Abstract
Objective:
Lactoferrin is a pleiotropic glycoprotein of the innate immune system with known effects on immunomodulation and cell differentiation. To gain an insight into the interaction among obesity, inflammation and insulin action, we aimed to examine the effects of lactoferrin on adipogenesis and the response to insulin in human hepatocarcinoma (HepG2) and 3T3-L1 cell lines.
Design:
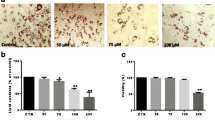
The cells were cultured with increasing lactoferrin concentration under non-inflammatory, inflammatory and standard conditions. The response to insulin was evaluated through 473SerAKT phosphorylation. The effects of lactoferrin on adipogenesis were studied through the expression of different lipogenic markers, AMP-activated protein kinase (AMPK) activation, retinoblastoma (Rb) activity and Oil Red O staining in 3T3-L1 cells.
Results:
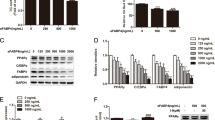
Lactoferrin increased dose-dependent insulin-induced 473SerAKT phosphorylation in both cell lines. Inflammation-induced decreased 473SerAKT phosphorylation was also rescued by lactoferrin. In addition, lactoferrin led to increased p172ThrAMPK during 3T3-L1 differentiation and to decreased adipogenesis (as shown by decreased expression of fatty acid synthase, acetyl-coenzyme A carboxylase-α and peroxisome proliferator-activated receptor-γ in parallel with decreased formation of lipid droplets). Lactoferrin also increased dose-dependent Rb activity (expression and hypophosphorylation) during 3T3-L1 differentiation.
Conclusion:
Lactoferrin administration increased insulin-induced 473SerAKT phosphorylation, even in those conditions wherein the response to insulin was downregulated, and led to blunted adipogenesis in the context of increased p172ThrAMPK and Rb activity.








Similar content being viewed by others
References
Lumeng CN, Deyoung SM, Saltiel AR . Macrophages block insulin action in adipocytes by altering expression of signaling and glucose transport proteins. Am J Physiol Endocrinol Metab 2007; 292: E166–E174.
Constant VA, Gagnon A, Landry A, Sorisky A . Macrophage-conditioned medium inhibits the differentiation of 3T3-L1 and human abdominal preadipocytes. Diabetologia 2006; 49: 1402–1411.
Zhou L, Sell H, Eckardt K, Yang Z, Eckel J . Conditioned medium obtained from in vitro differentiated adipocytes and resistin induce insulin resistance in human hepatocytes. FEBS Lett 2007; 581: 4303–4308.
Yarmo MN, Landry A, Molgat AS, Gagnon A, Sorisky A . Macrophage-conditioned medium inhibits differentiation-induced Rb phosphorylation in 3T3-L1 preadipocytes. Exp Cell Res 2009; 315: 411–418.
Jager J, Grémeaux T, Cormont M, Le Marchand-Brustel Y, Tanti JF . Interleukin-1beta-induced insulin resistance in adipocytes through down-regulation of insulin receptor substrate-1 expression. Endocrinology 2007; 148: 241–251.
Krogh-Madsen R, Plomgaard P, Moller K, Mittendorfer B, Pedersen BK . Influence of TNF-alpha and IL-6 infusions on insulin sensitivity and expression of IL-18 in humans. Am J Physiol Endocrinol Metab 2006; 291: E108–E114.
Lagathu C, Yvan-Charvet L, Bastard JP, Maachi M, Quignard-Boulangé A, Capeau J et al. Long-term treatment with interleukin-1beta induces insulin resistance in murine and human adipocytes. Diabetologia 2006; 49: 2162–2173.
Stephens JM, Lee J, Pilch PF . Tumor necrosis factor-alpha-induced insulin resistance in 3T3-L1 adipocytes is accompanied by a loss of insulin receptor substrate-1 and GLUT4 expression without a loss of insulin receptor-mediated signal transduction. J Biol Chem 1997; 272: 971–976.
Senn JJ, Klover PJ, Nowak IA, Mooney RA . Interleukin-6 induces cellular insulin resistance in hepatocytes. Diabetes 2002; 51: 3391–3399.
Ward PP, Paz E, Conneely OM . Multifunctional roles of lactoferrin: a critical overview. Cell Mol Life Sci 2005; 62: 2540–2548.
Otsuki K, Yakuwa K, Sawada M, Hasegawa A, Sasaki Y, Mitsukawa K et al. Recombinant human lactoferrin has preventive effects on lipopolysaccharide-induced preterm delivery and production of inflammatory cytokines in mice. J Perinat Med 2005; 33: 320–323.
Conneely OM . Antiinflammatory activities of lactoferrin. J Am Coll Nutr 2001; 20: 389S–395S, discussion 396S-397S.
Hayashida K, Kaneko T, Takeuchi T, Shimizu H, Ando K, Harada E . Oral administration of lactoferrin inhibits inflammation and nociception in rat adjuvant-induced arthritis. J Vet Med Sci 2004; 66: 149–154.
Kruzel ML, Harari Y, Chen CY, Castro GA . The gut. A key metabolic organ protected by lactoferrin during experimental systemic inflammation in mice. Adv Exp Med Biol 1998; 443: 167–173.
