Abstract
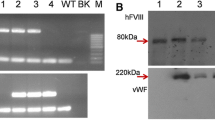
Deficiency or abnormality of coagulation factor VIII (FVIII) causes a bleeding disorder called hemophilia A. Treatment involves FVIII concentrates prepared from pooled human plasma or recombinant FVIII (rFVIII) prepared from mammalian cell culture. The cost of highly purified FVIII or rFVIII is a major factor in hemophilia therapy and restricts prophylaxis. We have sought to generate a new source of rFVIII by targeting expression of the human FVIII cDNA to the mammary gland of transgenic pigs using the regulatory sequences of the mouse whey acidic protein gene. The identity of processed heterodimeric rFVIII was confirmed using specific antibodies, by thrombin digestion and activity assays. The secretion of as much as 2.7 μg/ml of rFVIII in milk was over tenfold higher than in normal plasma. Up to 0.62 U/ml of rFVIII was detected in an assay in which rFVIII restored normal clotting activity to FVIII-deficient human plasma.
Similar content being viewed by others
References
Vehar, G.A., Keyt, B., Eaton, D., Rodriguez, H., O'Brien, D.P., Rotblat, F. et al. 1984. Structure of human factor VIII. Nature 312: 337 342.
Toole, J.J., Knopf, J.L. and Wozney, J.M. 1984. Molecular cloning of a cDNA encoding human antihemophilic factor. Nature 312: 342 347.
Wood, W.I., Gapon, D.J., Simonsen, C.C., Eaton, D.L., Gitschier, J., Keyt, B. et al. 1984. Expression of active human factor VIII from recombinant DNA clones. Nature 312: 330 337.
Hoyer, L.W. 1993. Characterization of dysfunctional factor VIII molecules. Methods EnzymoL. 222: 169 176.
Weiss, H.J., Sussman, I.I. and Hoyer, L.W. 1977. Stabilization of factor VI11 in plasma by the von Willebrand factor. Studies on posttransfusion and dissociated factor VIM and in patients with von Willebrahd's disease. J. Clin. Invest. 60: 390 404.
Ehmann, W.C., Eyster, M.E., Aledort, L.M. and Goedert, J.J. 1995. Causes of death in hemophilia. Nature 378: 124.
Gitschier, J., Wood, W.I., Goralka, T.M., Wion, K.L., Chen, E.Y., Eaton, D.H. et al. 1984. Characterization of the human factor VIII gene. Nature 312: 326 330.
Eaton, D.L., Wood, W.I., Eaton, D., Hass, P.E., Hollingshead, P., Wion, K., Mather, et al. 1986. Construction and characterization of an active factor VIII variant lacking the central one-third of the molecule. Biochem. 25: 8343 8347.
Toole, J.J., Pittman, D.D., Orr, E.C., Murtha, P., Wasley, L.C. and Kaufman, R.J. 1986. A large region (˜95 kD) of human factor VIII is dispensable for in vitro procoagulant activity.Proc. Natl. Acad. Sci. USA 83: 5939 5942.
Kaufman, R.J., Wasley, L.C. and Dorner, A.J. 1988. Synthesis, processing and secretion of recombinant human factor VIII expressed in mammalian cells. J. Biol. Chem. 263: 6352 6362.
Kaufman, R.J., Wasley, L.C., Davies, M.V., Wise, R.J., Israel, D.I. and Dorner, A.J. 1989. Effect of von Willebrand factor coexpression on the synthesis and secretion of factor VIII in Chinese hamster ovary cells. Mol. Cell Biol. 9: 1233 1242.
Lynch, C.M., Israel, D.I., Kaufman, R.J. and Miller, A.D. 1993. Sequences in the coding region of clotting factor VIII act as dominant inhibitors of RNA accumulation and protein production. Hum. Gene Ther. 4: 259 272.
