Abstract
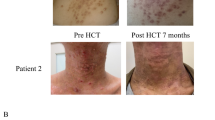
Autosomal-recessive hyper-IgE syndrome (AR-HIES) is a combined immunodeficiency recently found to be associated with mutations of DOCK8. Clinically, this disorder is characterized beside recurrent bacterial complications, in particular by an unusual susceptibility to extensive cutaneous viral complications and by a high risk for squamous cell carcinoma. Here, we report on lasting control over the disorder in two patients by hematopoietic cell transplantation (HCT). Both patients were suffering from extensive long-lasting cutaneous viral complications, in particular from disfiguring molluscum contagiosum infections, when treated at the age of 10 and 17 years. Donors were matched unrelated, and conditioning was carried out with a combination of fludarabine, melphalan and BM-targeted radioimmunotherapy. Both patients developed stable, full donor cell chimerism, with the exception of persistent low-IgA serum levels and the exception of normal immune functions. Over the course of several months, cutaneous manifestations of viral disease resolved completely and both patients remain clinically well and free of infectious complications at 4 and 2 years, respectively, after transplantation. This represents the first report indicating HCT to be curative in patients with AR-HIES, which should be considered early before life-threatening complications develop, which include malignancies.


Similar content being viewed by others
References
Zhang Q, Davis JC, Lamborn IT, Freeman AF, Jing H, Favreau AJ et al. Combined immunodeficiency associated with DOCK8 mutations. N Engl J Med 2009; 361: 2046–2055.
Renner ED, Puck JM, Holland SM, Schmitt M, Weiss M, Frosch M et al. Autosomal recessive hyperimmunoglobulin E syndrome: a distinct disease entity. J Pediatr 2004; 144: 93–99.
Engelhardt KR, McGhee S, Winkler S, Sassi A, Woellner C, Lopez-Herrera G et al. Large deletions and point mutations involving the dedicator of cytokinesis 8 (DOCK8) in the autosomal-recessive form of hyper-IgE syndrome. J Allergy Clin Immunol 2009; 124: 1289–1302.
Minegishi Y, Saito M, Morio T, Watanabe K, Agematsu K, Tsuchiya S et al. Human tyrosine kinase 2 deficiency reveals its requisite roles in multiple cytokine signals involved in innate and acquired immunity. Immunity 2006; 25: 745–755.
Freeman AF, Holland SM . The hyper-IgE syndromes. Immunol Allergy Clin North Am 2008; 28: 277–291.
Holland SM, DeLeo FR, Elloumi HZ, Hsu AP, Uzel G, Brodsky N et al. STAT3 mutations in the hyper-IgE syndrome. N Engl J Med 2007; 357: 1608–1619.
Milner JD, Brenchley JM, Laurence A, Freeman AF, Hill BJ, Elias KM et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008; 452: 773–776.
Schulz A, Gatz S, Schuetz C, Hoenig M, Debatin KM, Friedrich W et al. Radioimmunotherapy for myeloablation before SCT in paediatric patients with malignant and non-malignant diseases. Blood 2007; 110: 624.
Acknowledgements
We thank Katja Heinrich and Eva-Maria Rump for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gatz, S., Benninghoff, U., Schütz, C. et al. Curative treatment of autosomal-recessive hyper-IgE syndrome by hematopoietic cell transplantation. Bone Marrow Transplant 46, 552–556 (2011). https://doi.org/10.1038/bmt.2010.169
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.169
- Springer Nature Limited
Keywords
This article is cited by
-
RHO GTPases: from new partners to complex immune syndromes
Nature Reviews Immunology (2021)
-
CNS vasculitis and stroke as a complication of DOCK8 deficiency: a case report
BMC Neurology (2016)
-
Recent Advances in DOCK8 Immunodeficiency Syndrome
Journal of Clinical Immunology (2016)
-
Delayed control of herpes simplex virus infection and impaired CD4+ T‐cell migration to the skin in mouse models of DOCK8 deficiency
Immunology & Cell Biology (2015)
-
DOCK8 Deficiency: Clinical and Immunological Phenotype and Treatment Options - a Review of 136 Patients
Journal of Clinical Immunology (2015)