Abstract
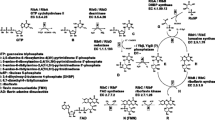
The pentose phosphate pathway and the pyruvate shunt were identified as major pathways of glucose catabolism in a recombinant, riboflavin-producing Bacillus subtilis strain. Reactions connecting the tricarboxylic acid cycle and glycolysis, catalyzed by the malic enzyme and phosphoenolpyruvate carboxykinase, consume up to 23% of the metabolized glucose. These are examples of important fluxes that can be accessed explicitly using a novel analysis based on synergistic application of flux balancing and recently introduced techniques of fractional 13C-labeling and two-dimensional nuclear magnetic resonance spectroscopy. The overall flux distribution also suggests that B. subtilis metabolism has an unusually high capacity for the reoxidation of NADPH. Under the conditions investigated, riboflavin formation in B. subtilis is limited by the fluxes through the biosynthetic rather than the central carbon pathways, which suggests a focus for future metabolic engineering of this system.
Similar content being viewed by others
References
Bailey, J.E. 1991. Toward a science of metabolic engineering. Science 252: 1668–1675.
Stephanopoulos, G. and Sinskey, A.J. 1993. Metabolic engineering—methodologies and future prospects. Trends. Biotechnol. 11: 392–396.
Vallino, J.J. and Stephanopoulos, G. 1993. Metabolic flux distribution in Corynebacterium glutamicum during growth and lysine overproduction. Biotechnol. Bioeng. 41: 633–646.
van Gulik, W.M. and Heijnen, J.J. 1995. A metabolic network stoichiometry analysis of microbial growth and product formation. Biotechnol. Bioeng. 48: 681–698.
Eggeling, L., Sahm, H., and de Graaf, A.A 1996. Quantification and directing metabolite flux: application to amino acid overproduction. Adv. Biochem. Eng. 54 1–30.
Varma, A. and Palsson, B.O. 1994. Metabolic flux balancing: basic concepts, scientific, and practical use. Bio/Technology 12: 994–998.
Galazzo, J.L. and Bailey, J.E. 1990. Fermentation pathway kinetics and metabolic flux control in suspended and immobilized Saccharomyces cerevisiae. Enzyme Microb. Technol. 12: 162–172.
Hatzimanikatis, V., Floudas, C.A. and Bailey, J.E. 1995. Analysis and design of metabolic reaction networks via mixed-integer linear optimization. AlChE J. 42: 1277–1292.
Schlosser, P.M., Riedy, G. and Bailey, J.E. 1994. Ethanol production in baker's yeast: application of experimental perturbation techniques for model development and resultant changes in flux control analysis. Biotechnol. Prog. 10: 141–154.
Holms, W.H. 1986. The central metabolic pathways of Escherichia coli: relationship between flux and control at a branch point, efficiency of conversion to biomass, and excretion of acetate. Curr. Top. Cell. Regul. 28: 69–105.
Sauer, U., Hatzimanikatis, V., Hohmann, H.-R., Manneberg, M., van Loon, A.P.G.M., and Bailey, J.E. 1996. Physiology and metabolic fluxes of wild-type and riboflavin-producing Bacillus subtilis. Appl. Environ. Microbiol. 62: 3687–3696.
Tsai, P., Hatzimanikatis, V., and Bailey, J.E. 1996. Effect of Vitreoscilla hemoglobin dosage on microaerobic Escherichia coli carbon and energy metabolism. Biotechnol. Bioeng. 49: 139–150.
Varma, A., Boesch, B.W. and Palsson, B.O. 1993. Biochemical production capabilities of Escherichia coli. Biotechnol. Bioeng. 42: 59–73.
Goel, A., Ferrance, J., Jeong, J. and Ataa, M.M. 1993. Analysis of metabolic fluxes in batch and continuous cultures of Bacillus subtilis. Biotechnol. Bioeng. 42: 686–696.
Walsh, K. and J., Koshland, D.E. 1984. Determination of flux through the branch point of two metabolic cycles. J. Biol. Chem. 259: 9646–9654.
Ishino, S., Shimomura-Nishimuta, J., Yamaguchi, K., Shirahata, K. and Araki, K. 1991. 13C nuclear magnetic resonance studies of glucose metabolism in L-glutamic acid and L-lysine fermentation by Corynebacterium glutamicum. J. Gen. Appl. Microbiol. 37: 157–165.
