Abstract
Background:
High-risk human papillomavirus (hrHPV)-positive women require triage to identify those with cervical high-grade intraepithelial neoplasia and cancer (⩾CIN3 (cervical intraepithelial neoplasia grade 3)). FAM19A4 methylation analysis, which detects advanced CIN and cancer, is applicable to different sample types. However, studies comparing the performance of FAM19A4 methylation analysis in hrHPV-positive self-samples and paired physician-taken scrapes are lacking.
Methods:
We compared the performance of FAM19A4 methylation analysis (and/or HPV16/18 genotyping) in self-samples and paired physician-taken scrapes for ⩾CIN3 detection in hrHPV-positive women (n=450,18–66 years).
Results:
Overall FAM19A4 methylation levels between sample types were significantly correlated, with strongest correlation in women with ⩾CIN3 (Spearman’s ρ 0.697, P<0.001). The performance of FAM19A4 methylation analysis and/or HPV16/18 genotyping did not differ significantly between sample types. In women ⩾30 years, ⩾CIN3 sensitivity of FAM19A4 methylation analysis was 78.4% in self-samples and 88.2% in scrapes (ratio 0.89; CI: 0.75–1.05). In women <30 years, ⩾CIN3 sensitivities were 37.5% and 45.8%, respectively (ratio 0.82; CI: 0.55–1.21). In both groups, ⩾CIN3 specificity of FAM19A4 methylation analysis was significantly higher in self-samples compared with scrapes.
Conclusions:
FAM19A4 methylation analysis in hrHPV-positive self-samples had a slightly lower sensitivity and a higher specificity for ⩾CIN3 compared with paired physician-taken scrapes. With a similarly good clinical performance in both sample types, combined FAM19A4 methylation analysis and HPV16/18 genotyping provides a feasible triage strategy for hrHPV-positive women, with direct applicability on self-samples.
Similar content being viewed by others
Main
Epidemiological studies have established that a persistent infection with a high-risk type of the human papillomavirus (hrHPV) is a necessary aetiological factor for cervical cancer. Several randomised trials have demonstrated that testing for the presence of hrHPV is a more sensitive screening tool compared with cytology, providing improved detection of cervical intraepithelial neoplasia grade 3 (CIN3) and cervical cancer (Dillner, 2013; Ronco et al, 2014). However, the majority of hrHPV infections is transient, and only upon accumulation of genetic and epigenetic changes in the host and/or viral genomes, progression towards invasive cancer may occur (Snijders et al, 2006; Steenbergen et al, 2014). To avoid unnecessary referral to the gynaecologist of women with transient hrHPV infections, identification of women with lesions reflecting clinically relevant infections with a high risk of progression towards cancer is required. Currently, cytology is the most advocated triage tool for this purpose (Zorzi et al, 2013; Dijkstra et al, 2014). However, cytology triage requires repeat testing to ensure sufficient sensitivity (Rijkaart et al, 2012; Dijkstra et al, 2014) and has a subjective test result (Dijkstra et al, 2014). Moreover, prior knowledge of the hrHPV status influences cytology reading, resulting in an easier classification of abnormal cytology, thus increasing the number of false-positives (Moriarty et al, 2014). Although HPV16/18 genotyping might be a valuable addition to cytology by eliminating the necessity of a repeat test (Cox et al, 2013; Dijkstra et al, 2014), this combination may suffer from non-detection of (pre)cancers associated with non-HPV16/18 hrHPV types.
As gene silencing by promoter hypermethylation has been shown to contribute to cervical carcinogenesis, methylation analysis of cancer-specific genes has been suggested as a valuable, alternative or additive triage tool (Steenbergen et al, 2004; Overmeer et al, 2009; Wilting et al, 2010; Saavedra et al, 2012). Furthermore, in contrast to cytology, DNA methylation analysis detects virtually all cervical carcinomas and is assumed to detect ‘advanced’ CIN lesions, that is, lesions with a long duration of existence and with many chromosomal aberrations, which have a high risk of short-term progression to cancer (De Strooper et al, 2014; Steenbergen et al, 2014). Gene promoter methylation can be easily accessed by sensitive, quantitative methylation-specific PCR (qMSP) providing an objective test outcome. Previous results in hrHPV-positive cervical scrapes have been promising (Huang et al, 2010; Hesselink et al, 2011, 2014; Eijsink et al, 2012; Verhoef et al, 2014a), with sensitivities for CIN3 and cervical cancer (⩾CIN3) equalling those of cytology analysis (Hesselink et al, 2011; Verhoef et al, 2014a).
An important development in cervical cancer screening is the use of self-collected cervicovaginal samples for women who do not attend screening (Ogilvie et al, 2005; Snijders et al, 2013; Arbyn et al, 2014). However, as cytology cannot be reliably performed on self-sampled material (Nobbenhuis et al, 2002; Garcia et al, 2003), triage of hrHPV-positive women by cytology still requires a visit to a physician. In contrast, methylation analysis can be performed directly on the self-collected sample (Eijsink et al, 2011; Hesselink et al, 2014; Verhoef et al, 2014a). Indeed, a prospective trial in non-responders has shown that detection of high-grade CIN with direct MAL/miR124-2 methylation analysis on hrHPV-positive lavage-collected self-samples is non-inferior to cytology triage on hrHPV-positive physician-taken scrapes, thereby preventing diagnostic delay and loss to follow-up (Verhoef et al, 2014a). Recently, methylation analysis of the FAM19A4 gene has been shown to yield attractive ⩾CIN3 sensitivities and specificities in both hrHPV-positive brush-collected and lavage-collected self-samples (De Strooper et al, 2016) supporting the concept that it serves as a universal triage test for HPV-positive cervicovaginal self-samples collected by different self-collection devices.
