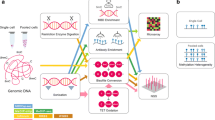
Abstract
Studies of epigenetic modifications would benefit from improved methods for high-throughput methylation profiling. We introduce two complementary approaches that use next-generation sequencing technology to detect cytosine methylation. In the first method, we designed ∼10,000 bisulfite padlock probes to profile ∼7,000 CpG locations distributed over the ENCODE pilot project regions and applied them to human B-lymphocytes, fibroblasts and induced pluripotent stem cells. This unbiased choice of targets takes advantage of existing expression and chromatin immunoprecipitation data and enabled us to observe a pattern of low promoter methylation and high gene-body methylation in highly expressed genes. The second method, methyl-sensitive cut counting, generated nontargeted genome-scale data for ∼1.4 million HpaII sites in the DNA of B-lymphocytes and confirmed that gene-body methylation in highly expressed genes is a consistent phenomenon throughout the human genome. Our observations highlight the usefulness of techniques that are not inherently or intentionally biased towards particular subsets like CpG islands or promoter regions.





Similar content being viewed by others
Accession codes
Change history
08 May 2009
In the version of this article initially published, the second affilliation for Yuan Gao was omitted: Department of Computer Science, Virginia Commonwealth University, Richmond, Virginia, USA. The affiliation has been added to the HTML and PDF versions of the article.
References
Suzuki, M.M. & Bird, A. DNA methylation landscapes: provocative insights from epigenomics. Nat. Rev. Genet. 9, 465–476 (2008).
Goll, M.G. & Bestor, T.H. Eukaryotic cytosine methyltransferases. Annu. Rev. Biochem. 74, 481–514 (2005).
Feinberg, A.P. & Tycko, B. The history of cancer epigenetics. Nat. Rev. Cancer 4, 143–153 (2004).
Jiang, Y.H., Bressler, J. & Beaudet, A.L. Epigenetics and human disease. Annu. Rev. Genomics Hum. Genet. 5, 479–510 (2004).
Clark, S.J., Harrison, J., Paul, C.L. & Frommer, M. High sensitivity mapping of methylated cytosines. Nucleic Acids Res. 22, 2990–2997 (1994).
Bird, A.P. & Southern, E.M. Use of restriction enzymes to study eukaryotic DNA methylation: I. The methylation pattern in ribosomal DNA from Xenopus laevis. J. Mol. Biol. 118, 27–47 (1978).
Keshet, I. et al. Evidence for an instructive mechanism of de novo methylation in cancer cells. Nat. Genet. 38, 149–153 (2006).
Cross, S.H., Charlton, J.A., Nan, X. & Bird, A.P. Purification of CpG islands using a methylated DNA binding column. Nat. Genet. 6, 236–244 (1994).
Rauch, T.A., Wu, X., Zhong, X., Riggs, A.D. & Pfeifer, G.P. A human B cell methylome at 100-base pair resolution. Proc. Natl. Acad. Sci. USA 106, 671–678 (2009).
Hellman, A. & Chess, A. Gene body-specific methylation on the active X chromosome. Science 315, 1141–1143 (2007).
Khulan, B. et al. Comparative isoschizomer profiling of cytosine methylation: the HELP assay. Genome Res. 16, 1046–1055 (2006).
Zhang, X. et al. Genome-wide high-resolution mapping and functional analysis of DNA methylation in Arabidopsis. Cell 126, 1189–1201 (2006).
Zilberman, D., Gehring, M., Tran, R.K., Ballinger, T. & Henikoff, S. Genome-wide analysis of Arabidopsis thaliana DNA methylation uncovers an interdependence between methylation and transcription. Nat. Genet. 39, 61–69 (2007).
Bibikova, M. et al. High-throughput DNA methylation profiling using universal bead arrays. Genome Res. 16, 383–393 (2006).
Irizarry, R.A. et al. The human colon cancer methylome shows similar hypo- and hypermethylation at conserved tissue-specific CpG island shores. Nat. Genet. 41, 178–186 (2009).
Meissner, A. et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 454, 766–770 (2008).
Lister, R. et al. Highly integrated single-base resolution maps of the epigenome in Arabidopsis. Cell 133, 523–536 (2008).
Cokus, S.J. et al. Shotgun bisulphite sequencing of the Arabidopsis genome reveals DNA methylation patterning. Nature 452, 215–219 (2008).
Bentley, D.R. et al. Accurate whole human genome sequencing using reversible terminator chemistry. Nature 456, 53–59 (2008).
Shendure, J. et al. Accurate multiplex polony sequencing of an evolved bacterial genome. Science 309, 1728–1732 (2005).
Birney, E. et al. Identification and analysis of functional elements in 1% of the human genome by the ENCODE pilot project. Nature 447, 799–816 (2007).
Shann, Y.J. et al. Genome-wide mapping and characterization of hypomethylated sites in human tissues and breast cancer cell lines. Genome Res. 18, 791–801 (2008).