Zimecki M, Artym J, Chodaczek G, Kocieba M, Kruzel ML . Protective effects of lactoferrin in Escherichia coli-induced bacteremia in mice: relationship to reduced serum TNF alpha level and increased turnover of neutrophils. Inflamm Res 2004; 53: 92–96.
Haversen L, Ohlsson BG, Hahn-Zoric M, Hanson LA, Mattsby-Baltzer I . Lactoferrin down-regulates the LPS-induced cytokine production in monocytic cells via NF-kappa B. Cell Immunol 2002; 220: 83–95.
Zhou Y, Zeng Z, Zhang W, Xiong W, Wu M, Tan Y et al. Lactotransferrin: a candidate tumor suppressor-deficient expression in human nasopharyngeal carcinoma and inhibition of NPC cell proliferation by modulating the mitogen-activated protein kinase pathway. Int J Cancer 2008; 123: 2065–2072.
Letchoumy PV, Mohan KV, Stegeman JJ, Gelboin HV, Hara Y, Nagini S . In vitro antioxidative potential of lactoferrin and black tea polyphenols and protective effects in vivo on carcinogen activation, DNA damage, proliferation, invasion, and angiogenesis during experimental oral carcinogenesis. Oncol Res 2008; 17: 193–203.
Naot D, Grey A, Reid IR, Cornish J . Lactoferrin—a novel bone growth factor. Clin Med Res 2005; 3: 93–101.
Maneva A, Taleva B, Maneva L . Lactoferrin-protector against oxidative stress and regulator of glycolysis in human erythrocytes. Z Naturforsch C 2003; 58: 256–262.
Buren J, Lai YC, Lundgren M, Eriksson JW, Jensen J . Insulin action and signalling in fat and muscle from dexamethasone-treated rats. Arch Biochem Biophys 2008; 474: 91–101.
Yagi M, Suzuki N, Takayama T, Arisue M, Kodama T, Yoda Y et al. Lactoferrin suppress the adipogenic differentiation of MC3T3-G2/PA6 cells. J Oral Sci 2008; 50: 419–425.
Yagi M, Suzuki N, Takayama T, Arisue M, Kodama T, Yoda Y et al. Effects of lactoferrin on the differentiation of pluripotent mesenchymal cells. Cell Biol Int 2009; 33: 283–289.
Rosen CJ, Bouxsein ML . Mechanisms of disease: is osteoporosis the obesity of bone? Nat Clin Pract Rheumatol 2006; 2: 35–43.
Gimble JM, Zvonic S, Floyd ZE, Kassem M, Nuttall ME . Playing with bone and fat. J Cell Biochem 2006; 98: 251–266.
Rossmeisl M, Flachs P, Brauner P, Sponarova J, Matejkova O, Prazak T et al. Role of energy charge and AMP-activated protein kinase in adipocytes in the control of body fat stores. Int J Obes Relat Metab Disord 2004; 28 Suppl 4: S38–S44.
Usui I, Haruta T, Iwata M, Takano A, Uno T, Kawahara J et al. Retinoblastoma protein phosphorylation via PI 3-kinase and mTOR pathway regulates adipocyte differentiation. Biochem Biophys Res Commun 2000; 275: 115–120.
Abella A, Dubus P, Malumbres M, Rane SG, Kiyokawa H, Sicard A et al. Cdk4 promotes adipogenesis through PPARgamma activation. Cell Metab 2005; 2: 239–249.
Son HJ, Lee SH, Choi SY . Human lactoferrin controls the level of retinoblastoma protein and its activity. Biochem Cell Biol 2006; 84: 345–350.
Sakoda H, Ogihara T, Anai M, Funaki M, Inukai K, Katagiri H et al. Dexamethasone-induced insulin resistance in 3T3-L1 adipocytes is due to inhibition of glucose transport rather than insulin signal transduction. Diabetes 2000; 49: 1700–1708.
Oda N, Nakai A, Mokuno T, Sawai Y, Nishida Y, Mano T et al. Dexamethasone-induced changes in glucose transporter 4 in rat heart muscle, skeletal muscle and adipocytes. Eur J Endocrinol 1995; 133: 121–126.
Acknowledgements
This work was partially supported by research grants from the Ministerio de Educación y Ciencia (SAF2008-0273).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moreno-Navarrete, J., Ortega, F., Ricart, W. et al. Lactoferrin increases 172ThrAMPK phosphorylation and insulin-induced p473SerAKT while impairing adipocyte differentiation. Int J Obes 33, 991–1000 (2009). https://doi.org/10.1038/ijo.2009.143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.143
- Springer Nature Limited
Keywords
This article is cited by
-
Antidiabetic efficacy of lactoferrin in type 2 diabetic pediatrics; controlling impact on PPAR-γ, SIRT-1, and TLR4 downstream signaling pathway
Diabetology & Metabolic Syndrome (2018)
-
Bovine lactoferrin promotes energy expenditure via the cAMP-PKA signaling pathway in human reprogrammed brown adipocytes
BioMetals (2018)
-
Protective effects of maternal nutritional supplementation with lactoferrin on growth and brain metabolism
Pediatric Research (2014)
-
Liver, but not adipose tissue PEDF gene expression is associated with insulin resistance
International Journal of Obesity (2013)