Studies evaluating the concordance of methylation analysis between self-collected cervicovaginal samples and physician-taken cervical scrapes are however limited (Boers et al, 2014; Chang et al, 2015). Such evaluations are crucial to reveal the relative clinical performance for ⩾CIN3 detection compared with physician-taken scrapes, as has been assessed for HPV testing (Arbyn et al, 2014). It is well known that each sample type has its own cellular composition with likely a different fraction of indicator cells exfoliated from ⩾CIN3, which may affect the performance of molecular reflex tests. The present study compared the clinical performance of FAM19A4 methylation analysis with and without additional HPV16/18 genotyping, for the detection of ⩾CIN3 in a unique cohort of paired self-collected cervicovaginal lavage samples and physician-taken cervical scrapes of hrHPV-positive women.
Materials and methods
Study design, participants and procedures
The present study was conducted as a post hoc analysis of data obtained in a prospective observational multicentre cohort study (COMETH), which aimed to compare different triage strategies in hrHPV-positive women (Luttmer et al, 2016). From December 2010 till December 2013, women (age 18–70 years) who visited a gynaecological outpatient clinic in one of six hospitals in the Netherlands were asked to participate in the study. Women could participate in the study regardless of their reason for referral to the gynaecology outpatient clinic. After providing informed consent, participants collected cervicovaginal lavage material (using the Delphi screener; Delphi Bioscience, Scherpenzeel, The Netherlands) for hrHPV testing. Women who met the inclusion criteria and tested hrHPV-positive on the cervicovaginal lavage were invited for a cervical scrape and colposcopy. The cervical scrape was taken by a physician using a Cervex-Brush (Rovers Medical Devices BV, Oss, The Netherlands) or a Medscand Cytobrush Plus (CooperSurgical Inc., Trumbull, CT, USA). Scrape material was stored in 20 ml of Thinprep PreservCyt solution (Hologic, Marlborough, MA, USA) and was evaluated by FAM19A4 methylation analysis and HPV16/18 genotyping (cytology results on these scrapes have been reported previously; Luttmer et al, 2016)). The study was approved by the Medical Ethical Committee of all participating hospitals (METc-VUmc2009/178) and registered as NTR2447. Women with a history of treatment for cervical dysplasia or cervical cancer, current cancer, pregnancy or lactation were excluded from participation (Luttmer et al, 2016).
For logistic reasons, from 141 (31.3%) women, cervicovaginal lavage material was collected by the physician before cervical scraping using a Delphi screener according to the protocol. The remaining 309 (68.7%) women self-collected lavage material at home. In 268 (59.5%) women, the cervical scrape was taken at a minimum of 2 weeks before colposcopy. In 182 (40.4%) women, for logistic reasons, the cervical scrape was taken immediately before colposcopy during the same visit to the gynaecologist.
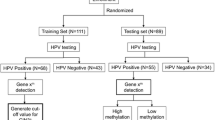
As shown in Figure 1, in all women who tested both hrHPV-positive on the cervicovaginal lavage and the cervical scrape (n=532), FAM19A4 methylation analysis was performed on each sample type. All of these women had colposcopy-directed biopsy. Women with invalid test results for FAM19A4 methylation analysis of the cervicovaginal lavage (n=48; 9.0%) or cervical scrape (n=43; 8.0%) were excluded from analyses. Of these, nine women had an invalid test result in both the lavage and scrape samples. The remaining women comprised the final study population (n=450).
Overview of the study population and histological endpoints. *Histological endpoints were based on the histological outcome of the colposcopy-directed biopsy, or, if classified worse, on the histology result of the specimen excised by LLETZ, conisation or hysterectomy. AIS=adenocarcinoma in situ; CIN=cervical intraepithelial neoplasia; hrHPV=high-risk human papillomavirus.
Virtually all women (65 of 66; 98.4%) with histologically confirmed CIN3 and adenocarcinoma in situ (AIS) in the biopsy specimen were treated by large loop excision of the transformation zone (LLETZ) or cervical conisation; one woman was followed up without treatment. Depending on the size of the lesion, also 55 of 85 (64.7%) women with a CIN2 biopsy underwent LLETZ. Of these women, eight (14.5%) were diagnosed with CIN3 in the LLETZ tissue, and categorised accordingly. In addition, one woman with a CIN0 in the biopsy specimen had a CIN3 in the tissue, which was obtained by (diagnostic) LLETZ.
Histological endpoints
In concordance with earlier work (Luttmer et al, 2016), we used histologically confirmed ⩾CIN3 as primary study end point and ⩾CIN2 as a secondary study end point. Unless otherwise specified, histological end points were based on the histological outcome of the colposcopy-directed biopsy, or, if classified worse, on the histology result of the specimen excised by LLETZ, conisation or hysterectomy. In addition, we attempted to provide a rough estimation of the volume of CIN lesions. Therefore, histology results of biopsy specimens and those of the corresponding LLETZ (or conisation/hysterectomy) specimens are presented (Table 1). The presence of ⩾CIN3 in the LLETZ (or conisation/hysterectomy) specimen, irrespective of the histological result of the biopsy, was considered indicative of a high-volume ⩾CIN3 lesion. The presence of CIN3 in the biopsy specimen, combined with ⩽CIN2 in the corresponding LLETZ specimen, was assumed to represent a low-volume CIN3 lesion. Similarly, the presence of CIN2 in the LLETZ specimen combined with ⩽CIN2 in the corresponding biopsy specimen was considered to indicate a high-volume CIN2 lesion. The presence of CIN2 in the biopsy with ⩽CIN1 in the LLETZ specimen was assumed to represent a low-volume CIN2 lesion. In women who were diagnosed with high-grade CIN on the biopsy specimen and who did not receive LLETZ (according to physician’s advice), lesions were classified as low volume.
