Abstract
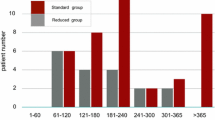
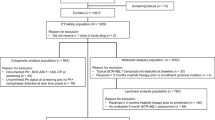
Nilotinib (Tasigna) is a potent and selective BCR-ABL inhibitor approved for use in patients with newly diagnosed chronic myeloid leukemia (CML) in chronic phase (CML-CP) and in patients with CML-CP and accelerated phase (CML-AP) who are resistant to or intolerant of imatinib. Patients with CML-AP (N=137) with at least 24 months of follow-up or who discontinued early were evaluated to determine the efficacy and tolerability of nilotinib. The majority (55%) of patients achieved a confirmed hematologic response, and 31% attained a confirmed complete hematologic response on nilotinib treatment. Overall, 32% of patients achieved major cytogenetic responses (MCyR), with most being complete cytogenetic responses. Responses were durable, with 66% of patients maintaining MCyR at 24 months. The estimated overall and progression-free survival rates at 24 months were 70% and 33%, respectively. Grade 3/4 neutropenia and thrombocytopenia were each observed in 42% of patients. Non-hematologic adverse events were mostly mild to moderate; the safety profile of nilotinib has not changed with longer follow-up. In all, 20 (15%) patients remained on study at data cutoff. In summary, nilotinib has a manageable safety profile, and can provide favorable long-term outcomes in the pretreated CML-AP patient population for whom treatment options are limited.


Similar content being viewed by others
References
Faderl S, Talpaz M, Estrov Z, O’Brien S, Kurzrock R, Kantarjian HM . The biology of chronic myeloid leukemia. N Engl J Med 1999; 341: 164–172.
Sawyers CL . Chronic myeloid leukemia. N Engl J Med 1999; 340: 1330–1340.
Sokal JE, Baccarani M, Russo D, Tura S . Staging and prognosis in chronic myelogenous leukemia. Semin Hematol 1988; 25: 49–61.
Kantarjian HM, Dixon D, Keating MJ, Talpaz M, Walters RS, McCredie KB et al. Characteristics of accelerated disease in chronic myelogenous leukemia. Cancer 1988; 61: 1441–1446.
Palandri F, Castagnetti F, Alimena G, Testoni N, Breccia M, Luatti S et al. The long-term durability of cytogenetic response in patients with accelerated phase chronic myeloid leukemia treated with imatinib 600 mg: the GIMEMA CML Working Party experience after a 7-year follow-up. Haematologica 2009; 94: 205–212.
Cortes JE, Talpaz M, O’Brien S, Faderl S, Garcia-Manero G, Ferrajoli A et al. Staging of chronic myeloid leukemia in the imatinib era: an evaluation of the World Health Organization proposal. Cancer 2006; 106: 1306–1315.
Gorre ME, Mohammed M, Ellwood K, Hsu N, Paquette R, Rao PN et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 2001; 293: 876–880.
Soverini S, Colarossi S, Gnani A, Rosti G, Castagnetti F, Poerio A et al. Contribution of ABL kinase domain mutations to imatinib resistance in different subsets of Philadelphia-positive patients: by the GIMEMA Working Party on chronic myeloid leukemia. Clin Cancer Res 2006; 12: 7374–7379.
Jabbour E, Kantarjian H, Jones D, Talpaz M, Bekele N, O’Brien S et al. Frequency and clinical significance of BCR-ABL mutations in patients with chronic myeloid leukemia treated with imatinib mesylate. Leukemia 2006; 20: 1767–1773.
Weisberg E, Manley PW, Breitenstein W, Brûggen J, Cowan-Jacob SW, Ray A et al. Characterization of AMN107, a selective inhibitor of native and mutant Bcr-Abl. Cancer Cell 2005; 7: 129–141.
Golemovic M, Verstovsek S, Giles F, Cortes J, Manshouri T, Manley PW et al. AMN107, a novel aminopyrimidine inhibitor of Bcr-Abl, has in vitro activity against imatinib-resistant chronic myeloid leukemia. Clin Cancer Res 2005; 11: 4941–4947.
