Abstract
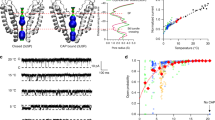
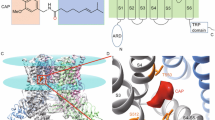
Capsaicin bestows spiciness by activating TRPV1 channel with exquisite potency and selectivity. Although a capsaicin-bound channel structure was previously resolved by cryo-EM at 4.2- to 4.5-Å resolution, capsaicin was registered as a small electron density, reflecting neither its chemical structure nor specific ligand-channel interactions—important details required for mechanistic understanding. We obtained the missing atomic-level details by iterative computation and confirmed them by systematic site-specific functional tests. We observed that the bound capsaicin takes a 'tail-up, head-down' configuration. The vanillyl and amide groups form specific interactions to anchor its bound position, while the aliphatic tail may sample a range of conformations, making it invisible in cryo-EM images. Capsaicin stabilizes TRPV1's open state by 'pull-and-contact' interactions between the vanillyl group and the S4-S5 linker. Our study provides a structural mechanism for the agonistic function of capsaicin and its analogs, and demonstrates an effective approach to obtain atomic-level information from cryo-EM structures.






Similar content being viewed by others
References
Nilius, B. & Appendino, G. Spices: the savory and beneficial science of pungency. Rev. Physiol. Biochem. Pharmacol. 164, 1–76 (2013).
Jordt, S.E. & Julius, D. Molecular basis for species-specific sensitivity to “hot” chili peppers. Cell 108, 421–430 (2002).
Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 29, 355–384 (2013).
Ludy, M.J., Moore, G.E. & Mattes, R.D. The effects of capsaicin and capsiate on energy balance: critical review and meta-analyses of studies in humans. Chem. Senses 37, 103–121 (2012).
Gavva, N.R. et al. The vanilloid receptor TRPV1 is tonically activated in vivo and involved in body temperature regulation. J. Neurosci. 27, 3366–3374 (2007).
Díaz-Laviada, I. & Rodriguez-Henche, N. The potential antitumor effects of capsaicin. Prog. Drug Res. 68, 181–208 (2014).
Caterina et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 288, 306–313 (2000).
Caterina, M.J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 (1997).
Zheng, J. Molecular mechanism of TRP channels. Compr. Physiol. 3, 221–242 (2013).
Gavva, N.R. et al. Molecular determinants of vanilloid sensitivity in TRPV1. J. Biol. Chem. 279, 20283–20295 (2004).
Liao, M., Cao, E., Julius, D. & Cheng, Y. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504, 107–112 (2013).
Cao, E., Liao, M., Cheng, Y. & Julius, D. TRPV1 structures in distinct conformations reveal activation mechanisms. Nature 504, 113–118 (2013).
Salazar, H. et al. Structural determinants of gating in the TRPV1 channel. Nat. Struct. Mol. Biol. 16, 704–710 (2009).
Zheng, J. & Ma, L. Structure and function of the thermoTRP channel pore. Curr. Top. Membr. 74, 233–257 (2014).
Yarov-Yarovoy, V., Schonbrun, J. & Baker, D. Multipass membrane protein structure prediction using Rosetta. Proteins 62, 1010–1025 (2006).
Fleishman, S.J. et al. RosettaScripts: a scripting language interface to the Rosetta macromolecular modeling suite. PLoS ONE 6, e20161 (2011).
Davis, I.W. & Baker, D. RosettaLigand docking with full ligand and receptor flexibility. J. Mol. Biol. 385, 381–392 (2009).
Lemmon, G. & Meiler, J. Rosetta Ligand docking with flexible XML protocols. Methods Mol. Biol. 819, 143–155 (2012).
Bronowska, A.K. in Thermodynamics—Interaction Studies—Solids, Liquids and Gases (ed. Juan Carlos Moreno-Pirajan) Ch. 1, 5–12 (InTech, Rijeka, 2011).
Jones, J.E. On the determination of molecular fields. II. From the equation of state of a gas. Proc. R. Soc. Lond. A 106, 463–477 (1924).
Lalatonne, Y., Richardi, J. & Pileni, M.P. Van der Waals versus dipolar forces controlling mesoscopic organizations of magnetic nanocrystals. Nat. Mater. 3, 121–125 (2004).
Horovitz, A. & Fersht, A.R. Strategy for analysing the co-operativity of intramolecular interactions in peptides and proteins. J. Mol. Biol. 214, 613–617 (1990).
Hidalgo, P. & MacKinnon, R. Revealing the architecture of a K+ channel pore through mutant cycles with a peptide inhibitor. Science 268, 307–310 (1995).