HPV genotyping
DNA was isolated from one-fifth of cervicovaginal lavage specimens and one-tenth of cervical scrape material using the Nucleo-Mag 96 Tissue Kit (Macherey-Nagel, Düren, Germany) and a Microlab Star robotic system (Hamilton, Planegg, Germany) according to the manufacturer’s instructions (Hesselink et al, 2011). Isolated DNA was subjected to GP5+/6+ PCR-enzyme immunoassay analysis (EIA Kit HPV GP HR; Diassay BV, Rijswijk, The Netherlands) (Jacobs et al, 1997). Subsequent genotyping for the high-risk HPV types 16/18/31/33/35/39/45/51/52/56/58/59/66/68 was performed using a microsphere bead-based assay (Luminex xMAP, Luminex Corp, Austin, TX, USA) as described previously (Schmitt et al, 2006).
qMSP analysis
Extracted DNA from hrHPV-positive cervicovaginal lavage specimens and cervical scrapes was treated with bisulphite using the EZ DNA Methylation Kit (Zymo Research, Irvine, CA, USA) as described previously (Overmeer et al, 2008, 2009). Bisulphite-converted DNA was used as template for FAM19A4 methylation analysis by qMSP using housekeeping gene β-actin (ACTB) as a reference (Steenbergen et al, 2013; De Strooper et al, 2014). Quantitative methylation-specific PCR analysis was carried out using an ABI 7500 Real-Time PCR-System (Applied Biosystems, Waltham, MA, USA). For each target, quantification cycle (Cq) values were measured at a fixed fluorescence threshold. To assure sample quality, all samples included in the study had a Cq value for ACTB <30. In case of poor DNA concentration and an invalid test result, DNA isolation, bisulphite treatment and qMSP were repeated with double sample input if sufficient material was available for analysis. Cq ratios were calculated for each sample using the following formula: 2(Cq (ACTB)−Cq (FAM19A4)) × 100. For both cervicovaginal lavage specimens and cervical scrapes, assay thresholds, which gave rise to a ⩾CIN3 specificity of 70% using a training-validation set approach (De Strooper et al, 2014), were chosen to consider a specimen positive for FAM19A4 methylation.
Statistical analysis
The sample size was set such that 90% power was achieved for demonstrating non-inferiority of FAM19A4 methylation analysis in cervicovaginal lavages to FAM19A4 methylation analysis in cervical scrapes using a matched-sample score test (Tang et al, 2003; Meijer et al, 2009). A minimum of 300 hrHPV-positive women needed to be included at rejection rate α of 0.05. Finally, 450 hrHPV-positive women were included with results for all markers.
For assessing overall genotype concordance, results were either scored as concordant (sample types yielded completely identical genotype results), compatible (one or more of the same genotypes were detected) or discordant (no genotype similarities detected). Paired evaluation of FAM19A4 Cq ratios in cervicovaginal lavages and in cervical scrapes was carried out by Spearman’s rank analysis.
The relation of the classification combining histological severity and lesion volume with the percentage of FAM19A4 methylation-positive and/or HPV16/18-positive samples was assessed using Fisher’s exact test.
For both cervicovaginal lavages and cervical scrapes, clinical performance of FAM19A4 methylation analysis and the combined use of FAM19A4 methylation analysis and HPV16/18 genotyping was evaluated. Clinical performance indicators were sensitivity, specificity, positive predictive value (PPV) and complemented NPV (1-negative predictive value, a measure of disease risk after a negative result) for ⩾CIN3 and ⩾CIN2, and referral rate (based on % marker positivity) were calculated. To enable comparisons, relative sensitivity (ratio of the sensitivity of a marker in one sample type to its sensitivity in the other sample type) and relative specificity (ratio of the specificity of a marker in one sample type to its specificity in the other sample type) were calculated with 95% CIs. If the 95% CI of the relative sensitivity or specificity was entirely below or above one, the difference in sensitivity or specificity was considered statistically significant. In case of non-significant differences in sensitivity or specificity, an additional test was performed to evaluate non-inferiority. Non-inferiority was defined as a relative sensitivity or specificity of at least 90% using a matched-sample score test (Tang et al, 2003; Meijer et al, 2009).
Using logistic regression, we analysed the influence of several factors on the sensitivity and specificity of FAM19A4 methylation analysis in this study population: the age of the participants (<30 years and ⩾30 years), the reason for referral to the gynaecologist (abnormal cytology result or other, non-cervix-related, gynaecological complaints), the sampling method of the cervicovaginal lavage (collected by the participant at home or by the physician) and the sampling moment of the cervical scrape (during a separate visit 2–3 weeks before colposcopy or combined with the colposcopy procedure). After finding a factor that significantly influenced the performance of the different markers, data were stratified for this factor. P-values <0.05 were considered statistically significant. All statistical analyses and computations of graphs were performed in IBM SPSS Statistics 20 (IBM Corporation, Armonk, NY, USA), STATA 11.0 (StataCorp LP, College Station, TX, USA) and Excel.
Results
Study population
The study flowchart and histological end points are shown in Figure 1. The final analysis comprised 450 women (age 18–66 years) who tested hrHPV-positive on both the cervicovaginal lavage material and the cervical scrape, had valid results for FAM19A4 methylation in both sample types and a histological end point. FAM19A4 methylation scored positive in 26.4% (119 of 450) of the cervicovaginal lavage specimens and in 35.8% (161 of 450) of the cervical scrape specimens. Human papillomavirus16 and/or HPV18 was present in 46.4% (209 of 450) of the cervicovaginal lavage specimens and 47.3% (213 of 450) of the cervical scrape specimens. Two women (0.4%) had invasive cervical carcinoma (i.e., one squamous cell carcinoma (SCC) and one adenocarcinoma), 73 women (16.2%) had CIN3 (including one AIS), 78 women (17.3%) had CIN2, 124 (27.6%) had CIN1 and 173 (38.4%) had no CIN. In Table 1, histological results of biopsy specimens and corresponding LLETZ results are presented.
