA 95, 14395–14399 (1998).
Naka, T. et al. Accelerated apoptosis of lymphocytes by augmented induction of Bax in SSI-1 (STAT-induced STAT inhibitor-1) deficient mice. Proc. Natl. Acad. Sci. USA 95, 15577–15582 (1998).
Rashid, A. et al. Genetic alterations in hepatocellular carcinomas from the People's Republic of China: association between loss of heterozygosity on chromosome 4q and abnormalities of p53 tumor suppressor gene. Br. J. Cancer 80, 59–66 (1999).
Yoshikawa, H., Fujiyama, A., Nakai, K., Inazawa, J. & Matsubara, K. Detection and isolation of a novel human gene located on Xp11.2–p11.4 that escapes X-inactivation using a two-dimensional DNA mapping method. Genomics 49, 237–246 (1998).
Herman, J.G., Graff, J.R., Myohanen, S., Nelkin, B.D. & Baylin, S.B. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc. Natl. Acad. Sci. USA 93, 9821–9826 (1996).
Kramer, J.A. & Krawetz, S.A. PCR-based assay to determine nuclear matrix association. Biotechniques 22, 826–828 (1997).
Acknowledgements
We thank A. Hancock and E. Spillare for technical assistance; M. Rountree for technical advice; S. Baylin for critical comments on the manuscript; and S. Tamai for providing liver samples. H. Yoshikawa was supported in part by the NCI-JFCR scientist exchange program. Supported in part by P50 CA58184.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshikawa, H., Matsubara, K., Qian, GS. et al. SOCS-1, a negative regulator of the JAK/STAT pathway, is silenced by methylation in human hepatocellular carcinoma and shows growth-suppression activity. Nat Genet 28, 29–35 (2001). https://doi.org/10.1038/ng0501-29
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0501-29
- Springer Nature America, Inc.
This article is cited by
-
Identification and validation of the association of Janus kinase 2 mutations with the response to immune checkpoint inhibitor therapy
Inflammation Research (2024)
-
Alterations of SOCS1 and SOCS3 transcript levels, but not promoter methylation levels in subcutaneous adipose tissues in obese women
BMC Endocrine Disorders (2023)
-
Demethylation of CADM1 and SOCS1 using capsaicin in cervical cancer cell line
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)
-
Quantitative detection of methylated SOCS-1 in schizophrenia and bipolar disorder considering SOCS-1 -1478CA/del polymorphism and clinical parameters
Irish Journal of Medical Science (1971 -) (2023)
-
Relation of the methylation state of RUNX3 and p16 gene promoters with hepatocellular carcinoma in Egyptian patients
Egyptian Journal of Medical Human Genetics (2022)