Ranganathan, R., Lewis, J.H. & MacKinnon, R. Spatial localization of the K+ channel selectivity filter by mutant cycle-based structure analysis. Neuron 16, 131–139 (1996).
Carter, P.J., Winter, G., Wilkinson, A.J. & Fersht, A.R. The use of double mutants to detect structural changes in the active site of the tyrosyl-tRNA synthetase (Bacillus stearothermophilus). Cell 38, 835–840 (1984).
Schreiber, G. & Fersht, A.R. Energetics of protein-protein interactions: analysis of the barnase-barstar interface by single mutations and double mutant cycles. J. Mol. Biol. 248, 478–486 (1995).
Kobata, K., Todo, T., Yazawa, S., Iwai, K. & Watanabe, T. Novel capsaicinoid-like substances, capsiate and dihydrocapsiate, from the fruits of a nonpungent cultivar, CH-19 Sweet, of pepper (Capsicum annuum L.). J. Agric. Food Chem. 46, 1695–1697 (1998).
Bevan, S. et al. Capsazepine: a competitive antagonist of the sensory neurone excitant capsaicin. Br. J. Pharmacol. 107, 544–552 (1992).
Colquhoun, D. Binding, gating, affinity and efficacy. Br. J. Pharmacol. 125, 923–947 (1998).
Franks, F. Water: A Matrix of Life 2nd edn. (The Royal Society of Chemistry, Cambridge, 2000).
Sheu, S.Y., Yang, D.Y., Selzle, H.L. & Schlag, E.W. Energetics of hydrogen bonds in peptides. Proc. Natl. Acad. Sci. USA 100, 12683–12687 (2003).
Lu, Z., Klem, A.M. & Ramu, Y. Coupling between voltage sensors and activation gate in voltage-gated K+ channels. J. Gen. Physiol. 120, 663–676 (2002).
Yarov-Yarovoy, V. et al. Structural basis for gating charge movement in the voltage sensor of a sodium channel. Proc. Natl. Acad. Sci. USA 109, E93–E102 (2012).
Boukalova, S., Marsakova, L., Teisinger, J. & Vlachova, V. Conserved residues within the putative S4–S5 region serve distinct functions among thermosensitive vanilloid transient receptor potential (TRPV) channels. J. Biol. Chem. 285, 41455–41462 (2010).
Voets, T., Owsianik, G., Janssens, A., Talavera, K. & Nilius, B. TRPM8 voltage sensor mutants reveal a mechanism for integrating thermal and chemical stimuli. Nat. Chem. Biol. 3, 174–182 (2007).
Zheng, W. & Auerbach, A. Decrypting the sequence of structural events during the gating transition of pentameric ligand-gated ion channels based on an interpolated elastic network model. PLoS Comput. Biol. 7, e1001046 (2011).
Puljung, M.C., DeBerg, H.A., Zagotta, W.N. & Stoll, S. Double electron-electron resonance reveals cAMP-induced conformational change in HCN channels. Proc. Natl. Acad. Sci. USA 111, 9816–9821 (2014).
Tekpinar, M. & Zheng, W. Predicting order of conformational changes during protein conformational transitions using an interpolated elastic network model. Proteins 78, 2469–2481 (2010).
Steinberg, X., Lespay-Rebolledo, C. & Brauchi, S. A structural view of ligand-dependent activation in thermoTRP channels. Front. Physiol. 5, 171 (2014).
Szallasi, A. & Blumberg, P.M. Resiniferatoxin, a phorbol-related diterpene, acts as an ultrapotent analog of capsaicin, the irritant constituent in red pepper. Neuroscience 30, 515–520 (1989).
Thomas, K.C. et al. Structure-activity relationship of capsaicin analogs and transient receptor potential vanilloid 1-mediated human lung epithelial cell toxicity. J. Pharmacol. Exp. Ther. 337, 400–410 (2011).
Wahl, P., Foged, C., Tullin, S. & Thomsen, C. Iodo-resiniferatoxin, a new potent vanilloid receptor antagonist. Mol. Pharmacol. 59, 9–15 (2001).
Appendino, G. et al. Halogenation of a capsaicin analogue leads to novel vanilloid TRPV1 receptor antagonists. Br. J. Pharmacol. 139, 1417–1424 (2003).
Appendino, G. et al. The taming of capsaicin. Reversal of the vanilloid activity of N-acylvanillamines by aromatic iodination. J. Med. Chem. 48, 4663–4669 (2005).
Liao, M., Cao, E., Julius, D. & Cheng, Y. Single particle electron cryo-microscopy of a mammalian ion channel. Curr. Opin. Struct. Biol. 27, 1–7 (2014).
Alexander, N.S. et al. Energetic analysis of the rhodopsin–G-protein complex links the α5 helix to GDP release. Nat. Struct. Mol. Biol. 21, 56–63 (2014).