Concordance of HPV genotypes and FAM19A4 methylation analysis
Genotype agreement between the cervical scrape and cervicovaginal lavage was concordant (sample types yielded completely identical genotype results) in 60.7% (273 of 450) and compatible (one or more of the same genotypes were detected) in 28.9% (130 of 450) of cases. In the remaining cases (47 of 450; 10.4%), genotypes were discordant between both sample types. As shown in Table 2, for the majority of hrHPV types, genotype concordance between cervical scrapes and cervicovaginal lavages was substantial to almost perfect (Landis and Koch, 1977), both in the total group and in the strata of histological severity. FAM19A4 Cq ratios in lavage samples were significantly correlated to those in cervical scrapes in the total study population (Spearman’s ρ 0.394, P<0.001). In the strata of histological severity, this correlation was found to be strongest in women with ⩾CIN3 (Spearman’s ρ 0.697, P<0.001), followed by CIN2 (Spearman’s ρ 0.255, P=0.024) and ⩽CIN1 (Spearman’s ρ 0.257, P<0.001). The correlation between methylation Cq ratio in cervical scrapes and lavage samples in the strata of histological severity is presented in Supplementary Figure 1. Both women diagnosed with cervical cancer tested positive for FAM19A4 methylation analysis in both sample types. The woman with an adenocarcinoma tested HPV16-positive, whereas the woman with an SCC harboured HPV39.
Performance of FAM19A4 methylation analysis in total study population
Test specifications of FAM19A4 methylation analysis in cervical scrapes (which were described previously; Luttmer et al, 2016) and in cervicovaginal lavages for detection of ⩾CIN3 and ⩾CIN2 in the total study population are shown in Table 3 (upper panel). Although the sensitivity of FAM19A4 methylation analysis for the detection of ⩾CIN3 was lower in cervicovaginal lavage material (65.3%) compared with that in cervical scrapes (74.7%), this difference was not statistically significant (ratio 0.88; CI: 0.75–1.02). However, statistical non-inferiority (of lavages relative to scrapes) could not be demonstrated (P=0.64). The ⩾CIN3 specificity of FAM19A4 methylation analysis was significantly higher in cervicovaginal lavages (81.3%) compared with that in cervical scrapes (72.0%; ratio 1.13; CI: 1.05–1.21). For ⩾CIN2 outcome in the total study population (Table 3; upper panel), FAM19A4 methylation analysis had a significantly lower sensitivity in cervicovaginal lavages (44.4%) compared with that in cervical scrapes (56.9%), whereas its specificity in lavages (82.8%) was significantly higher compared with that in scrapes (75.1%; relative specificity 1.10; CI: 1.03–1.18).
Factors influencing the performance of FAM19A4 methylation analysis: age
The performance of FAM19A4 methylation analysis was significantly influenced by the age of the participants in both sample types (Supplementary Tables 1A and B). Neither the reason for referral of the participant (abnormal cytology result or other, non-cervix-related, gynaecologic complaints) nor the sampling method of the lavage (self-collected by the participant at home or physician-collected) or scrape (during a separate visit 2–3 weeks before colposcopy or combined with the colposcopy procedure) influenced the performance of FAM19A4 methylation analysis significantly (Supplementary Tables 1A and B). Given the influence of age, the performance of FAM19A4 methylation analysis was assessed in subgroups of women 18–30 years (referred to as <30years) and women 30–66 years (referred to as ⩾30 years). Sensitivity, specificity, PPV and complemented NPV of FAM19A4 methylation analysis in both sample types and in both age strata are shown in Table 3.
In women ⩾30 years of age (n=252), 51 ⩾CIN3, 43 CIN2 and 158 ⩽CIN1 were present (Table 3; middle panel). In this sub-population, the sensitivity of FAM19A4 methylation analysis for ⩾CIN3 was lower in cervicovaginal lavages (78.4%) compared with that in cervical scrapes (88.2%). Although this difference was not statistically significant (ratio 0.89; CI: 0.75–1.05), statistical non-inferiority could not be demonstrated (P=0.56). The specificity of FAM19A4 methylation analysis for ⩾CIN3 was significantly higher in lavages (73.1%) compared with that in scrapes (63.2%; ratio 1.16; CI: 1.03–1.30). For detection of ⩾CIN2 in women aged ⩾30 years, the sensitivity of FAM19A4 methylation analysis in lavages (58.5%) was lower compared with that in scrapes (70.2%; ratio 0.83; CI: 0.70–0.999), and its specificity in lavages (75.3%) was higher compared with that in scrapes (66.5%; ratio 1.13; CI: 1.01–1.28).
In the subgroup of women <30 years of age (n=198), 24 ⩾CIN3, 35 CIN2 and 139 ⩽CIN1 were present (Table 3, lower panel). The sensitivity of FAM19A4 methylation analysis for ⩾CIN3 in lavages did not differ significantly from its sensitivity in scrapes (37.5% vs 45.8%; ratio 0.82; CI: 0.55–1.21), but statistical non-inferiority could not be established (P=0.69). The specificity of FAM19A4 methylation analysis for ⩾CIN3 in lavages (90.8%) was significantly higher compared with that in scrapes (82.2%; ratio 1.10; CI: 1.02–1.20). The sensitivity of FAM19A4 methylation for ⩾CIN2 in lavages was significantly lower in lavages (22.0%) compared with that in scrapes (35.6%; ratio 0.62; CI: 0.40–0.96), whereas the ⩾CIN2 specificity of this marker in lavages (91.4%) was non-inferior to its specificity in scrapes (84.9%; ratio 1.08; CI: 0.99–1.17; non-inferiority test: P=0.01).
The performance of FAM19A4 methylation analysis combined with HPV16/18 genotyping
Table 4 presents the clinical performance of FAM19A4 methylation analysis combined with HPV16/18 genotyping for ⩾CIN3 and ⩾CIN2 in both sample types. Compared with FAM19A4 methylation analysis alone (Table 3), the combined marker panel (Table 4) reached significantly higher ⩾CIN3 and ⩾CIN2 sensitivities, at significantly lower specificities in both sample types and in both age categories. In contrast to FAM19A4 methylation analysis alone, the performance of combined FAM19A4 methylation analysis and HPV16/18 genotyping did not differ significantly between cervicovaginal lavages and cervical scrapes (Table 4).