Manley PW, Stiefl N, Cowan-Jacob SW, Kaufman S, Mestan J, Wartmann M et al. Structural resemblances and comparisons of the relative pharmacological properties of imatinib and nilotinib. Bioorg Med Chem 2010; 18: 6977–6986.
O’Hare T, Walters DK, Stoffregen EP, Jia T, Manley PW, Mestan J et al. In vitro activity of Bcr-Abl inhibitors AMN107 and BMS-354825 against clinically relevant imatinib-resistant Abl kinase domain mutants. Cancer Res 2005; 65: 4500–4505.
Weisberg E, Manley P, Mestan J, Cowan-Jacob S, Ray A, Griffin JD . AMN107 (nilotinib): a novel and selective inhibitor of BCR-ABL. Br J Cancer 2006; 94: 1765–1769.
Kantarjian H, Giles F, Wunderle L, Bhalla K, O’Brien S, Wassmann B et al. Nilotinib in imatinib-resistant CML and Philadelphia chromosome-positive ALL. N Engl J Med 2006; 354: 2542–2551.
le Coutre P, Ottmann OG, Giles F, Kim DW, Cortes J, Gattermann N et al. Nilotinib (formerly AMN107), a highly selective BCR-ABL tyrosine kinase inhibitor, is active in patients with imatinib-resistant or -intolerant accelerated-phase chronic myelogenous leukemia. Blood 2008; 111: 1834–1839.
Hughes T, Deininger M, Hochhaus A, Branford S, Radich J, Kaeda J et al. Monitoring CML patients responding to treatment with tyrosine kinase inhibitors: review and recommendations for harmonizing current methodology for detecting BCR-ABL transcripts and kinase domain mutations and for expressing results. Blood 2006; 108: 28–37.
Branford S, Cross NC, Hochhaus A, Radich J, Saglio G, Kaeda J et al. Rationale for the recommendations for harmonizing current methodology for detecting BCR-ABL transcripts in patients with chronic myeloid leukaemia. Leukemia 2006; 20: 1925–1930.
Muller MC, Cross NC, Erben P, Schenk T, Hanfstein B, Ernst T et al. Harmonization of molecular monitoring of CML therapy in Europe. Leukemia 2009; 23: 1957–1963.
Branford S, Fletcher L, Cross NC, Muller MC, Hochhaus A, Kim DW et al. Desirable performance characteristics for BCR-ABL measurement on an international reporting scale to allow consistent interpretation of individual patient response and comparison of response rates between clinical trials. Blood 2008; 112: 3330–3338.
Hughes T, Saglio G, Branford S, Soverini S, Kim DW, Muller MC et al. Impact of baseline BCR-ABL mutations on response to nilotinib in patients with chronic myeloid leukemia in chronic phase. J Clin Oncol 2009; 27: 4204–4210.
Talpaz M, Silver RT, Druker BJ, Goldman JM, Gambacorti-Passerini C, Guilhot F et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase 2 study. Blood 2002; 99: 1928–1937.
Silver RT, Talpaz M, Sawyers CL, Druker BJ, Hochhaus A, Schiffer CA et al. Four years of follow-up of 1027 patients with late chronic phase (L-CP), accelerated phase (AP), or blast crisis (BC) chronic myeloid leukemia (CML) treated with imatinib in three large phase II trials. Blood 2004; 104: 11a (abstract 23).
Silver RT, Cortes J, Waltzman R, Mone M, Kantarjian H . Sustained durability of responses and improved progression-free and overall survival with imatinib treatment for accelerated phase and blast crisis chronic myeloid leukemia: long-term follow-up of the STI571 0102 and 0109 trials. Haematologica 2009; 94: 743–744.
Vajpai N, Strauss A, Fendrich G, Cowan-Jacob SW, Manley PW, Grzesiek S et al. Solution conformations and dynamics of ABL kinase-inhibitor complexes determined by NMR substantiate the different binding modes of imatinib/nilotinib and dasatinib. J Biol Chem 2008; 283: 18292–18302.