Koeberl, D.D., Halbert, C.L., Krumm, A. and Miller, A.D. 1995. Sequences within the coding regions of clotting factor VIII and CFTR block transcriptional elongation. Hum. Gene Ther. 6: 469 479.
Hoeben, R.C., Fallaux, F.J., Cramer, S.J. van den Wollenberg, D.J.M., van Ormondt, H., Briet, E. et al. 1995. Expression of the blood-clotting factor VIII cDNA is repressed by a transcriptional silencer located in its coding region. Blood 86: 2447 2454.
Koedam, J.A., Meijers, J.C.M., Sixma, J.J. and Bouma, B.N. 1988. Inactivation of human factor VIII by activated protein C. Cofactor activity of protein S and protective effect of von Willebrand factor. J. Clin. Invest. 82: 1236 1243.
Lubon, H., Paleyanda, R.K., Velander, W.H. and Drohan, W.N. 1996. Blood proteins from transgenic animal bioreactors. Transfus. Med. Revs. X: 131 143.
Campbell, S.M., Rosen, J.M., Hennighausen, L.G., Strech-Jurk, U. and Sippel, A.E. 1982. Comparison of the whey acidic protein genes of the rat and mouse. Nucleic Acids Res. 12: 8685 8697.
Fulcher, C.A. and Zimmerman, T.S. 1982. Characterization of the human factor VIII procoagulant protein with a heterologous precipitating antibody. Proc. Natl. Acad. Sci. USA 79: 1648 1652.
Eaton, D., Rodriguez, H. and Vehar, G.A. 1986. Proteolytic processing of human factor VIII. Correlation of specific cleavages by thrombin, factor Xa and activated protein C with activation and inactivation of factor VIII coagulant activity. Biochem. 25: 505 512.
Fulcher, C.A., Roberts, J.R. and Zimmerman, T.S. 1983. Thrombin proteolysis of purified factor VIII. Correlation of activation with generation of a specific polypeptide. Blood 61: 807 811.
Pittman, D.D. and Kaufman, R.J. 1988. Proteolytic requirements for thrombin activation of ariti-hemophilic factor (factor VIII). Proc. Natl. Acad. Sci. USA 8: 2429 2433.
Rosen, S. 1984. Assay of factor VIII:C with a chromogenic substrate. Scand. J. Haematol. 33 (suppl. 40): 139 145.
Over, J. 1984. Methodology of the one-stage assay of factor VIII (VIII:C). Scand. J. Haematol. 41: 13 24.
Clark, A.J., Bessos, H., Bishop, J.O., Brown, P., Harris, S., Lathe, R. et al. 1989. Expression of human anti-hemophilic factor IX in the milk of transgenic sheep. Bio/Technology 7: 487 492.
Lee, T., Drohan, W.N. and Lubon, H. 1995. Proteolytic processing of human protein C in swine mammary gland. J. Biochem. 118: 81 87.
Yarus, S., Greenberg, N.M., Wei, Y., Whitsett, J.A., Weaver, T.E. and ROsen, J.M. 1997. Secretion of unprocessed human surfactant protein B in the milk of transgenic mice. Transgenic Res. 6: 51 57.
Drohan, W.N., Zhang, D.-W., Paleyanda, R.K., Chang, R., Wroble, M., Velander, W. et al. 1994. Inefficient processing of human protein C in the mouse mammary gland. Transgenic Res. 3: 355 364.
Denman, J., Hayes, M., O'Day, C., Edmunds, T., Bartlett, C., Hirani, S. et al. 1991. Transgenic expression of a variant of human tissue-type plasminogen activator in goat milk: Purification and characterization Of the recombinant enzyme. Bio/Technology 9: 839 >843.
Mikkelsen, T.R., Brandt, J., Larsen, H.J., Larsen, B.B., Poulsen, K., Ingerslev, J. et al. 1992. Tissue-specific expression in the salivary glands of transgenic mice. Nucl. Acids Res. 20: 2249 2255.