FAM19A4 methylation analysis in relation to histological severity
In an attempt to evaluate FAM19A4 methylation analysis in relation to lesion severity and volume, participants were stratified on the basis of a combination of histological severity and volume of the CIN lesion (Table 1). FAM19A4 methylation positivity rates in cervicovaginal lavages and cervical scrapes, in relation to this combined classification, are presented in Table 5. In both cervicovaginal lavage samples and cervical scrapes, the percentage of FAM19A4 methylation-positive cases increased with the lesion severity and volume (P<0.001 for both sample types; Table 5). A similar increase in positivity rate was observed when combining FAM19A4 methylation analysis and HPV16/18 genotyping, yet at overall higher positivity rates for each volume category (Table 5).
Discussion
This study compared the performance of FAM19A4 methylation analysis in large series of paired self-collected cervicovaginal lavage samples and physician-taken cervical scrapes for the detection of ⩾CIN3 in hrHPV-positive women from a gynaecologic outpatient population. FAM19A4 methylation analysis on self-collected lavage material had a slightly lower sensitivity and a significantly higher specificity compared with FAM19A4 methylation analysis on physician-taken scrapes.
This study is the first large prospective multicentre cohort study comparing the performance of methylation marker analysis in hrHPV-positive cervicovaginal lavage samples to cervical scrapes from the same women. The required number of participants with and without ⩾CIN3 lesions in this study was calculated in advance to allow comparison of sensitivity and specificity in both sample types. Until now, limited data comparing methylation marker performance in paired samples were available. The methylation markers studied by Boers et al (2014) (C13ORF18, EPB41L3, JAM3 and TERT) (Boers et al, 2014) and Chang et al (2015) (PAX1, SOX1 and ZNF582) have shown moderate to good concordance in self-samples and cervical scrapes, yet these studies were performed on very small study populations with limited controls and require confirmation from other prospective studies.
In the current study, the differences in clinical performance between the two sample types can most likely be attributed to a different cellular composition and proportion of hypermethylation-positive cervical indicator cells. During cervical scrape collection, a physician will target the transformation zone of the cervix, contributing to a likely large number of cervical indicator cells. In case of self-collection by a lavage device, a more random distribution of vaginal and cervical indicator cells can be expected. Consequently, the lower fraction of hypermethylated indicator cells in self-collected lavages might explain the slightly lower sensitivity and higher specificity of FAM19A4 methylation analysis.
Our previous studies have shown that FAM19A4 methylation analysis and cytology on HPV-positive cervical scrapes have a similar ⩾CIN3 sensitivity (De Strooper et al, 2014; Luttmer et al, 2016). However, FAM19A4 methylation analysis has proven to be more sensitive for the detection of CIN3 lesions with a longer duration of existence (the so-called advanced lesions) and cervical carcinoma (Luttmer et al, 2016). An advantage of the present study is the availability of both biopsy and LLETZ histological results from all participants with a high-grade lesion enabling a semiquantitative assessment of lesion volume. We showed that in both hrHPV-positive cervicovaginal lavages and cervical scrapes, FAM19A4 methylation-positivity increases with the volume of high-grade CIN lesions. These results support the concept that FAM19A4 methylation analysis has a preference for detecting the larger, probably more advanced CIN lesions (De Strooper et al, 2014). Of note, combined FAM19A4 methylation analysis and HPV16/18 genotyping detected all but one high-volume ⩾CIN3 lesions in both cervical scrapes and cervicovaginal lavages.
As expected, and found previously (Verhoef et al, 2014b; De Strooper et al, 2016), the addition of HPV16/18 genotyping to FAM19A4 methylation analysis yielded a significant increase in sensitivity at the cost of a marked decrease in specificity. Of interest, whereas FAM19A4 methylation analysis alone was found to be slightly less sensitive and more specific in the cervicovaginal lavages compared with the cervical scrapes, no significant differences were found for the marker combining FAM19A4 methylation analysis and HPV16/18 genotyping. Thus, the combination of FAM19A4 methylation analysis with HPV16/18 genotyping might be a safe alternative triage strategy that performs similarly on both sample types.
In line with earlier studies (Luttmer et al, 2016), our data showed a significant influence of age on FAM19A4 methylation positivity in both cervical scrapes and cervicovaginal lavage material. Accordingly, in young women, FAM19A4 methylation analysis results in quite low ⩾CIN2/3 sensitivities of only 37.5–45.8%, at relatively high specificities of 82.2–90.0%. In young women, hrHPV prevalence is known to be high, but the majority of infections are transient and most lesions, also a substantial part of CIN3 lesions, tend to regress spontaneously (Winer et al, 2003; Insinga et al, 2010; Jaisamrarn et al, 2013), contributing to a very low cancer incidence in this age category (Benard et al, 2012). As FAM19A4 methylation analysis has previously shown to preferably detect advanced CIN lesions and cervical cancer (De Strooper et al, 2014; Steenbergen et al, 2014), this lower sensitivity has the clinical benefit that it could protect young women from overtreatment. This is particularly relevant for women in their reproductive age as treatment may lead to adverse pregnancy outcome.
Owing to the selection of an outpatient population, the translation of our results into screening settings should be handled with care. Further confirmation in population-based screening trials is required as the percentage of HPV-positive women with ⩾CIN3 lesions in a screening population is lower compared with that in a gynaecologic referral population. Another limitation of our study is the relatively large number of samples with an invalid test result for FAM19A4 methylation analysis of 8% in the cervical scrapes and 9% in the cervicovaginal lavage samples. These invalid test results on the cervical scrapes might result from cautious scraping during colposcopy to prevent bleeding and thus poor visualisation, leading to low DNA concentrations (Overmeer et al, 2011). Indeed, 88.4% of the invalid results were found in scrapes that were collected directly before colposcopy, a procedure that is not likely to be applied in a routine cervical screening setting. Also in self-samples, the number of cells can be limited or the sample can be of poor quality, for example, by collection or storage failures. This problem can be circumvented by using a larger fraction of the scrape or self-sample in the DNA isolation procedure, or by decreasing the elution volume during isolation. Given the study setting aiming to evaluate different triage strategies, leftover sample material was not always sufficient to perform repeat methylation analysis. In routine setting, this limitation will likely not be applicable, as also supported by the limited occurrence of methylation-invalid samples in a randomised controlled trial in screening setting (Verhoef et al, 2014a).