Jensen, M.Ø. et al. Mechanism of voltage gating in potassium channels. Science 336, 229–233 (2012).
Lindert, S. et al. EM-fold: de novo atomic-detail protein structure determination from medium-resolution density maps. Structure 20, 464–478 (2012).
Cheng, W., Yang, F., Takanishi, C.L. & Zheng, J. Thermosensitive TRPV channel subunits co-assemble into heteromeric channels with intermediate conductance and gating properties. J. Gen. Physiol. 129, 191–207 (2007).
Barbero, G.F. et al. Application of Hansch's model to capsaicinoids and capsinoids: a study using the quantitative structure-activity relationship. A novel method for the synthesis of capsinoids. J. Agric. Food Chem. 58, 3342–3349 (2010).
Gannett, P.M. et al. Capsaicinoids-their separation, synthesis, and mutagenicity. J. Org. Chem. 53, 1064–1071 (1988).
Challis, A.A.L. & Clemo, G.R. Some amines and amides derived from vanillin. J. Chem. Soc. 613–618 (1947).
Davis, I.W., Raha, K., Head, M.S. & Baker, D. Blind docking of pharmaceutically relevant compounds using RosettaLigand. Protein Sci. 18, 1998–2002 (2009).
Meiler, J. & Baker, D. ROSETTALIGAND: protein-small molecule docking with full side-chain flexibility. Proteins 65, 538–548 (2006).
Yarov-Yarovoy, V., Baker, D. & Catterall, W.A. Voltage sensor conformations in the open and closed states in ROSETTA structural models of K+ channels. Proc. Natl. Acad. Sci. USA 103, 7292–7297 (2006).
Hawkins, P.C., Skillman, A.G., Warren, G.L., Ellingson, B.A. & Stahl, M.T. Conformer generation with OMEGA: algorithm and validation using high quality structures from the Protein Databank and Cambridge Structural Database. J. Chem. Inf. Model. 50, 572–584 (2010).
Pettersen, E.F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Qin, F. Restoration of single-channel currents using the segmental k-means method based on hidden Markov modeling. Biophys. J. 86, 1488–1501 (2004).
Sakmann, B. & Neher, E. Single-Channel Recording 2nd edn. (Springer, 2009).
Sigworth, F.J. The variance of sodium current fluctuations at the node of Ranvier. J. Physiol. (Lond.) 307, 97–129 (1980).
Yang, F., Cui, Y., Wang, K. & Zheng, J. Thermosensitive TRP channel pore turret is part of the temperature activation pathway. Proc. Natl. Acad. Sci. USA 107, 7083–7088 (2010).
Acknowledgements
We thank members of the Zheng laboratory for assistance and constructive discussion. This work was supported by funding from National Institutes of Health (R01NS072377) and National Natural Science Foundation of China (31328011) to J.Z., National Natural Science Foundation of China (31328011, 81301720) to W.C., National Basic Research Program of China (2013CB910204), National Natural Science Foundation of China (81371302), China Scholarship Council to W.Y. and American Heart Association (14POST19820027) to F.Y.
Author information
Authors and Affiliations
Contributions
F.Y. and X.X. conducted most of experiments including molecular docking, patch-clamp recordings and data analysis; W.Y., P.Y., Z.S. and W.C. synthesized and analyzed capsaicin analogs; V.Y.-Y. supervised molecular docking and revised the manuscript; J.Z. and F.Y. prepared the manuscript; J.Z. conceived and supervised the project and participated in data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1-11 and Supplementary Tables 1-4. (PDF 7671 kb)
Supplementary Notes
Supplementary Notes (DOCX 3147 kb)
Rights and permissions
About this article
Cite this article
Yang, F., Xiao, X., Cheng, W. et al. Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel. Nat Chem Biol 11, 518–524 (2015). https://doi.org/10.1038/nchembio.1835
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1835
- Springer Nature America, Inc.
This article is cited by
-
Structure-guided peptide engineering of a positive allosteric modulator targeting the outer pore of TRPV1 for long-lasting analgesia
Nature Communications (2023)
-
Structural mechanisms of TRPV2 modulation by endogenous and exogenous ligands
Nature Chemical Biology (2023)
-
Cannabinoid non-cannabidiol site modulation of TRPV2 structure and function
Nature Communications (2022)
-
Safety, Pharmacokinetic, and Pharmacodynamic Study of a Sublingual Formula for the Treatment of Vasovagal Syncope
Drugs in R&D (2022)
-
A capsaicinoid-based soft drug, AG1529, for attenuating TRPV1-mediated histaminergic and inflammatory sensory neuron excitability
Scientific Reports (2021)