Pittius, C.W., Hennighausen, L., Lee, E., Westphal, H., Nicol, E., Vitale, J. et al. 1988. A milk protein gene promoter directs the expression of human tissue plasminogen activator cDNA to the mammary gland in transgenic mice. Proc. Natl. Acad. Sci. USA 85: 5874 5878.
Reddy, V.B., Vitale, J., Wei, C., Montoya-Zavala, M., Stice, S.L., Balise, J. et al. 1991. Expression of human growth hormone in the milk of transgenic mice. Animal Biotechnoi. 2: 15 29.
Günzburg, W.H., Salmons, B., Zimmermann, B., Muller, M., Erfle, V. and Brem, G. 1991. A mammary-specific promoter directs expression of growth hormone not Only to the mammary gland, but also to Bergman glia cells in transgenic mice.Mol. Endocrinol. 5: 123 133.
Wen, J., Kawamata, Y., Tojo, H., Tanaka, S. and Tachi, C. 1995. Expression of whey acidic protein (WAP) genes in tissues other than the mammary gland in normal and transgenic mice expressing mWAP/hGH fusion gene. Mol. Reprod. Dev. 41: 399 406.
Wall, R.J., Pursel, V.G., Shamay, A., McKnight, R.A., Pittius, C.W. and Hennighausen, L. 1991. High-level synthesis of a heterologous milk protein in the mammary glands of transgenic swine. Proc. Natl. Acad. Sci. USA 88: 1696 1700.
Drohan, W.N., Wilkins, T.D., Latimer, E., Zhou, D., Velander, W., Lee, T.K. et al. 1994. A scalable method for the purification of recombinant human protein C from the milk of transgenic swine, pp. 501 507 in Advances in bioprocess engineering. Galindo, E. and Ramirez, O.T. (eds.) Kluwer Academic Publishers,.Dordrecht, The Netherlands.
Velander, W.H., Johnson, J.L., Page, R.L., Russell, C.G., Subramanian, A., Wilkins, T.D. et al. 1992. High-level expression of a heterologous protein in the milk of transgenic swine using the cDNA encoding human protein C. Proc. Natl. Acad. Sci. USA 89: 12003 12007.
Wall, R.J., Pursel, V.G., Hammer, R.E. and Brinster, R.L. 1985. Development of porcine ova that were centrifuged to permit visualization of pronuclei and nuclei. Biol. Reprod. 32: 645 651.
Fulcher, C.A., Roberts, J.R., Holland, L.Z. and Zimmerman, T.S. 1985. Human factor VIII procoagulant protein. Monoclonal antibodies define precursor-product relationships and functional epitopes. J. Clin. Invest. 76: 117 124.
Lubin, I.M., Healey, J.F., Scandelia, D., Runge, M.S. and Lollar, P. 1994. Elimination of a major inhibitor epitope in Factor VIII. J. Biol. Chem. 269: 8639 8641.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paleyanda, R., Velander, W., Lee, T. et al. Transgenic pigs produce functional human factor VIII in milk. Nat Biotechnol 15, 971–975 (1997). https://doi.org/10.1038/nbt1097-971
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1097-971
- Springer Nature America, Inc.
This article is cited by
-
Expression of Active Fluorophore Proteins in the Milk of Transgenic Pigs Bypassing the Secretory Pathway
Scientific Reports (2016)
-
Genome editing revolutionize the creation of genetically modified pigs for modeling human diseases
Human Genetics (2016)
-
Establishment of a novel, eco-friendly transgenic pig model using porcine pancreatic amylase promoter-driven fungal cellulase transgenes
Transgenic Research (2015)
-
Efficient stabilization of recombinant human coagulation factor VIII in the milk of transgenic mice using hFVIII and vWF co-expression vector transduction
Biotechnology Letters (2015)
-
A bioinformatic evaluation of potential allergenicity of 85 candidate genes in transgenic organisms
Chinese Science Bulletin (2012)