In conclusion, FAM19A4 methylation marker analysis in hrHPV-positive self-collected lavage samples had a slightly lower sensitivity and a higher specificity for ⩾CIN3 compared with FAM19A4 methylation analysis in paired physician-taken scrapes. Combined FAM19A4 methylation analysis and HPV16/18 genotyping revealed a similarly good clinical performance, which was similar in both sample types. Therefore, this combination could provide a feasible triage strategy for hrHPV-positive women, with the advantage of direct applicability on self-collected material.
Change history
23 August 2016
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Arbyn M, Verdoodt F, Snijders PJF, Verhoef VMJ, Suonio E, Dillner L, Minozzi S, Bellisario C, Banzi R, Zhao F-H, Hillemanns P, Anttila A (2014) Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: a meta-analysis. Lancet Oncol 15: 172–183.
Benard VB, Watson M, Castle PE, Saraiya M (2012) Cervical carcinoma rates among young females in the United States. Obstet Gynecol 120: 1117–1123.
Boers A, Bosgraaf RP, van Leeuwen RW, Schuuring E, Heideman DAM, Massuger LFAG, Verhoef VMJ, Bulten J, Melchers WJG, van der Zee AGJ, Bekkers RLM, Wisman GBA (2014) DNA methylation analysis in self-sampled brush material as a triage test in hrHPV-positive women. Br J Cancer 111: 1095–1101.
Chang C-C, Huang R-L, Liao Y-P, Su P-H, Hsu Y-W, Wang H-C, Tien C-Y, Yu M-H, Lin Y-W, Lai H-C (2015) Concordance analysis of methylation biomarkers detection in self-collected and physician-collected samples in cervical neoplasm. BMC Cancer 15: 418.
Cox JT, Castle PE, Behrens CM, Sharma A, Wright TC, Cuzick J (2013) Comparison of cervical cancer screening strategies incorporating different combinations of cytology, HPV testing, and genotyping for HPV 16/18: results from the ATHENA HPV study. Am J Obstet Gynecol 208: 184.e1–e184.e11.
De Strooper LMA, Meijer CJLM, Berkhof J, Hesselink AT, Snijders PJF, Steenbergen RDM, Heideman DAM (2014) Methylation analysis of the FAM19A4 gene in cervical scrapes is highly efficient in detecting cervical carcinomas and advanced CIN2/3 lesions. Cancer Prev Res (Phila) 7: 1251–1257.
De Strooper LMA, Verhoef VMJ, Berkhof J, Hesselink AT, de Bruin HM, van Kemenade FJ, Bosgraaf RP, Bekkers RLM, Massuger LFAG, Melchers WJG, Steenbergen RDM, Snijders PJF, Meijer CJLM, Heideman DAM (2016) Validation of the FAM19A4/mir124-2 DNA methylation test for both lavage- and brush-based self-samples to detect cervical (pre)cancer in HPV-positive women. Gynecol Oncol 141 (2): 341–347.
Dijkstra M, van Niekerk D, Rijkaart D, van Kemenade FJ, Heideman DAM, Snijders PJF, Meijer CJ, Berkhof J (2014) Primary hrHPV DNA testing in Cervical Cancer screening: how to manage screen positive women? A POBASCAM Trial sub study. Cancer Epidemiol Biomarkers Prev 23: 55–63.
Dillner J (2013) Primary human papillomavirus testing in organized cervical screening. Curr Opin Obstet Gynecol 25: 11–16.
Eijsink JJH, Lendvai Á, Deregowski V, Klip HG, Verpooten G, Dehaspe L, de Bock GH, Hollema H, van Criekinge W, Schuuring E, van der Zee AG, Wisman GB (2012) A four-gene methylation marker panel as triage test in high-risk human papillomavirus positive patients. Int J Cancer 130: 1861–1869.
Eijsink JJH, Yang N, Lendvai A, Klip HG, Volders HH, Buikema HJ, van Hemel BM, Voll M, Coelingh Bennink HJT, Schuuring E, Wisman GB, van der Zee AG (2011) Detection of cervical neoplasia by DNA methylation analysis in cervico-vaginal lavages, a feasibility study. Gynecol Oncol 120: 280–283.
Garcia F, Barker B, Santos C, Brown EM, Nuño T, Giuliano A, Davis J (2003) Cross-sectional study of patient- and physician-collected cervical cytology and human papillomavirus. Obstet Gynecol 102: 266–272.
Hesselink AT, Heideman DAM, Steenbergen RDM, Coupé VMH, Overmeer RM, Rijkaart D, Berkhof J, Meijer CJLM, Snijders PJF (2011) Combined promoter methylation analysis of CADM1 and MAL: an objective triage tool for high-risk human papillomavirus DNA-positive women. Clin Cancer Res 17: 2459–2465.
Hesselink AT, Heideman DAM, Steenbergen RDM, Gök M, van Kemenade FJ, Wilting SM, Berkhof J, Meijer CJLM, Snijders PJF (2014) Methylation marker analysis of self-sampled cervico-vaginal lavage specimens to triage high-risk HPV-positive women for colposcopy. Int J Cancer 135: 880–886.
Huang T-H, Lai H-C, Liu H-W, Lin CJ, Wang K-H, Ding D-C, Chu T-Y (2010) Quantitative analysis of methylation status of the PAX1 gene for detection of cervical cancer. Int J Gynecol Cancer 20: 513–519.
Insinga RP, Perez G, Wheeler CM, Koutsky LA, Garland SM, Leodolter S, Joura EA, Ferris DG, Steben M, Brown DR, Elbasha EH, Paavonen J, Haupt RM (2010) Incidence, duration, and reappearance of type-specific cervical human papillomavirus infections in young women. Cancer Epidemiol Biomarkers Prev 19: 1585–1594.
Jacobs MV, Snijders PJ, van den Brule AJ, Helmerhorst TJ, Meijer CJ, Walboomers JM (1997) A general primer GP5+/GP6(+)-mediated PCR-enzyme immunoassay method for rapid detection of 14 high-risk and 6 low-risk human papillomavirus genotypes in cervical scrapings. J Clin Microbiol 35: 791–795.