Portais, J.-C., Schuster, R., Merle, M. and Canioni, P. 1993. Metabolic flux determination in C6 glioma cells using carbon-13 distribution upon [1-13C]glucose incubation. Eur. J. Biochem. 217: 457–468.
Rollin, C., Morgant, V., Guyonvarch, A. and Guerquin-Kern, J.-L. 1995. 13C-NMR studies of Corynebacterium melassecola metabolic pathways. Eur. J. Biochem. 227: 488–493.
Marx, A., de Graaf, A.A., Wiechert, W., Eggeling, L., and Sahm, H. 1996. Determination of the fluxes in the central metabolism of Corynebacterium glutamicum by nuclear magnetic resonance spectroscopy combined with metabolite balancing. Biotechnol. Bioeng. 49: 111–129.
Neri, D., Szyperski, T., Otting, G., Senn, H., and Wuthrich, K. 1989. Stereospecific nuclear magnetic resonance assignments of the methyl groups of valine and leucine in the DMA-binding domain of the 434-repressor by biosynthetically directed fractional 13C labeling. Biochemistry 28: 7510–7516.
Senn, H., Werner, B., Messerle, B.A., Weber, C., Traber, R. and Wuthrich, K. 1989. Stereospecific assignment of the methyl 1H-NMR lines of valine and isoleucine in polypeptides by nonrandom 13C labeling. FEBS Lett. 249: 113–118.
Wüthrich, K., Szyperski, T., Leiting, B., and Otting, G. 1992. Biosynthetic pathways of the common proteinogenic amino acids investigated by fractional 13C labeling and NMR spectroscopy, pp. 41–48 in Frontiers and new horizons in amino acid research. Takai, K. (ed.). Elsevier, Amsterdam.
Szyperski, T. 1995. Biosynthetically directed fractional 13C-labeling of proteinogenic amino acids. An efficient analytical tool to investigate intermediary metabolism. Eur. J. Biochem. 232: 433–448.
Szyperski, T., Bailey, J.E., and Wuthrich, K. 1996. Detecting and dissecting metabolic fluxes using biosynthetic fractional 13C-labeling and two-dimensional NMR spectroscopy. Trends Biotechnol. 14: 453–459.
Perkins, J.B., Pero, J.G. and Sloma, A. 1991. Riboflavin overproducing strains of bacteria. Europ. Pat. Appl. 0 405 370 A1.
Diesterhaft, M.D. and Freese, E. 1973. Role of pyruvate carboxylase, phospho-enolpyruvate carboxykinase, and malic enzyme during growth and sporulation of Bacillus subtilis. J. Biol. Chem. 248: 6062–6070.
Freese, E. and Fortnagel, U. 1969. Growth and sporulation of Bacillus subtilis mutants blocked in the pyruvate dehydrogenase complex. J. Bacteriol. 99: 745–756.
Volk, R. and Bacher, A. 1990. Studies on the 4-carbon precursor in the biosynthesis of riboflavin. J. Biol. Chem. 265: 19479–19485.
Wood, T.D. 1985. The pentose phosphate pathway. Academic Press Inc., Orlando, FL.
Sasajima, K. and Yoneda, M. 1984. Production of pentoses by micro-organisms. Biotechnol. Genet. Eng. Rev. 2: 175–213.
Gottschalk, G. 1986. Bacterial metabolism, 2nd ed. Springer-Verlag, New York.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sauer, U., Hatzimanikatis, V., Bailey, J. et al. Metabolic fluxes in riboflavin-producing Bacillus subtilis. Nat Biotechnol 15, 448–452 (1997). https://doi.org/10.1038/nbt0597-448
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0597-448
- Springer Nature America, Inc.
This article is cited by
-
Rational design of GDP‑d‑mannose mannosyl hydrolase for microbial l‑fucose production
Microbial Cell Factories (2023)
-
Genomic, transcriptomic, and metabolic characterizations of Escherichia coli adapted to branched-chain higher alcohol tolerance
Applied Microbiology and Biotechnology (2020)
-
Effects of gltA and arcA Mutations on Biomass and 1,3-Propanediol Production in Klebsiella pneumoniae
Biotechnology and Bioprocess Engineering (2019)
-
PFA toolbox: a MATLAB tool for Metabolic Flux Analysis
BMC Systems Biology (2016)
-
Biotechnology of riboflavin
Applied Microbiology and Biotechnology (2016)