Nilsson, M. et al. Padlock probes: circularizing oligonucleotides for localized DNA detection. Science 265, 2085–2088 (1994).
Hardenbol, P. et al. Multiplexed genotyping with sequence-tagged molecular inversion probes. Nat. Biotechnol. 21, 673–678 (2003).
Porreca, G.J. et al. Multiplex amplification of large sets of human exons. Nat. Methods 4, 931–936 (2007).
Deng, J. et al. Targeted bisulfite sequencing reveals changes in DNA methylation associated with nuclear reprogramming. Nat. Biotechnol. advance online publication, doi:10/1038/nbt.1530 (29 March 2009).
Eckhardt, F. et al. DNA methylation profiling of human chromosomes 6, 20 and 22. Nat. Genet. 38, 1378–1385 (2006).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Church, G.M. The personal genome project. Mol. Syst. Biol. 1, 0030 (2005.).
Appanah, R., Dickerson, D.R., Goyal, P., Groudine, M. & Lorincz, M.C. An unmethylated 3′ promoter-proximal region is required for efficient transcription initiation. PLoS Genet. 3, e27 (2007).
Buratowski, S. Transcription. Gene expression–where to start? Science 322, 1804–1805 (2008).
Weber, M. et al. Distribution, silencing potential and evolutionary impact of promoter DNA methylation in the human genome. Nat. Genet. 39, 457–466 (2007).
Shendure, J. & Ji, H. Next-generation DNA sequencing. Nat. Biotechnol. 26, 1135–1145 (2008).
Schuster, S.C. Next-generation sequencing transforms today's biology. Nat. Methods 5, 16–18 (2008).
Mardis, E.R. Next-generation DNA sequencing methods. Annu. Rev. Genomics Hum. Genet. 9, 387–402 (2008).
Kahvejian, A., Quackenbush, J. & Thompson, J.F. What would you do if you could sequence everything? Nat. Biotechnol. 26, 1125–1133 (2008).
Jones, P.A. The DNA methylation paradox. Trends Genet. 15, 34–37 (1999).
Illingworth, R. et al. A novel CpG island set identifies tissue-specific methylation at developmental gene loci. PLoS Biol. 6, e22 (2008).
Park, I.H., Lerou, P.H., Zhao, R., Huo, H. & Daley, G.Q. Generation of human-induced pluripotent stem cells. Nat. Protoc. 3, 1180–1186 (2008).
Ge, B. et al. Survey of allelic expression using EST mining. Genome Res. 15, 1584–1591 (2005).
Lewin, J., Schmitt, A.O., Adorjan, P., Hildmann, T. & Piepenbrock, C. Quantitative DNA methylation analysis based on four-dye trace data from direct sequencing of PCR amplificates. Bioinformatics 20, 3005–3012 (2004).
Acknowledgements
We thank Kun Zhang for discussion throughout this work; Wei Lin for help with computational design; Andrew Chess and Ravid Straussman for discussion and critical reading of the manuscript; Harvard Biopolymers Facility for Solexa sequencing; and Harvard Partners Center for Genetics and Genomics for gene expression profiling. This work was supported by the NHGRI-Centers of Excellence in Genomic Science (to G.M.C.).
Author information
Authors and Affiliations
Contributions
M.P.B., J.B.L. and G.M.C. conceived the study, designed the research and wrote the manuscript. M.P.B. and J.B.L. performed experiments and data analysis. Y.G. and B.X. carried out initial Solexa sequencing. J.-H.L. helped with culturing cell lines and isolating DNA/RNA. E.M.L. synthesized the padlock oligos. I.-H.P. and G.Q.D. generated the iPS cell lines.
Corresponding authors
Ethics declarations
Competing interests
E.M.L. is an employee of Agilent Technologies. G.M.C. is involved in eight next-generation sequencing companies. M.P.B., J.B.L. and G.M.C. are named as inventors on a patent application on technologies described in this article.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10; Supplementary Tables 2–5, 7,8 (PDF 6072 kb)
Supplementary Table 1
BSPP data (TXT 2113 kb)
Supplementary Table 6
MSCC data (TXT 60051 kb)
Rights and permissions
About this article
Cite this article
Ball, M., Li, J., Gao, Y. et al. Targeted and genome-scale strategies reveal gene-body methylation signatures in human cells. Nat Biotechnol 27, 361–368 (2009). https://doi.org/10.1038/nbt.1533
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1533
- Springer Nature America, Inc.
This article is cited by
-
Gbdmr: identifying differentially methylated CpG regions in the human genome via generalized beta regressions
BMC Bioinformatics (2024)
-
A role for SETD2 loss in tumorigenesis through DNA methylation dysregulation
BMC Cancer (2023)
-
The impact of spatial correlation on methylation entropy with application to mouse brain methylome
Epigenetics & Chromatin (2023)
-
The methylome and cell-free DNA: current applications in medicine and pediatric disease
Pediatric Research (2023)
-
Acute Transcriptomic and Epigenetic Alterations at T12 After Rat T10 Spinal Cord Contusive Injury
Molecular Neurobiology (2023)