Jaisamrarn U, Castellsagué X, Garland SM, Naud P, Palmroth J, Del Rosario-Raymundo MR, Wheeler CM, Salmerón J, Chow S-N, Apter D, Teixeira JC, Skinner SR, Hedrick J, Szarewski A, Romanowski B, Aoki FY, Schwarz TF, Poppe WAJ, Bosch FX, de Carvalho NS, Germar MJ, Peters K, Paavonen J, Bozonnat M-C, Descamps D, Struyf F, Dubin GO, Rosillon D, Baril L (2013) Natural history of progression of HPV infection to cervical lesion or clearance: analysis of the control arm of the large, randomised PATRICIA study. PLoS One 8: e79260.
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33: 159–174.
Luttmer R, De Strooper LMA, Berkhof J, Snijders PJF, Dijkstra MG, Uijterwaal MH, Steenbergen RDM, Kemenade FJ, Van, Rozendaal L, Helmerhorst TJM, Verheijen RHM, Harmsel WA, Ter, Baal WM, Van, Heideman AM, Meijer CJLM (2016) Comparing the performance of FAM19A4 methylation analysis, cytology and HPV16/18 genotyping for the detection of cervical (pre)cancer in high-risk HPV-positive women of a gynecologic outpatient population (COMETH study). Int J Cancer 138: 992–1002.
Meijer CJLM, Berkhof J, Castle PE, Hesselink AT, Franco EL, Ronco G, Arbyn M, Bosch FX, Cuzick J, Dillner J, Heideman DAM, Snijders PJF (2009) Guidelines for human papillomavirus DNA test requirements for primary cervical cancer screening in women 30 years and older. Int J Cancer 124: 516–520.
Moriarty AT, Nayar R, Arnold T, Gearries L, Renshaw A, Thomas N, Souers R (2014) The Tahoe Study: bias in the interpretation of Papanicolaou test results when human papillomavirus status is known. Arch Pathol Lab Med 138: 1182–1185.
Nobbenhuis MAE, Helmerhorst TJM, van den Brule AJ, Rozendaal L, Jaspars LH, Voorhorst FJ, Verheijen RHM, Meijer CJLM (2002) Primary screening for high risk HPV by home obtained cervicovaginal lavage is an alternative screening tool for unscreened women. J Clin Pathol 55: 435–439.
Ogilvie GS, Patrick DM, Schulzer M, Sellors JW, Petric M, Chambers K, White R, FitzGerald JM (2005) Diagnostic accuracy of self collected vaginal specimens for human papillomavirus compared to clinician collected human papillomavirus specimens: a meta-analysis. Sex Transm Infect 81: 207–212.
Overmeer RM, Henken FE, Bierkens M, Wilting SM, Timmerman I, Meijer CJLM, Snijders PJF, Steenbergen RDM (2009) Repression of MAL tumour suppressor activity by promoter methylation during cervical carcinogenesis. J Pathol 219: 327–336.
Overmeer RM, Henken FE, Snijders PJF, Claassen-Kramer D, Berkhof J, Helmerhorst TJM, Heideman DAM, Wilting SM, Murakami Y, Ito A, Meijer CJLM, Steenbergen RDM (2008) Association between dense CADM1 promoter methylation and reduced protein expression in high-grade CIN and cervical SCC. J Pathol 215: 388–397.
Overmeer RM, Louwers JA, Meijer CJLM, van Kemenade FJ, Hesselink AT, Daalmeijer NF, Wilting SM, Heideman DAM, Verheijen RHM, Zaal A, van Baal WM, Berkhof J, Snijders PJF, Steenbergen RDM (2011) Combined CADM1 and MAL promoter methylation analysis to detect (pre-)malignant cervical lesions in high-risk HPV-positive women. Int J cancer 129: 2218–2225.
Rijkaart DC, Berkhof J, van Kemenade FJ, Coupé VMH, Hesselink AT, Rozendaal L, Heideman DAM, Verheijen RH, Bulk S, Verweij WM, Snijders PJF, Meijer CJLM (2012) Evaluation of 14 triage strategies for HPV DNA-positive women in population-based cervical screening. Int J Cancer 130: 602–610.
Ronco G, Dillner J, Elfström KM, Tunesi S, Snijders PJF, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi P, Berkhof J, Peto J, Meijer CJLM (2014) Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet 383: 524–532.
Saavedra KF, Brebi PS, Roa JC (2012) Epigenetics alterations in preneoplastic and neoplastic lesions of the cervix. Clin Epigenet 4: 13.
Schmitt M, Bravo IG, Snijders PJF, Pawlita M, Waterboer T, Gissmann L (2006) Bead-based multiplex genotyping of human papillomaviruses bead-based multiplex genotyping of human papillomaviruses. J Clin Microbiol 44: 504–512.
Snijders PJF, Steenbergen RDM, Heideman DAM, Meijer CJLM (2006) HPV-mediated cervical carcinogenesis: concepts and clinical implications. J Pathol 208: 152–164.
Snijders PJF, Verhoef VMJ, Arbyn M, Ogilvie G, Minozzi S, Banzi R, van Kemenade FJ, Heideman DAM, Meijer CJLM (2013) High-risk HPV testing on self-sampled versus clinician-collected specimens: a review on the clinical accuracy and impact on population attendance in cervical cancer screening. Int J Cancer 132: 2223–2236.
Steenbergen RDM, Kramer D, Braakhuis BJM, Stern PL, Verheijen RHM, Meijer CJLM, Snijders PJF (2004) TSLC1 gene silencing in cervical cancer cell lines and cervical neoplasia. J Natl Cancer Inst 96: 294–305.
Steenbergen RDM, Ongenaert M, Snellenberg S, Trooskens G, van der Meide WF, Pandey D, Bloushtain-Qimron N, Polyak K, CJLM Meijer, Snijders PJF, Van Criekinge W (2013) Methylation-specific digital karyotyping of HPV16E6E7 expressing human keratinocytes identifies novel methylation events in cervical carcinogenesis. J Pathol 231: 53–62.