Kantarjian HM, Giles F, Gattermann N, Bhalla K, Alimena G, Palandri F et al. Nilotinib (formerly AMN107), a highly selective BCR-ABL tyrosine kinase inhibitor, is effective in patients with Philadelphia chromosome-positive chronic myelogenous leukemia in chronic phase following imatinib resistance and intolerance. Blood 2007; 110: 3540–3546.
Shimoni A, Leiba M, Schleuning M, Martineau G, Renaud M, Koren-Michowitz M et al. Prior treatment with the tyrosine kinase inhibitors dasatinib and nilotinib allows stem cell transplantation (SCT) in a less advanced disease phase and does not increase SCT toxicity in patients with chronic myelogenous leukemia and Philadelphia positive acute lymphoblastic leukemia. Leukemia 2009; 23: 190–194.
Kantarjian HM, Kim D, Dorlhiac-Llacer P, Pasquini R, Khoroshko N, DiPersio JF et al. Dasatinib 140 mg once daily (QD) demonstrates equivalent efficacy and improved safety compared with 70 mg twice daily (BID) in patients with accelerated phase chronic myeloid leukemia (CML-AP): 2-year follow-up data from CA180-035. Blood 2008; 112: 1106–1107.
Acknowledgements
We thank Analysis Group Inc., Boston, MA, USA for conducting and assisting with statistical analyses. JFA is grateful for support from the NIHR Biomedical Research Centre funding scheme. Financial support for medical editorial assistance was provided by Novartis Pharmaceuticals. We thank Daniel Hutta, PhD and Erinn Goldman, PhD for medical editorial assistance with this manuscript. Financial support for medical editorial assistance was provided by Novartis Pharmaceuticals, sponsors of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PDleC received research funding and honoraria from Bristol–Myers Squibb and Novartis, and participated in speakers’ bureaus for Bristol–Myers Squibb and Novartis. FJG acted as a consultant for and received research funding and honoraria from Novartis. AH acted as a consultant for Ariad, Bristol–Myers Squibb and Novartis, received research funding from Bristol–Myers Squibb, Novartis and Pfizer, and received honoraria from and participated on advisory committees for Bristol–Meyers Squibb and Novartis. JFA received research funding from Novartis, received honoraria from Bristol–Myers Squibb, Chemgenex and Novartis, and participated on advisory committees for Ariad, Bristol–Myers Squibb, Chemgenex and Novartis. GJO acted as a consultant for and received research funding from Novartis, and has received honoraria and participated on advisory committees for Bristol–Myers Squibb and Novartis. RB and YS are Novartis employees. NJG is an employee and shareholder of Novartis. MB has received research funding from Novartis and acted as a consultant for, received honoraria from and participated in speakers’ bureaus for Bristol–Myers Squibb and Novartis. JC acted as a consultant for Bristol–Myers Squibb, Chemgenex and Pfizer, and received research funding from Ariad, Bristol–Myers Squibb, Chemgenex, Novartis and Pfizer. HMK acted as a consultant for Novartis, and received research funding from Bristol–Myers Squibb, Novartis and Pfizer.
Rights and permissions
About this article
Cite this article
le Coutre, P., Giles, F., Hochhaus, A. et al. Nilotinib in patients with Ph+ chronic myeloid leukemia in accelerated phase following imatinib resistance or intolerance: 24-month follow-up results. Leukemia 26, 1189–1194 (2012). https://doi.org/10.1038/leu.2011.323
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.323
- Springer Nature Limited
Keywords
This article is cited by
-
Pathogenesis and management of accelerated and blast phases of chronic myeloid leukemia
Leukemia (2023)
-
An Update on the Management of Advanced Phase Chronic Myeloid Leukemia
Current Hematologic Malignancy Reports (2023)
-
NOVEL-1st: an observational study to assess the safety and efficacy of nilotinib in newly diagnosed patients with Philadelphia chromosome-positive chronic myeloid leukemia in chronic phase in Taiwan
International Journal of Hematology (2022)
-
Genetics and tyrosine kinase inhibitors of chronic myeloid leukemia
The Nucleus (2019)
-
Modernes Management der chronischen myeloischen Leukämie*
Im Focus Onkologie (2018)