Steenbergen RDM, Snijders PJF, Heideman DAM, Meijer CJLM (2014) Clinical implications of (epi)genetic changes in HPV-induced cervical precancerous lesions. Nat Rev Cancer 14: 395–405.
Tang N-S, Tang M-L, Chan ISF (2003) On tests of equivalence via non-unity relative risk for matched-pair design. Stat Med 22: 1217–1233.
Verhoef VMJ, Bosgraaf RP, van Kemenade FJ, Rozendaal L, Heideman DAM, Hesselink AT, Bekkers RLM, Steenbergen RDM, Massuger LFAG, Melchers WJG, Bulten J, Overbeek LIH, Berkhof J, Snijders PJF, Meijer CJLM (2014a) Triage by methylation-marker testing versus cytology in women who test HPV-positive on self-collected cervicovaginal specimens (PROHTECT-3): a randomised controlled non-inferiority trial. Lancet Oncol 15: 315–322.
Verhoef VMJ, Heideman DAM, van Kemenade FJ, Rozendaal L, Bosgraaf RP, Hesselink AT, Bekkers RLM, Massuger LFAG, Steenbergen RDM, Snijders PJF, Berkhof J, Meijer CJLM (2014b) Methylation marker analysis and HPV16/18 genotyping in high-risk HPV positive self-sampled specimens to identify women with high grade CIN or cervical cancer. Gynecol Oncol 135: 58–63.
Wilting SM, van Boerdonk RAA, Henken FE, CJLM Meijer, Diosdado B, Meijer GA, le Sage C, Agami R, Snijders PJF, Steenbergen RDM (2010) Methylation-mediated silencing and tumour suppressive function of hsa-miR-124 in cervical cancer. Mol Cancer 9: 167.
Winer RL, Lee S-K, Hughes JP, Adam DE, Kiviat NB, Koutsky LA (2003) Genital human papillomavirus infection: incidence and risk factors in a cohort of female university students. Am J Epidemiol 157: 218–226.
Zorzi M, Del Mistro A, Farruggio A, de’Bartolomeis L, Frayle-Salamanca H, Baboci L, Bertazzo A, Cocco P, Fedato C, Gennaro M, Marchi N, Penon MG, Cogo C, Ferro A (2013) Use of a high-risk human papillomavirus DNA test as the primary test in a cervical cancer screening programme: a population-based cohort study. BJOG 120: 1260–1267, ; discussion 1267–1268.
Acknowledgements
This study was supported by grants from the Dutch Cancer Society (KWF2014-7238 awarded to DAMH, PJFS and CJLMM), the European Research Council (ERC advanced 2012-AdG, proposal 322986 Mass-Care) awarded to CJLMM and the Seventh Framework Program of DG Research of the European Commission (CoheaHr-Health-F3-2013-603019) awarded to CJLMM and JB. We thank the women who participated in this study. In addition, we thank the technicians and research staff of the unit of Molecular Pathology of the Department of Pathology at the VU University Medical Center, especially Helma de Bruin, Marjolein Bekker-Lettink and Martijn Bogaarts. We also thank the administrative workers and information technology team of the Department of Pathology at the VU University Medical Center for the supportive work. We acknowledge the gynaecologists, nurses and administrative workers of the Gynaecology Departments of the VU University Medical Center, University Medical Center Utrecht, Onze Lieve Vrouwe Gasthuis West, Reinier de Graaf Group, Sint Antonius Hospital and Flevo Hospital for their contribution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
JB has played an advisory role for Merck and Roche, has been on the speakers bureau of Qiagen and has received a travel reimbursement from DDL Diagnostic Laboratory. PJFS has been on the speakers bureau of Roche, Qiagen, Abbott, Gen-Probe and Seegene. PJFS is consultant for Crucell Holland BV. TJMH, RHMV and WAH have been principal investigators of a GlaxoSmithKline sponsored study. WGVQ is a minority shareholder of Diassay BV and obtained grants from GlaxoSmithKline. DAMH has been on the speakers bureau of Hologic/Gen-Probe and serves occasionally on the scientific advisory boards of AMGEN and Pfizer. CJLMM has been on the sponsored speakers bureau of GlaxoSmithKline, Qiagen, Merck, Roche, Menarini and Segeene, and served on the scientific advisory board of GlaxoSmithKline, Qiagen, Merck and Roche. CJLMM has been consultant for Qiagen and Genticel and is a minority shareholder of Diassay BV. Formerly, CJLMM was a minority shareholder of Delphi Biosciences. CJLMM, PJFS, RDMS and DAMH have minority stake in Self-screen BV, a spin-off company of VU University Medical Center Amsterdam, which holds patents related to the present work. RL, LMADS, MGD, FJvK, LR, WMvB, GCMG, JWMS and DKEvD do not have any conflict of interest to declare.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Luttmer, R., De Strooper, L., Dijkstra, M. et al. FAM19A4 methylation analysis in self-samples compared with cervical scrapes for detecting cervical (pre)cancer in HPV-positive women. Br J Cancer 115, 579–587 (2016). https://doi.org/10.1038/bjc.2016.200
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2016.200
- Springer Nature Limited
Keywords
This article is cited by
-
Identification of a methylation panel as an alternative triage to detect CIN3+ in hrHPV-positive self-samples from the population-based cervical cancer screening programme
Clinical Epigenetics (2023)
-
DNA methylation as a triage marker for colposcopy referral in HPV-based cervical cancer screening: a systematic review and meta-analysis
Clinical Epigenetics (2023)
-
Evaluation of DNA methylation biomarkers ASCL1 and LHX8 on HPV-positive self-collected samples from primary HPV-based screening
British Journal of Cancer (2023)
-
Triage of human papillomavirus infected women by methylation analysis in first-void urine
Scientific Reports (2021)
-
Cervical cancer detection by DNA methylation analysis in urine
Scientific Reports (2019)